ACCP Cardiology PRN Journal Club Dr Courtney Montepara

ACCP Cardiology PRN Journal Club

Dr. Courtney Montepara • Dr. Montepara completed her Pharm. D at the Philadelphia College of Pharmacy. She then went on to complete her PGY 1 at Atlantic Health System in Morristown, NJ. She is currently a PGY 2 Cardiology Resident at the Cleveland Clinic. 2

Dr. Michael Brenner • Dr. Brenner a pharmacist at the VA Ann Arbor Healthcare System. He is currently the PGY 2 Cardiology Pharmacy Residency program director and an adjunct clinical assistant professor at the University of Michigan College of Pharmacy. Dr. Brenner completed his Pharm. D at the Massachusetts College of Pharmacy and then completed a PGY 1 at West Palm Beach VA Medical Center in Florida. His practice site is predominantly ambulatory care, focusing hypertension, heart failure, and arrhythmias. 3

Further Cardiovascular Outcomes Research with PCSK 9 Inhibition in Subjects with Elevated Risk (FOURIER) Courtney Montepara, Pharm. D. PGY 2 Cardiology Pharmacy Resident Cleveland Clinic, Cleveland, OH Michael Brenner, Pharm. D. , BCPS-AQ Cardiology Clinical Pharmacy Specialist Director, PGY 2 Cardiology Pharmacy Residency Program VA Ann Arbor Healthcare System, Ann Arbor, MI 4

Disclosure The speaker has nothing to disclose concerning possible financial or personal relationships with commercial entities that may have direct or indirect interest in the subject matter of this presentation. 5
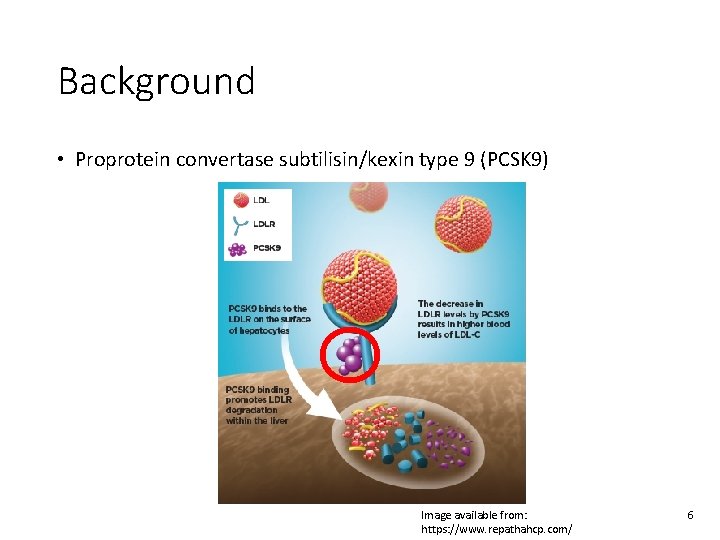
Background • Proprotein convertase subtilisin/kexin type 9 (PCSK 9) Image available from: https: //www. repathahcp. com/ 6
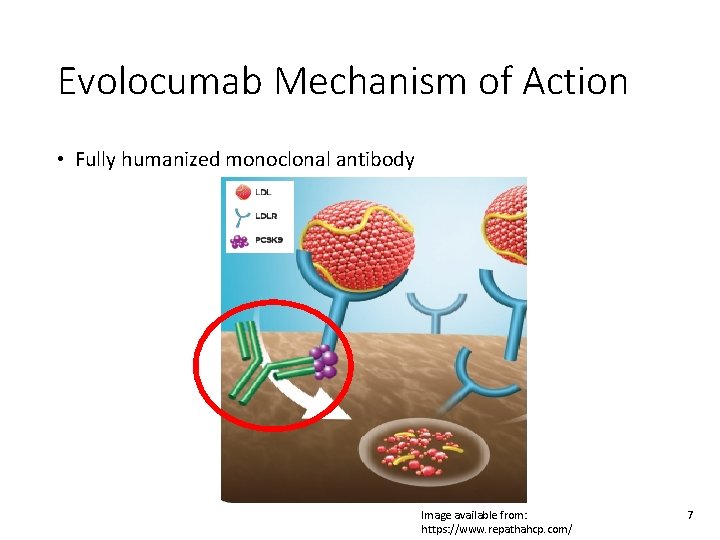
Evolocumab Mechanism of Action • Fully humanized monoclonal antibody Image available from: https: //www. repathahcp. com/ 7

Evolocumab (Repatha®) Indicated as an adjunct to diet and: • Maximally tolerated statin therapy for treatment of adults with He. FH or clinical ASCVD who require additional lowering of low density lipoprotein cholesterol (LDL-C) § 140 mg subcutaneously every 2 weeks OR 420 mg subcutaneously once monthly • Other LDL-lowering therapies (e. g. , statins, ezetimibe, LDL apheresis) in patients with Ho. FH who require additional lowering of LDL-C § 420 mg subcutaneously once monthly He. FH: heterozygous familial hypercholesterolemia Ho. FH: homozygous familial hypercholesterolemia ASCVD: atherosclerotic cardiovascular disease Repatha® [package insert]. Thousand Oaks, CA: Amgen; 2015. Images available from: https: //www. repathahcp. com/ 8

Previous Literature: OSLER-1 and 2 • Two open-label, randomized trials enrolled 4465 patients who had completed a phase 2 or 3 study of evolocumab • Patients were assigned to evolocumab (140 mg every 2 weeks or 420 mg monthly) plus standard therapy or standard therapy alone • Follow-up for median of 11. 1 months • Evolocumab plus standard therapy reduced LDL cholesterol by 61% versus standard therapy alone • Exploratory analysis showed that it was also associated with reduced incidence of cardiovascular events Sabatine MS, et al. N Engl J Med. 2015 Apr 16; 372(16): 1500 -9. 9

Sabatine MS, et al. N Engl J Med. 2017 Mar 17. [Epub ahead of print] 10

Study Objective To investigate the clinical efficacy and safety of evolocumab when added to high- or moderate-intensity statin therapy in patients with clinically evident atherosclerotic cardiovascular disease (ASCVD) 11

Eligibility Criteria Inclusion Criteria • • 40 -85 years of age Clinically evident ASCVD, defined as a history of myocardial infarction (MI), nonhemorrhagic stroke, or symptomatic peripheral artery disease (PAD) Fasting LDL-C ≥ 70 mg/d. L or a non-HDL-C ≥ 100 mg/d. L while taking an optimized regimen of lipid-lowering therapy, defined as at least atorvastatin 20 mg daily or its equivalent ± ezetimibe At least 1 major risk factor or at least 2 minor risk factors Exclusion Criteria • • MI or stroke within previous 4 weeks • Planned or expected cardiac surgery or revascularization within 3 months after randomization • • Uncontrolled hypertension • Severe renal dysfunction (e. GFR < 20 m. L/min/1. 73 m 2 at final screening) • Active liver disease or hepatic dysfunction (AST or ALT > 3 x ULN at final screening) NYHA class III or IV, or last known LVEF < 30% Known hemorrhagic stroke at any time Uncontrolled or recurrent ventricular tachycardia Prior use of PCSK 9 inhibition treatment other than evolocumab or use of evolocumab < 12 weeks prior to final lipid screening 12
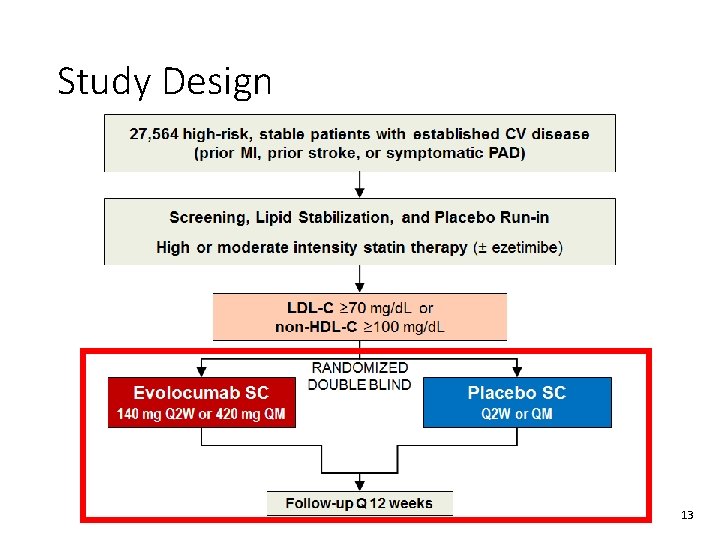
Study Design 13

Study Endpoints Primary Efficacy Endpoint • Major cardiovascular events, defined as the composite of cardiovascular death, MI, stroke, hospitalization for unstable angina, or coronary revascularization Key Secondary Efficacy Endpoint • Composite of cardiovascular death, MI, or stroke Safety Endpoints • Adverse events • Development of anti-evolocumab antibodies (binding and neutralizing) 14

Statistical Analysis • If the rate of the primary endpoint was significantly lower in the evolocumab group (P < 0. 05), the key secondary endpoint and then cardiovascular death were tested at a significance level of 0. 05 • Intention-to-treat • Estimated 1630 secondary endpoint events required to provide 90% power to detect a 15% relative risk reduction with evolocumab compared to placebo • Cox proportional-hazards model with stratification factors as covariates to determine hazard ratios and 95% confidence intervals • Log-rank tests to calculate P values for time-to-event analyses 15
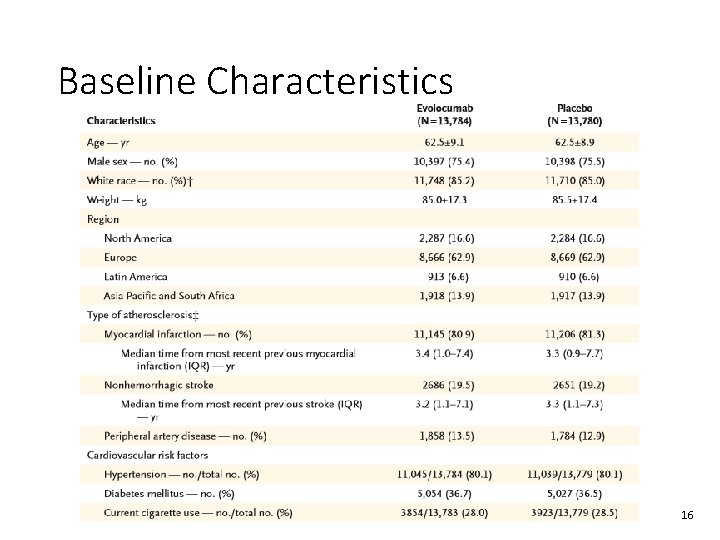
Baseline Characteristics 16
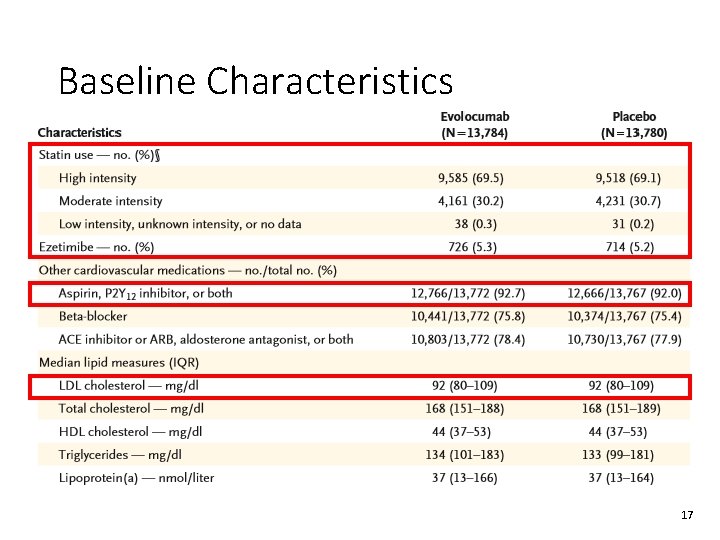
Baseline Characteristics 17
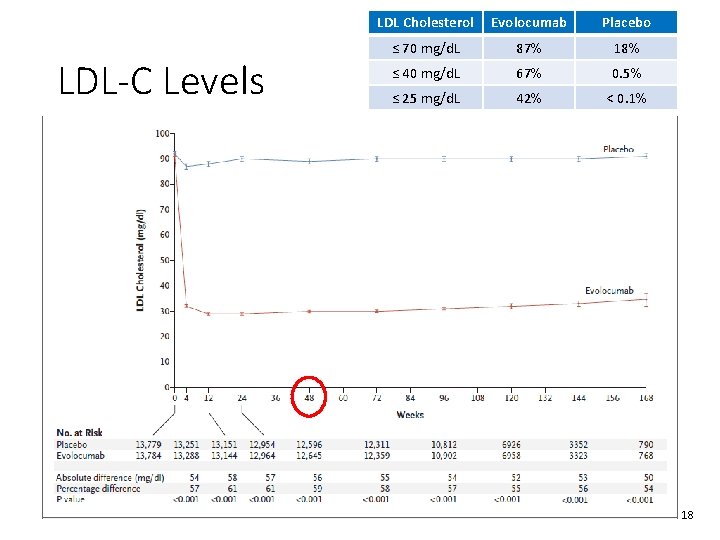
LDL-C Levels LDL Cholesterol Evolocumab Placebo ≤ 70 mg/d. L 87% 18% ≤ 40 mg/d. L 67% 0. 5% ≤ 25 mg/d. L 42% < 0. 1% 18

Primary and Secondary Endpoints NNT = 74 19
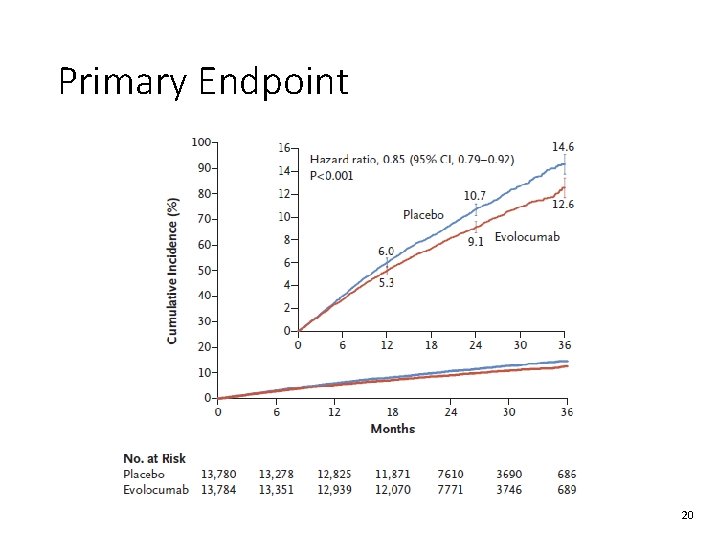
Primary Endpoint 20
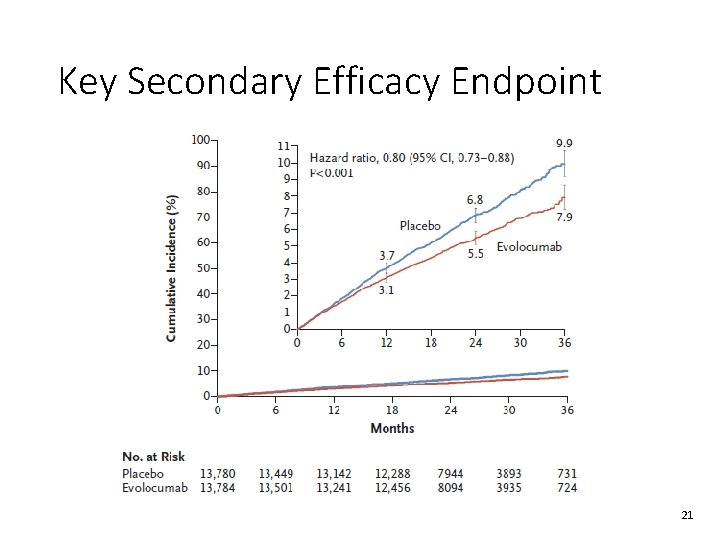
Key Secondary Efficacy Endpoint 21
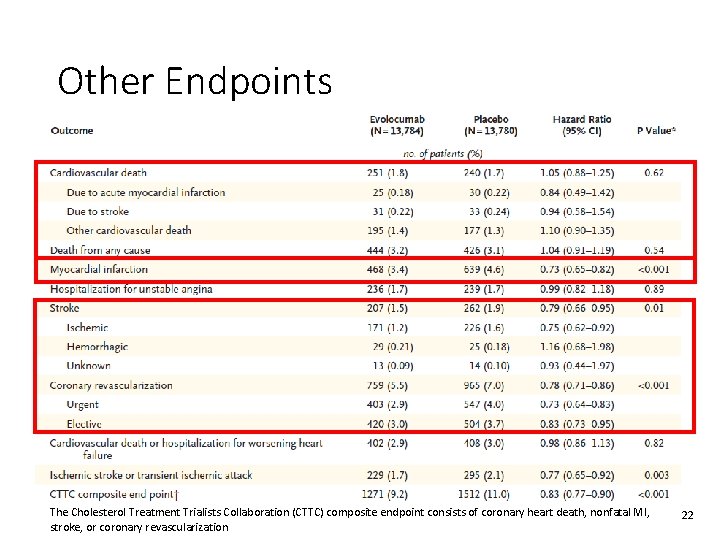
Other Endpoints The Cholesterol Treatment Trialists Collaboration (CTTC) composite endpoint consists of coronary heart death, nonfatal MI, stroke, or coronary revascularization 22
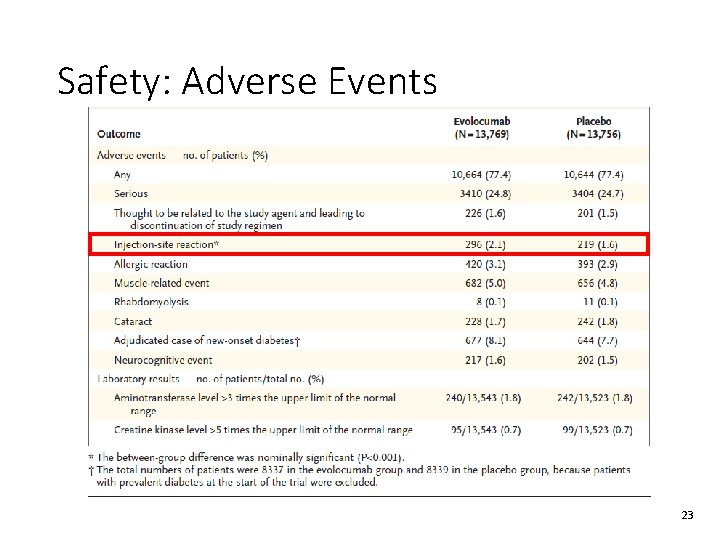
Safety: Adverse Events 23

Study Conclusions • When added to statin therapy, evolocumab lowered LDL cholesterol levels by 59% from baseline compared to placebo, from a median of 92 mg/d. L to 30 mg/d. L • risk of the primary composite endpoint by 15% and risk of the key secondary endpoint by 20% • Magnitude of risk reduction shown to increase over time • No effect of additional LDL-C lowering on cardiovascular death or all-cause mortality • Injection-site reactions were significantly higher in the evolocumab group compared to the placebo group 24

Study Critique • Strong study design • Short duration of follow-up 25

Cost • Evolocumab costs $14, 000 per year! • Institute for Clinical and Economic Review (ICER) has begun a “New Evidence Update” to incorporate the newly released data, including updated economic analyses and value-based price benchmarks of the PCSK 9 inhibitors • Amgen said if insurance companies loosen restrictions on coverage, they will refund the cost of evolocumab if patients have a heart attack or stroke while taking it Image available from: https: //www. repathahcp. com/ 26

Clinical Implications • Uses § Patients with familial hypercholesterolemia (FH) § Patients with hyperlipidemia not due to FH who are at elevated cardiovascular risk and either undertreated or cannot tolerate statin therapy • Prior authorizations/insurance approval • Compliance to dosing regimens • Administration technique education • Guideline changes? 27

Acknowledgments • Michael Brenner, Pharm. D. , BCPS-AQ Cardiology • Genevieve Hale, Pharm. D. , BCPS • Zachary Noel, Pharm. D. , BCPS • Thomas Szymanski, Pharm. D. Candidate 28

Further Cardiovascular Outcomes Research with PCSK 9 Inhibition in Subjects with Elevated Risk (FOURIER) Courtney Montepara, Pharm. D. PGY 2 Cardiology Pharmacy Resident Cleveland Clinic, Cleveland, OH Michael Brenner, Pharm. D. , BCPS-AQ Cardiology Clinical Pharmacy Specialist Director, PGY 2 Cardiology Pharmacy Residency Program VA Ann Arbor Healthcare System, Ann Arbor, MI 29

Announcements • Thank you for attending the ACCP Cardiology PRN Journal Club • There will be TWO journal clubs in April. The first will be on April 18 th at 3 PM EST. • For a copy of the slides and trial summary, please visit https: //accpcardsprnjournalclub. pbworks. com/ 30
- Slides: 30