Justin Davies MBBS Ph D Interventional Cardiologist Hammersmith

Justin Davies, MBBS, Ph. D Interventional Cardiologist Hammersmith Hospital, Imperial College London CRT, 2017

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Grant/Research Support Consulting Fees/Honoraria Royalty Income Intellectual Property Rights Company Volcano Philips

Ongoing studies using i. FR and FFR Study Countries Patients Endpoint summary Status SYNTAX II 1 The Netherlands, Poland, Spain, United Kingdom, Latvia 451 PCI with modern DES in 3 VD—applying SYNTAX score II, i. FR®/FFR and IVUS guidance to optimize DES Complete PROSPECT II 2 Sweden, Denmark, Norway 900 IVUS & NIRS (blinded) performed in culprit vessel(s) Successful PCI of all intended lesions by angiogram and FFR/i. FR® Recruiting MITNEC 3 Canada 450 Diagnostic yield of non-invasive tests vs FFR Performance of i. FR® vs FFR vs non-invasive tests Recruiting 10 prospective studies including >10, 000 patients enrolling with i. FR ORBITA 4 UK Estimated enrollment 200 Impact of PCI on Qo. L metrics vs OMT alone Ability of ‘Functional Gain’ to predict Qo. L gains Recruiting J-DEFINE 5 Japan Estimated enrollment 500 Practical use and events using ‘hybrid’ i. FR®/FFR Mismatch of non-invasive and angio w/ FFR/ IFR® Active, but not recruiting DEFINE-FLAIR 6 Europe, Asia, North America, Africa Estimated enrollment 2500 Non-inferiority of i. FR® vs FFR (stable, UA, ACS) Safety deferral of non-culprit during primary PCI Awaiting results Q 1 2017 i. FR SWEDEHEART 7 Denmark, Iceland, Sweden Estimated enrollment 2000 Non-inferiority of i. FR® vs FFR (stable, UA, ACS) Predictive value of post-PCI i. FR on outcomes Awaiting results Q 1 2017 DEFINE FLOW 8 Netherlands Estimated enrollment 450 Natural history of physiology (pressure and flow) Outcomes of CFR and FFR quadrants (MACE) Enrolling by invitation only DEFINE Dx 9 US, EU 2000 Appropriateness of patients with high risk factors to bypass nuclear and proceed to cath w/FFR Protocol development DEFINE PAD 9 US 300 Ability of FFR to predict event rates following PAD stenting and atherectomy Protocol development 1. SYNTAX II. http: //www. ecri-trials. com/studies/syntax-ii/participating-sites/. Accessed December 8 2016; 2. PROSPECT II, available at: https: //clinicaltrials. gov/ct 2/show/NCT 02171065. Accessed December 8, 2016; 3. MITNEC. https: //clinicaltrials. gov/ct 2/show/NCT 01972360. Accessed December 8, 2016; 4. ORBITA. https: //clinicaltrials. gov/ct 2/show/NCT 02062593. Accessed December 2016; 5. J-DEFINE. https: //clinicaltrials. gov/ct 2/show/NCT 02002910. Accessed December 8, 2016; 6. DEFINE-FLAIR. https: //clinicaltrials. gov/ct 2/show/NCT 02053038? term=DEFINE+FLAIR&rank=1. December 8, 2016; 7. i. FR SWEDEHEART. https: //clinicaltrials. gov/ct 2/show/NCT 02166736? term=ifr+swedeheart&rank=1. Accessed December 8, 2016; 8. DEFINE FLOW. https: //clinicaltrials. gov/ct 2/show/NCT 02328820? term=define+flow&rank=1. Accessed December 8, 2016; 9. Philips data on file 3
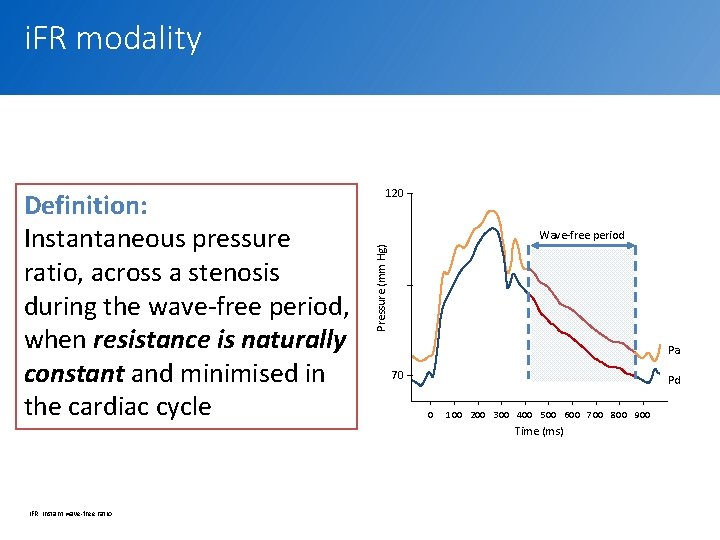
i. FR modality Wave-free period Pressure (mm Hg) Definition: Instantaneous pressure ratio, across a stenosis during the wave-free period, when resistance is naturally constant and minimised in the cardiac cycle 120 Pa 70 Pd 0 100 200 300 400 500 600 700 800 900 Time (ms) i. FR, instant wave-free ratio 4

Potential benefits of using i. FR • Improve patient experience 1, 2 • Avoid off-label use of adenosine in USA • Time savings 1 • Costs savings 1 • Avoid rare but serious complications 1 Under assessment in (1) DEFINE-FLAIR, (2) i. FR-Swede. Heart studies
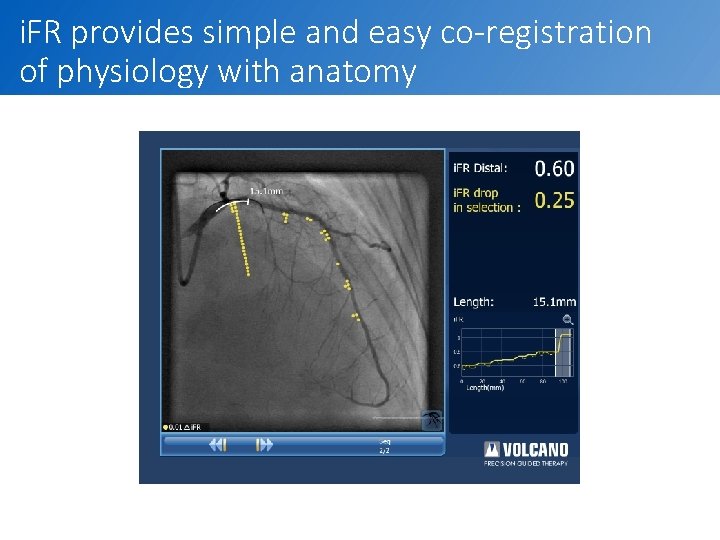
i. FR provides simple and easy co-registration of physiology with anatomy
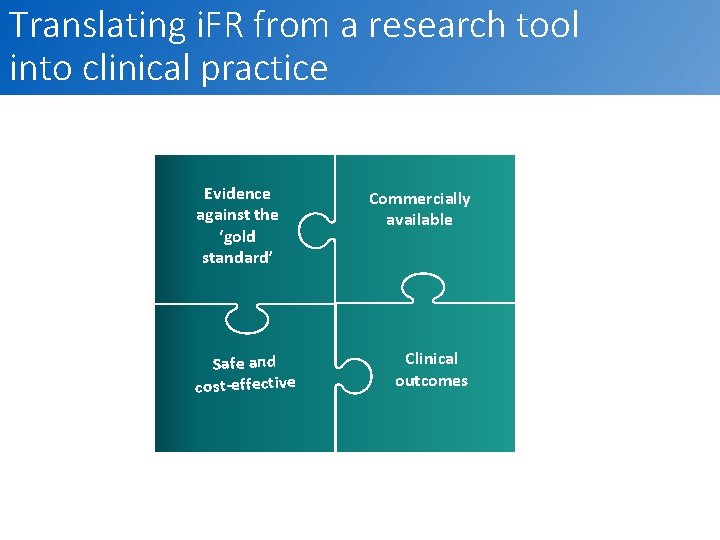
Translating i. FR from a research tool into clinical practice Evidence against the ‘gold standard’ Safe and cost-effective Commercially available Clinical outcomes 7
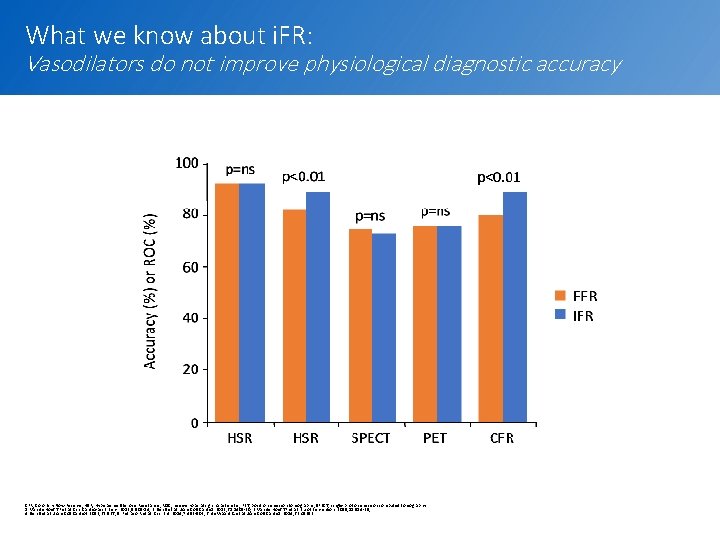
What we know about i. FR: Vasodilators do not improve physiological diagnostic accuracy CFR, Coronary Flow Reserve; HSR, Hyperaemic Stenosis Resistance; ROC, receiver-operating characteristic; PET, positron emission tomography; SPECT, single-photon emission computed tomography 1. Van de Hoef TP et al. Circ Cardiovasc Interv. 2012; 5: 508 -14; 2. Sen S et al. J Am Coll Cardiol. 2013; 61: 1409 -20; 3. Van de Hoef TP et al. Euro. Intervention. 2015; 11: 914 -25; 4. Sen S et al. J Am Coll Cardiol. 2013; 62: 566; 5. Petraco R et al. Circ. Int. 2014; 7: 492 -502; 6. de Waard G et al. J Am Coll Cardiol. 2014; 63: A 1692. 8

Why are we still using adenosine for physiological assessment?

Adenosine, a significant barrier to FFR adoption “If only adenosine were not so : difficult to mix, costly, time consuming, uncomfortable for the patient, and (rarely) potentially dangerous…. FFR would be easier to perform and therefore would be used more often” • M Kern, On the search for an “easy” FFR: Submaximal hyperemia and NTG-induced translesional pressure drop (Pd/Pa-NTG), Cathet. Cardiovasc. Intervent. February 1, 2016

Leadership Team Javier Escaned Co-Principal Investigator Clinico San Carlos, Madrid Justin Davies Co-Principal Investigator Imperial College, London

Leadership Team Patrick Serruys Co-Chairman Imperial College, London Manesh Patel Co-Chairman DCRI, Duke
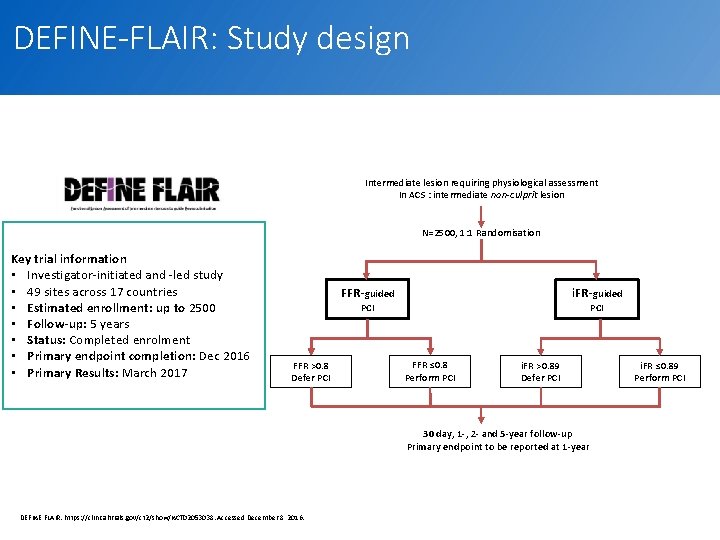
DEFINE-FLAIR: Study design Intermediate lesion requiring physiological assessment In ACS : intermediate non-culprit lesion N=2500, 1: 1 Randomisation Key trial information • Investigator-initiated and -led study • 49 sites across 17 countries • Estimated enrollment: up to 2500 • Follow-up: 5 years • Status: Completed enrolment • Primary endpoint completion: Dec 2016 • Primary Results: March 2017 FFR-guided i. FR-guided PCI FFR >0. 8 Defer PCI FFR ≤ 0. 8 Perform PCI i. FR >0. 89 Defer PCI i. FR ≤ 0. 89 Perform PCI 30 day, 1 -, 2 - and 5 -year follow-up Primary endpoint to be reported at 1 -year DEFINE FLAIR. https: //clinicaltrials. gov/ct 2/show/NCT 02053038. Accessed December 8, 2016. 13
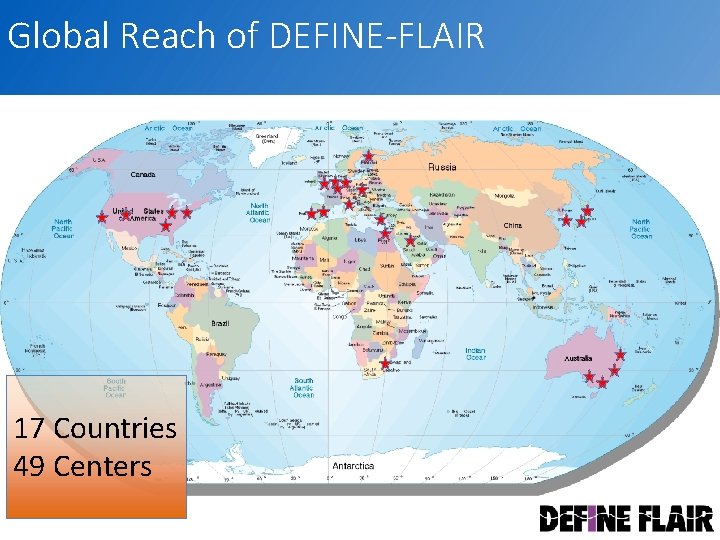
Global Reach of DEFINE-FLAIR 17 Countries 49 Centers

DEFINE-FLAIR: Study hypotheses and aims Study hypotheses In patients with coronary stenoses suitable for physiological assessment with FFR, the decision to perform coronary revascularisation based on i. FR measurements is: • Non-inferior to FFR in terms of safety and efficacy • Superior to FFR in terms of cost-effectiveness Aim • Comparison of clinical outcomes of patients whose treatment has been guided by i. FR vs those whose treatment has been guided by FFR If both study hypotheses are confirmed • i. FR could be used in a similar manner to FFR for the assessment of physiological stenosis severity • i. FR could lead to improved healthcare cost efficiencies DEFINE FLAIR. https: //clinicaltrials. gov/ct 2/show/NCT 02053038. Accessed December 8, 2016. 15

DEFINE-FLAIR: Endpoints Primary outcome • Major adverse cardiac events* in the i. FR and FFR groups at year 1 Secondary endpoints • Death (all cause) at 30 days, 1, 2 and 5 years • Death (cardiovascular) at 30 days, 1, 2 and 5 years • MI at 30 days, 1, 2 and 5 years • Repeat revascularisation by PCI or CABG at 30 days, 1, 2 and 5 years • Costs associated with i. FR or FFR guidance • Quality of life in patients included in the i. FR or FFR guidance groups *Major adverse cardiovascular events, defined as a combined endpoint of death, non-fatal myocardial infarction, or unplanned revascularisation. CABG, coronary artery bypass graft; MI, myocardial infarction. DEFINE FLAIR. https: //clinicaltrials. gov/ct 2/show/NCT 02053038. Accessed December 8, 2016. 16

Study Design – Key Features • Strategy study • Robust non-inferiority design • Margin similar to Noble/Excel/Syntax • Similar inclusion and exclusion to FAME/FAME 2 • Clinically relevant patient distributions (7080% in “critical intermediate zone”)

Study Design – Key Features • Blinding (patients/follow-up teams) • Core lab analysis of physiological screenshots • Monitoring 100% sites • Independent international clinical events committee
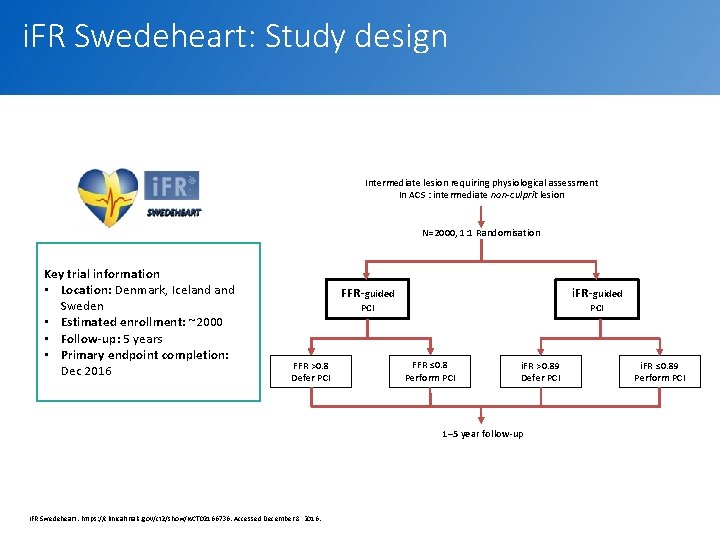
i. FR Swedeheart: Study design Intermediate lesion requiring physiological assessment In ACS : intermediate non-culprit lesion N=2000, 1: 1 Randomisation Key trial information • Location: Denmark, Iceland Sweden • Estimated enrollment: ~2000 • Follow-up: 5 years • Primary endpoint completion: Dec 2016 FFR-guided i. FR-guided PCI FFR >0. 8 Defer PCI FFR ≤ 0. 8 Perform PCI i. FR >0. 89 Defer PCI i. FR ≤ 0. 89 Perform PCI 1– 5 year follow-up i. FR Swedeheart. https: //clinicaltrials. gov/ct 2/show/NCT 02166736. Accessed December 8, 2016. 19
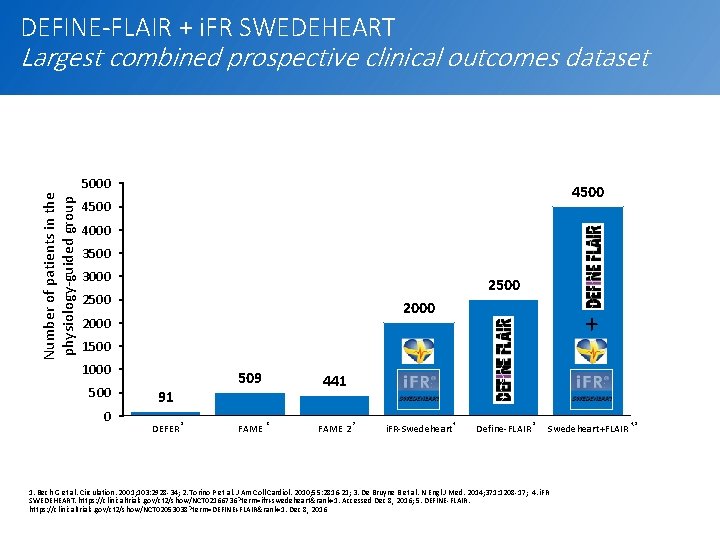
DEFINE-FLAIR + i. FR SWEDEHEART Number of patients in the physiology-guided group Largest combined prospective clinical outcomes dataset 5000 4500 4000 3500 3000 2500 2000 + 1500 1000 500 0 509 91 DEFER 1 FAME 441 2 FAME 2 3 i. FR-Swedeheart 4 Define-FLAIR 5 Swedeheart+FLAIR 1. Bech G et al. Circulation. 2001; 103: 2928 -34; 2. Torino P et al. J Am Coll Cardiol. 2010; 55: 2816 -21; 3. De Bruyne B et al. N Engl J Med. 2014; 371: 1208 -17; 4. i. FR SWEDEHEART. https: //clinicaltrials. gov/ct 2/show/NCT 02166736? term=ifr+swedeheart&rank=1. Accessed Dec 8, 2016; 5. DEFINE-FLAIR. https: //clinicaltrials. gov/ct 2/show/NCT 02053038? term=DEFINE+FLAIR&rank=1. Dec 8, 2016 4, 5

Main Arena (Hall D) Washington DC 18 th March, 11 am

Late Breaking Clinical Trial Presentations Main Arena (Hall D) Washington DC 18 th March, 11 am

Increased awareness and uptake of i. FR technology The great IFR vs. FFR debate: why sometimes “the wait and see approach” is the best tactic as the best pragmatic solution will always emerge and become established Patrick W. Serruys, Editor-in-Chief i. FR used in >4, 200 labs around the world (as of Jan 2017) Serruys PW. Euro. Intervention. 2013; 9: 11 -3 23
- Slides: 23