When and who In 1869 Russian Chemist Dimitri


















- Slides: 36


When and who? • In 1869 Russian Chemist Dimitri Mendeleev and German chemist Lothar Meyer published nearly identical ways of classifying

• But Mendeleev is generally more credited with the 1 st periodic table for 2 reasons: • He published first • He was better at explaining it than Meyer

• Mendeleev also saw the “periods” • Credited with publishing the first “periodic table”

Mendeleev got lots of credit because he left gaps for missing elements!

How was the periodic table of elements categorized at that time? • By increasing molar mass

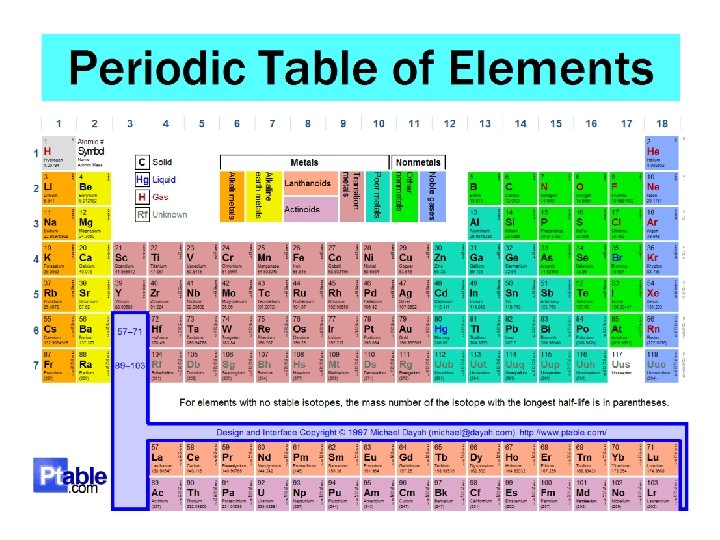
Periodic Law • When the elements are arranged in increasing order by their atomic numbers, their properties repeat periodically

The Modern Periodic Table • Organized in order of increasing atomic number

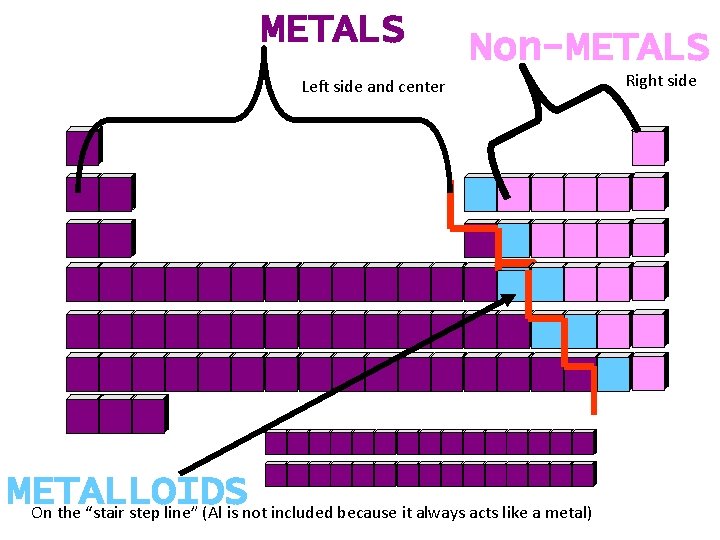
METALS Non-METALS Left side and center METALLOIDS On the “stair step line” (Al is not included because it always acts like a metal) Right side

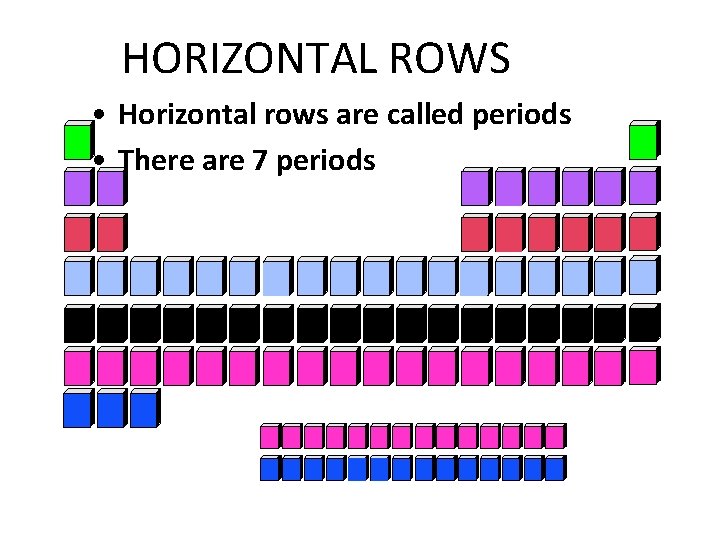
HORIZONTAL ROWS • Horizontal rows are called periods • There are 7 periods

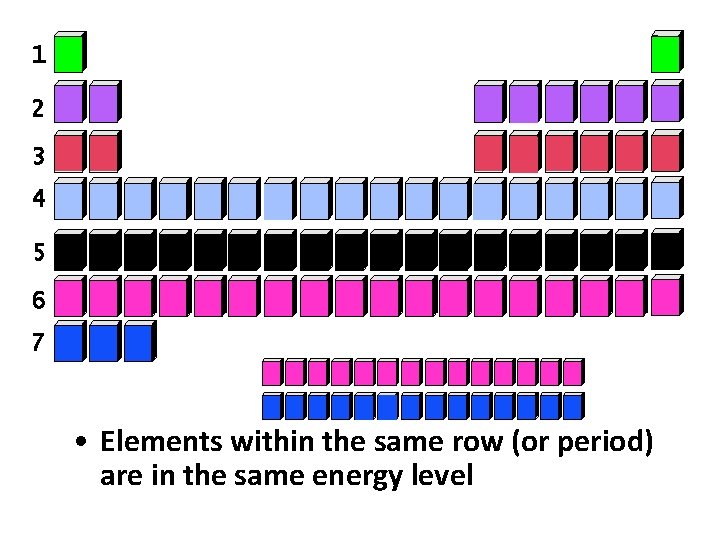
What do all of the elements in any given period have in common?

1 2 3 4 5 6 7 • Elements within the same row (or period) are in the same energy level

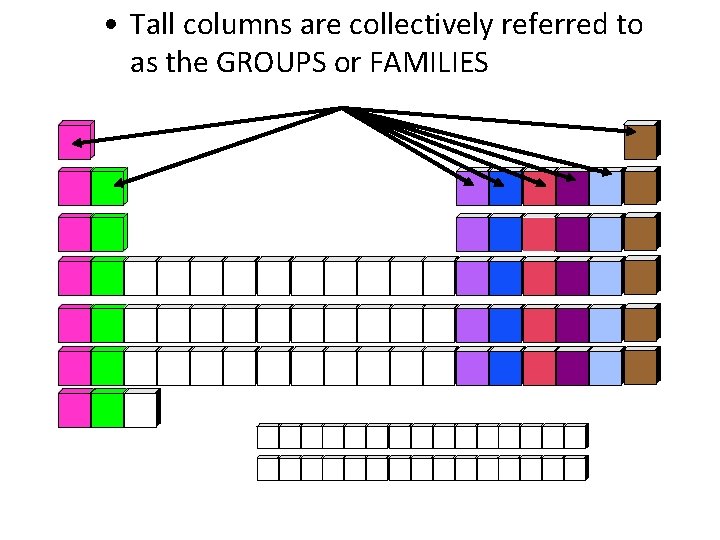
• Tall columns are collectively referred to as the GROUPS or FAMILIES

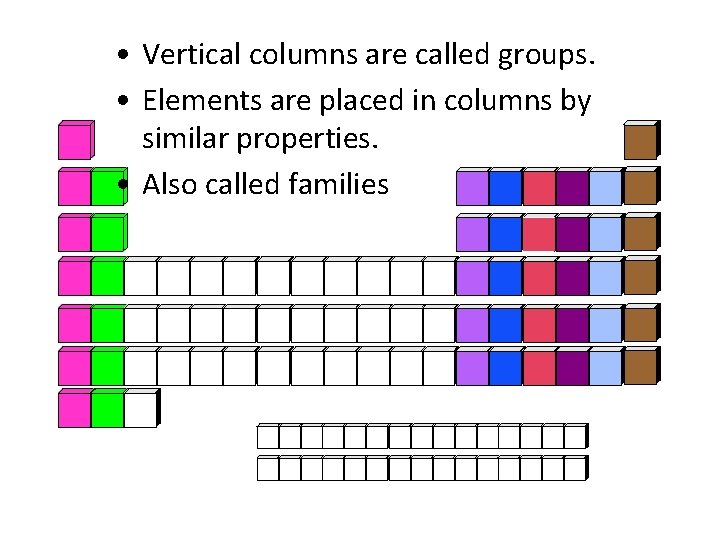
What are similarities between elements of the same group? • Same number of valence electrons • Same ending configuration, but different energy levels • Same charge and same chemical behavior

• Vertical columns are called groups. • Elements are placed in columns by similar properties. • Also called families

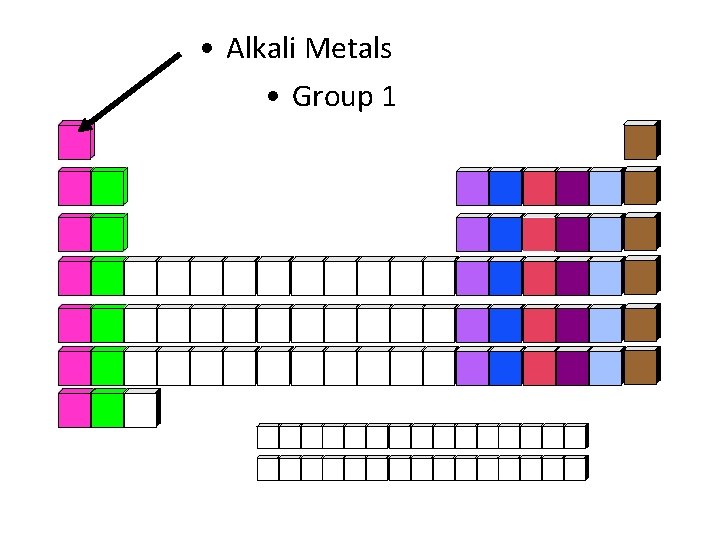
• Alkali Metals • Group 1

Alkali Metals • Elements: Li, Na, K, Rb, Cs, Fr • Chemical Properties: most highly reactive metals; stored in oil • Physical Properties: metals, silver gray, soft, have a low density (light weight) • Charge of Ions: +1 • Reaction with water: 2 x + H 2 O 2 x. OH + H 2

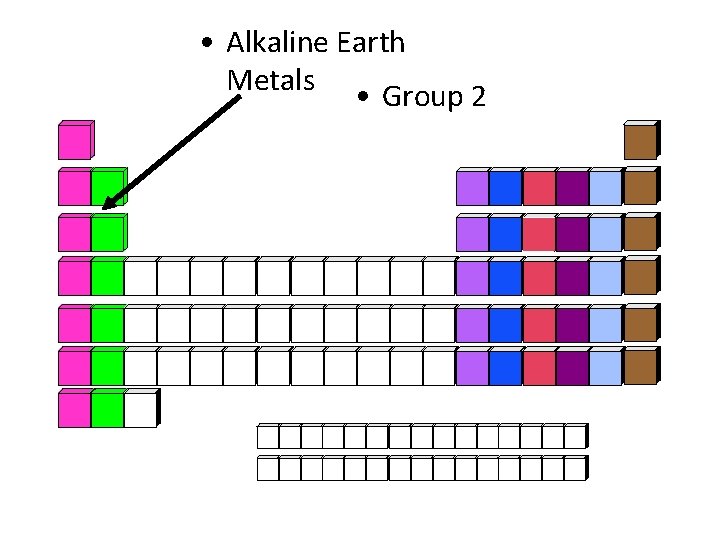
• Alkaline Earth Metals • Group 2

Alkaline Earth Metals • Elements: Be, Mg, Ca, Sr, Ba, Ra • Chemical Properties: extremely reactive; kept in oil • Physical Properties: shiny metals, gray silver • Charge of Ions: +2 • End of Electron Configuration: s 2

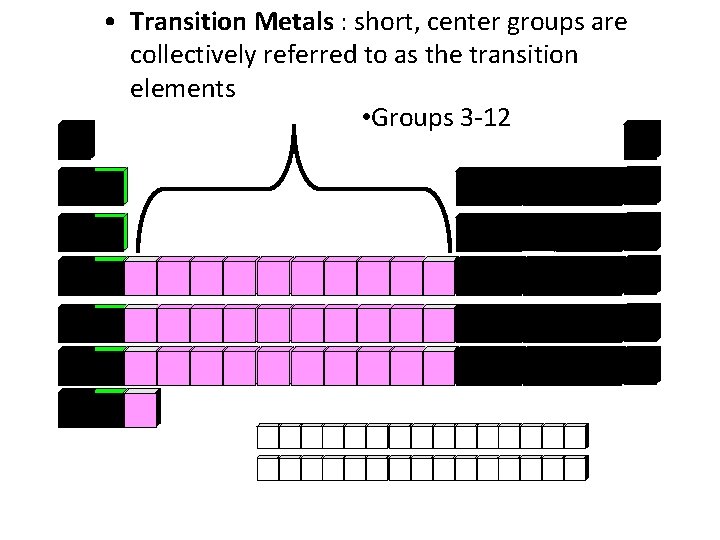
• Transition Metals : short, center groups are collectively referred to as the transition elements • Groups 3 -12

Transition Metals • They are given because they were believed to be chemically between metals and nonmetals • Some common elements: Zn, Au, Mn, Se, Ti, Rn • Charge of Ions: Variable • End of Electron Configuration: d

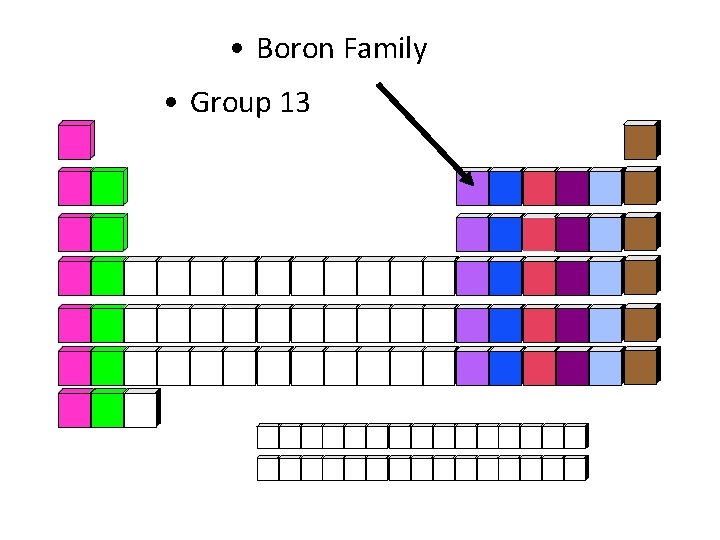
• Boron Family • Group 13

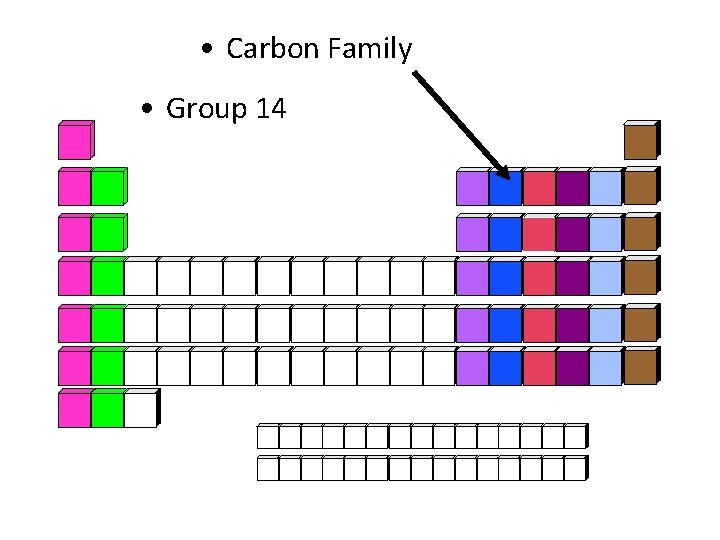
• Carbon Family • Group 14

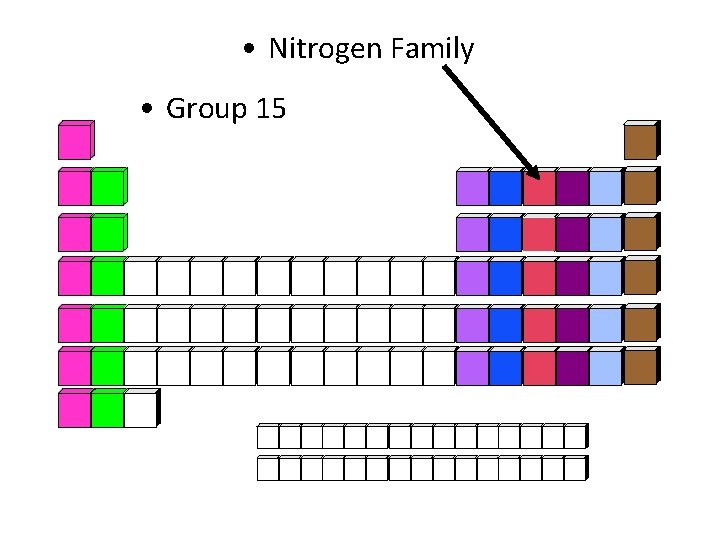
• Nitrogen Family • Group 15

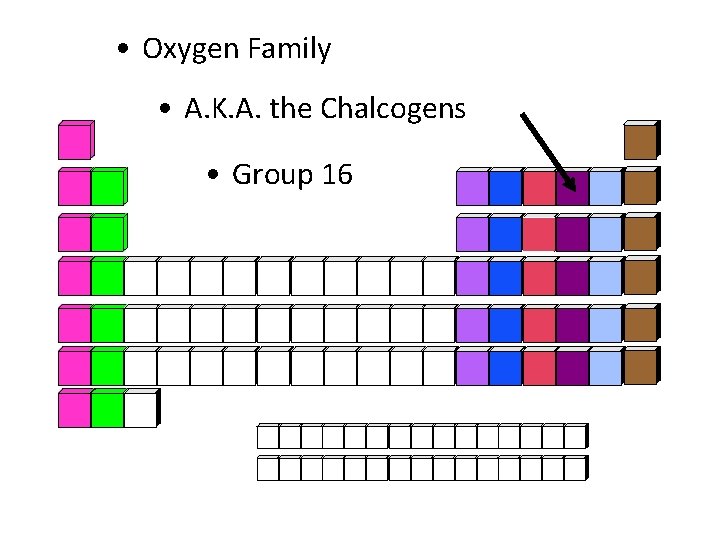
• Oxygen Family • A. K. A. the Chalcogens • Group 16

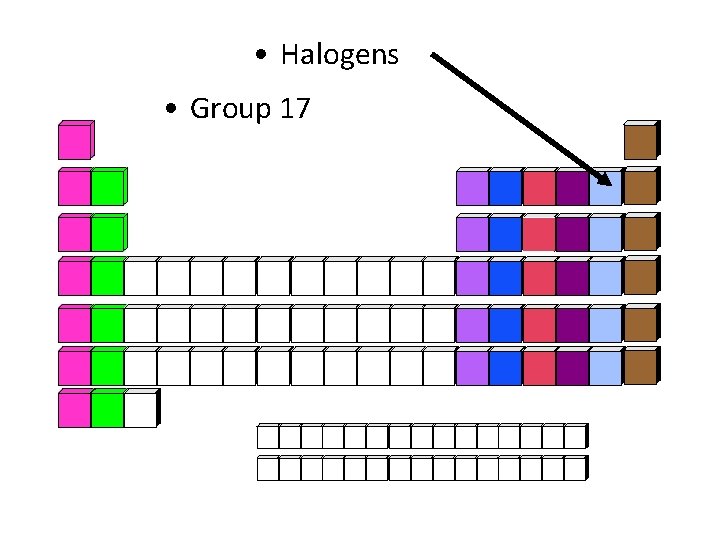
• Halogens • Group 17

Halogens • • Elements: F, Cl, Br, I, At Chemical Properties: most reactive non metals Charge of Ions: -1 End of Electron Configuration: p 5

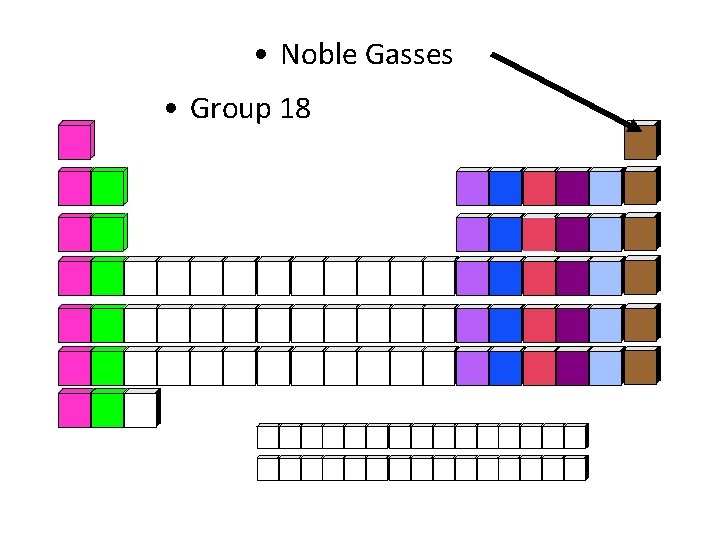
• Noble Gasses • Group 18

Noble Gases • Elements: He, Ne, Ar, Kr, Xe, Rn • Chemical Properties: very un-reactive; do not want to bond with anything • Charge of Ions: 0 • End of Electron Configuration: p 6 • Former Name: Inert Gases • Why is this former name inappropriate to use? Because under specialized conditions, noble gases are able to bond with other elements

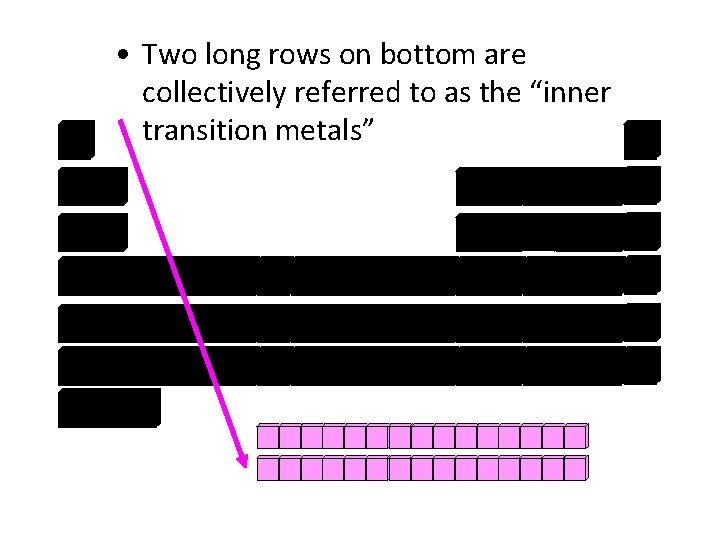
• Two long rows on bottom are collectively referred to as the “inner transition metals”

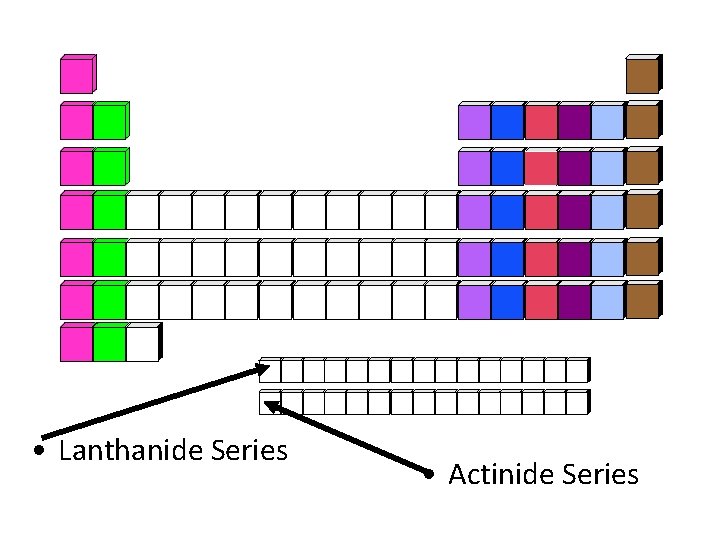
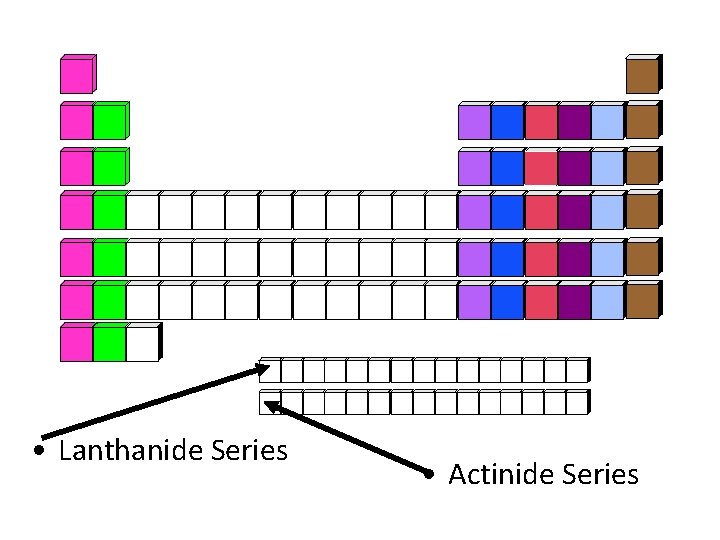
• Lanthanide Series • Actinide Series

Lanthanide Series • Elements Atomic # 58 - 71 • Elements: Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu • Located: first of the two rows below the periodic table • Also classified as part of the inner transition metals • Formerly named: Rare earth metals • Inappropriate name because not all of them are rare

• Lanthanide Series • Actinide Series

Actinide Series • Elements Atomic # 90 - 103 • Elements: Th, Pa, U, Np, Pu, Am, Cm, Bk, Cf, Es, Fm, Md, No, Lr • Located: second of the two rows below the periodic table • Also classified as part of the inner transition

Transuranium Elements • Lab created elements • Include elements 93 and higher

Using your own color scheme, color each group of the periodic table. Be sure to include a KEY Groups to label: Transition metals, Noble Gases, Alkali Metals, Lanthanide Series, Halogens, Alkaline Earth Metals, Actinide Series