8 3 The Concentration of Solutions What is














- Slides: 15

8. 3 The Concentration of Solutions

What is Concentration? • Concentration tell us the amount of solute per quantity of solvent.

Concentration Units • There are many units of measurement for concentration. 1. 2. 3. 4. 5. 6. 7. Mass/volume Mass/mass Volume/volume Parts per million Parts per billions Molar Concentration Solubility Concentration

1. Mass/Volume (m/v) • This quantity gives the mass of the solute dissolved in a volume of solution expressed as a percent. Mass/volume Percent = Mass of solute (g) x 100% Volume of Solution (m. L)

Example • A pharmacist adds 2. 00 m. L of distilled water to 4. 00 g of a powder drug. The final volume of the solution is 3. 00 m. L. What is the concentration of the drug in g/100 m. L of solution. What is the (m/v) of the solution? % m/v = Mass of solute (g) x 100% Volume of Solution (m. L) = 4. 00 g / 3. 00 m. L X 100% = 133%

2. Mass/mass (m/m) • Gives the mass of solute dissolved in a mass of solution and is expressed as a percent. • This is used for alloys, example stainless steel Fe (78%), Cr (15%), Ni (7%) Mass/mass Percent = Mass of solute (g) x 100% Mass of Solution (g)

Example • Calcium chloride, Ca. Cl 2 can be used instead of road salt to melt ice. A student collected a sample 23. 47 g sample of slush and allowed it to melt. The residue that was left had a mass of 4. 58 g (assume its only Ca. Cl 2). What is the mass/mass percentage of calcium chloride in the slush? X g = 4. 58 g 100 g 23. 47 g X = 19. 5 g or 19. 5 % Ca. Cl 2

3. Volume/volume (v/v) • When mixing 2 liquids together to form a solution, it is easier to measure their volumes rather than their masses. Volume/volume %= Volume of solute (m. L) x 100% Volume of Solution (m. L)

Example • Rubbing alcohol is commonly used as an antiseptic for small cuts. It is sold as 70% v/v solution of isopropyl alcohol in water. What is the volume of isopropyl alcohol used to make 500 m. L of rubbing alcohol? v/v %= Volume of solute (m. L) x 100% Volume of Solution (m. L) 70 % = Volume of solute (m. L) x 100% 500 m. L Volume of solute (m. L) = 70% 500 m. L = 350 m. L is needed 100%

4. Parts per million (ppm) and 5. Parts per billion (ppb) • The concentration of a very small quantity of a substance in the human body or the environment is usually expressing in ppm (or ppb, next slide)

ppm Calculations ppm = Mass of solute x 106 Mass of Solution • Or Mass of solute = Mass of Solution x (g) 106 g of solution

ppb Calculations ppb = Mass of solute x 109 Mass of Solution • Or Mass of solute = Mass of Solution x (g) 109 g of solution

6. Molar Concentration • The most useful and widely used in chemistry • Molar concentration is the number of moles of solute that can dissolve in 1 L of solution

• Is this a molar solution? Ouch!!

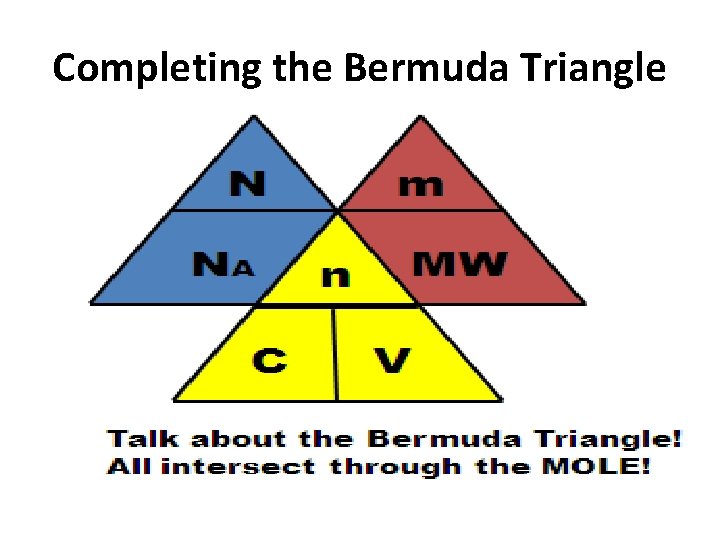
Completing the Bermuda Triangle