Concentration of Solutions Concentration Amount of a substance

Concentration of Solutions : Concentration: Amount of a substance per (solute). defined space. (solution or solvent) Percent by mass: Is the ratio of the solute’s mass to the solution’s mass expressed as a percent. Percent by mass = X 100

Example # 1: Calculate the percent by mass of solute in 55 g of solution with 15 g of potassium nitrate dissolved in water. Data: Solute’s mass = 15 g Solution’s mass = 55 g % by mass = X 100 % by mass = ? = X 100 = 27 % of KNO 3

Example # 2: Calculate the percent by mass of solute in a solution made by 25 g of sodium chloride & 50 g of water. Data: Solute’s mass = 25 g Solvent’s mass = 50 g Solution’s mass = 50 g + 25 g = 75 g % by mass = ? X 100 % by mass = = X 100 = 33 % of Na. Cl

Solution Concentration: % by volume = X 100 Example: Calculate the percent by volume of alcohol in 95 m. L of solution which contains 30 m. L of alcohol. Data: Solute = 30 m. L Solution = 95 m. L % by volume = ? X 100 % by volume = = X 100 = 32 % of alcohol

Mole Fraction: Is the ratio between the # of moles of each component & the total # of moles in the solution. ΧA = Mole fraction of A n. A = Moles of A n. B = Moles of B
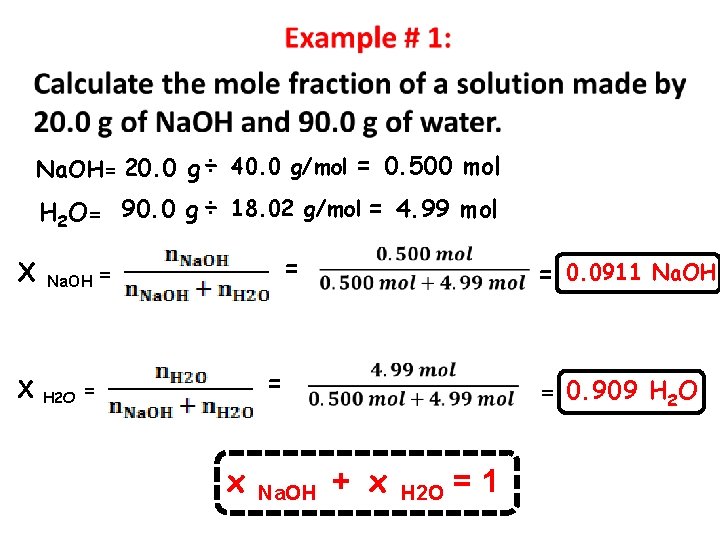
Na. OH= 20. 0 g ÷ 40. 0 g/mol = 0. 500 mol H 2 O= 90. 0 g ÷ 18. 02 g/mol = 4. 99 mol Χ Na. OH = Χ H 2 O = = = x Na. OH + x H 2 O =1 = 0. 0911 Na. OH = 0. 909 H 2 O
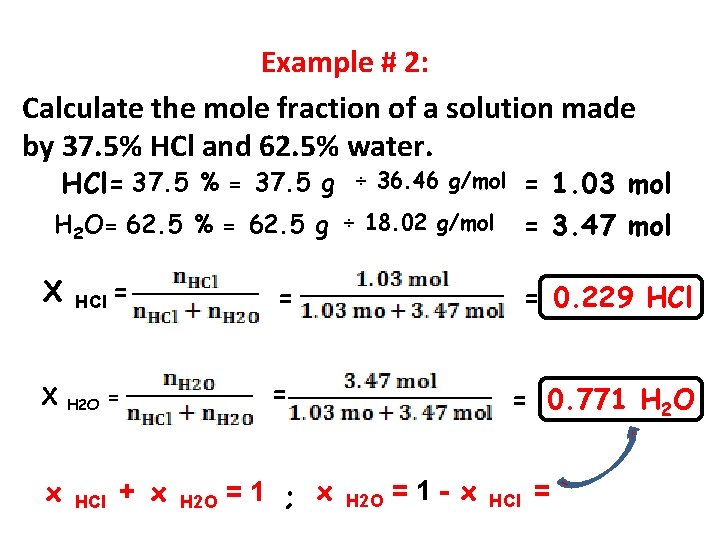
Example # 2: Calculate the mole fraction of a solution made by 37. 5% HCl and 62. 5% water. HCl= 37. 5 % = 37. 5 g = 1. 03 mol = 3. 47 mol ÷ 36. 46 g/mol H 2 O= 62. 5 % = 62. 5 g ÷ 18. 02 g/mol Χ Χ x HCl = H 2 O HCl = + x H 2 O = = 0. 229 HCl = = 0. 771 H 2 O =1 ; x H 2 O =1 - x HCl =
- Slides: 7