The Father of the Periodic Table Dimitri Mendeleev

The Father of the Periodic Table Dimitri Mendeleev ________was the first scientist to elements notice the relationship between the _____ atomic _______ mass Arranged his periodic table by ____ unknown Said properties of ______elements could be predicted by the properties of elements around the missing element Moseley _____later discovered that the periodic nature of the elements was associated with_____ atomic _____, number not atomic mass
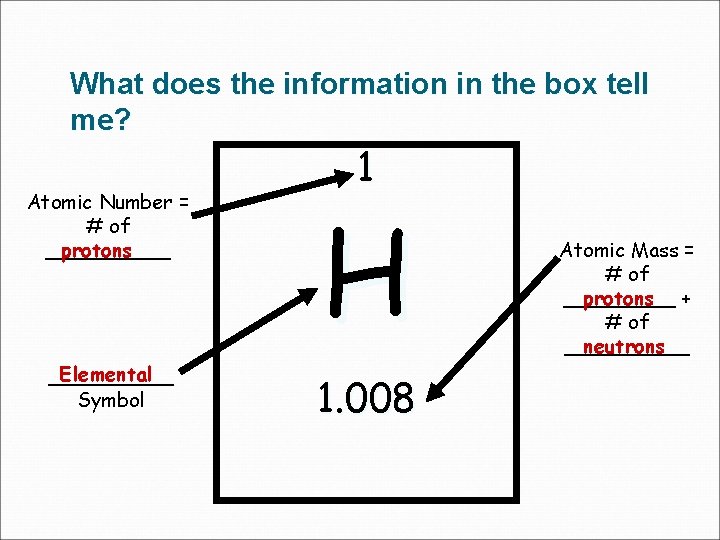
What does the information in the box tell me? 1 Atomic Number = # of _____ protons H Elemental _____ Symbol 1. 008 Atomic Mass = # of _____ protons + # of _____ neutrons
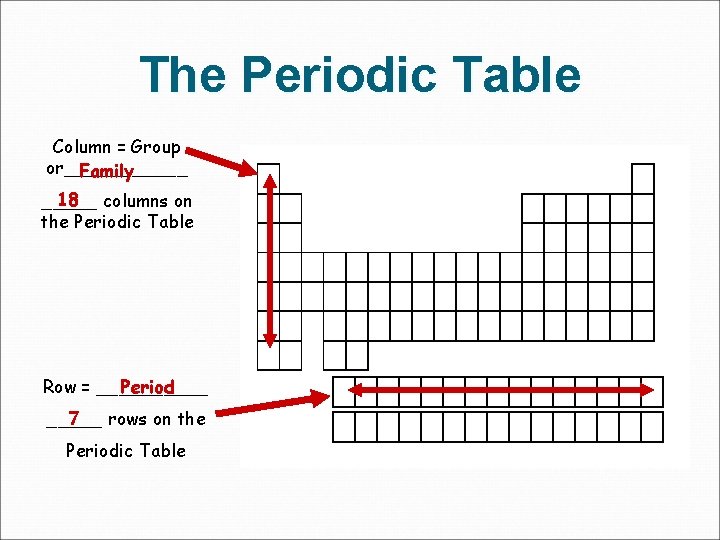
The Periodic Table Column = Group or______ Family 18 columns on _____ the Periodic Table Row = _____ Period 7 _____ rows on the Periodic Table
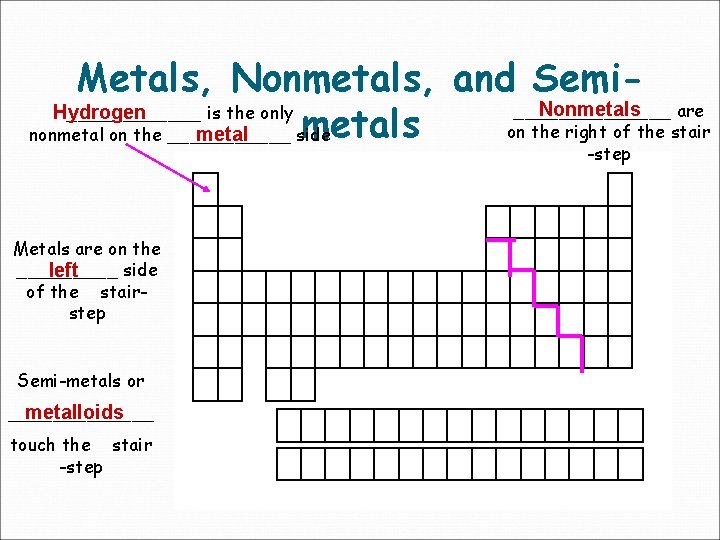
Metals, Nonmetals, and Semi. Nonmetals _______ are Hydrogen ______ is the only metals on the right of the stair nonmetal on the ______ side metal -step Metals are on the _____ side left of the stairstep Semi-metals or metalloids _______ touch the stair -step
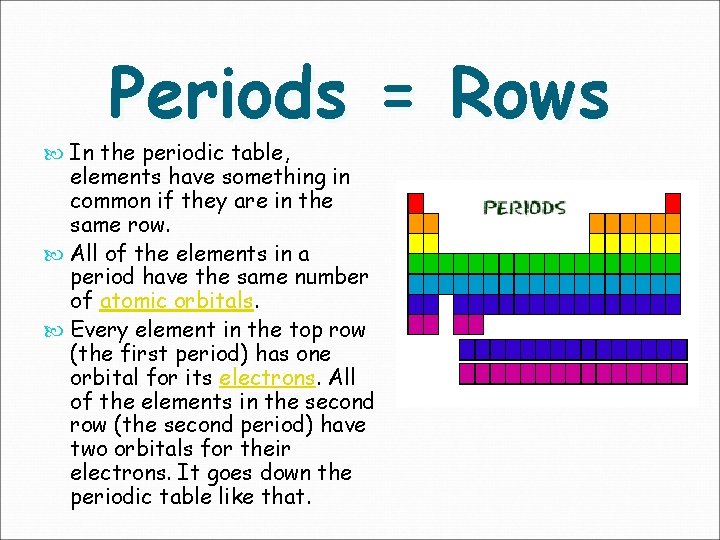
Periods = Rows In the periodic table, elements have something in common if they are in the same row. All of the elements in a period have the same number of atomic orbitals. Every element in the top row (the first period) has one orbital for its electrons. All of the elements in the second row (the second period) have two orbitals for their electrons. It goes down the periodic table like that.
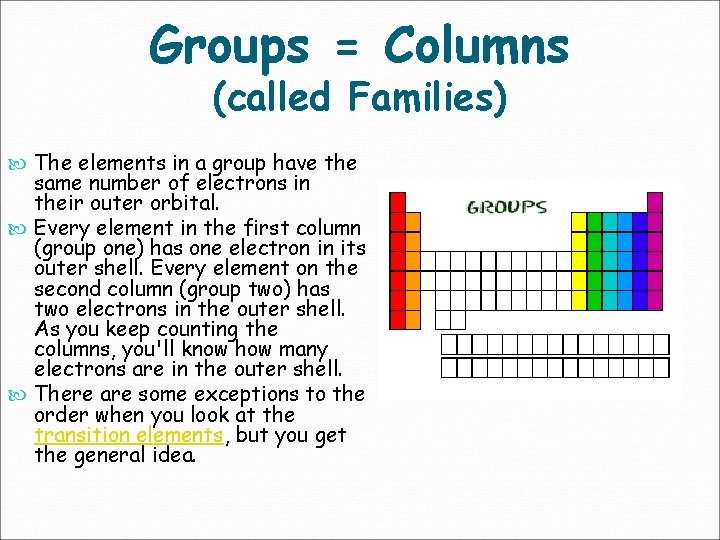
Groups = Columns (called Families) The elements in a group have the same number of electrons in their outer orbital. Every element in the first column (group one) has one electron in its outer shell. Every element on the second column (group two) has two electrons in the outer shell. As you keep counting the columns, you'll know how many electrons are in the outer shell. There are some exceptions to the order when you look at the transition elements, but you get the general idea.
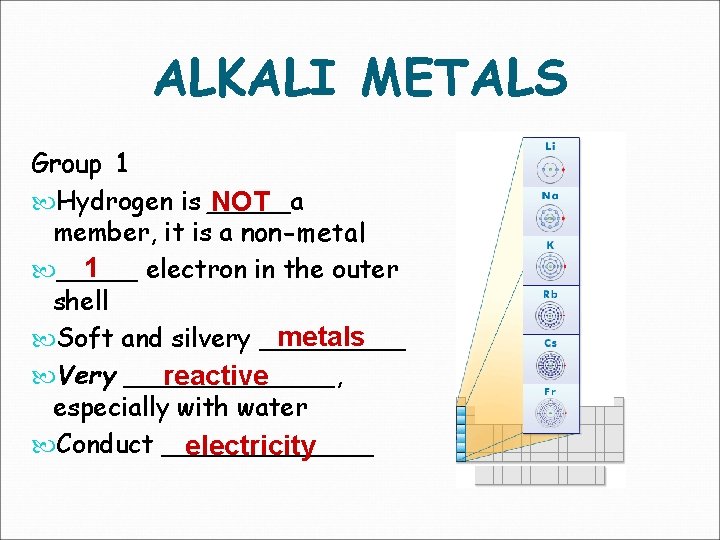
ALKALI METALS Group 1 Hydrogen is _____a NOT member, it is a non-metal 1 _____ electron in the outer shell metals Soft and silvery _____ Very _______, reactive especially with water Conduct _______ electricity
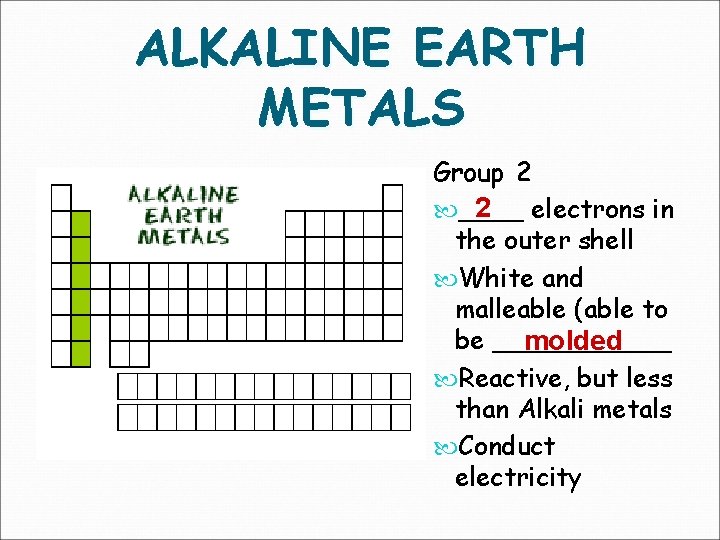
ALKALINE EARTH METALS Group 2 2 electrons in ____ the outer shell White and malleable (able to be ______ molded Reactive, but less than Alkali metals Conduct electricity
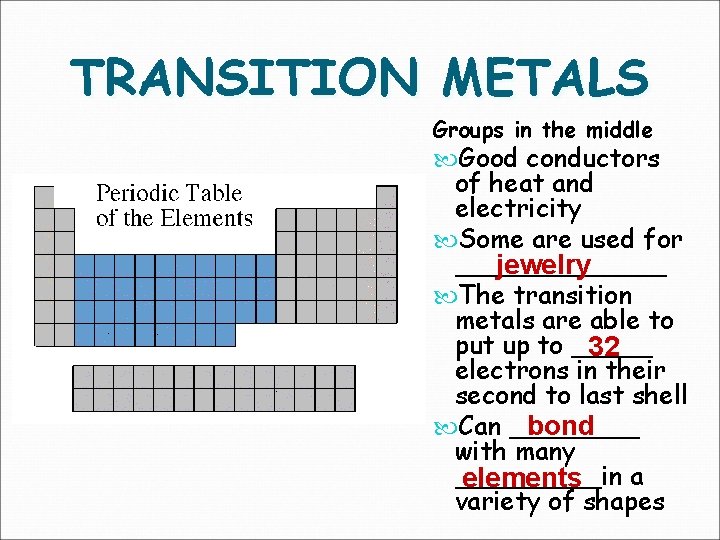
TRANSITION METALS Groups in the middle Good conductors of heat and electricity Some are used for _______ jewelry The transition metals are able to put up to _____ 32 electrons in their second to last shell Can ____ bond with many _____in a elements variety of shapes
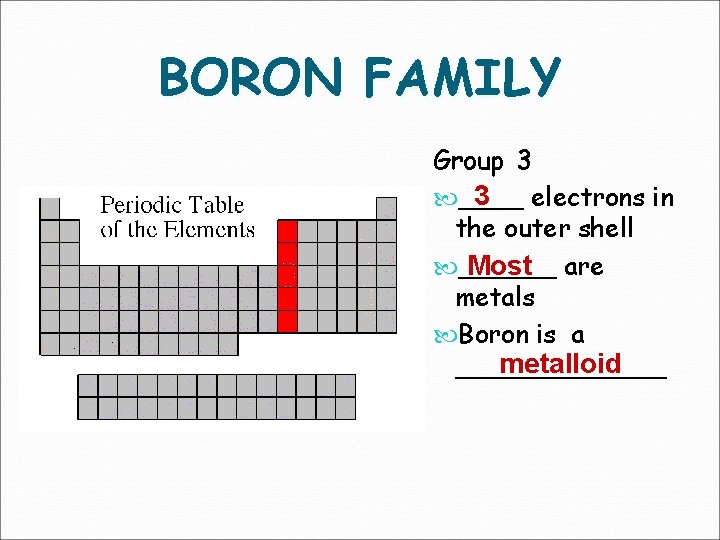
BORON FAMILY Group 3 3 electrons in ____ the outer shell Most are ______ metals Boron is a metalloid _______
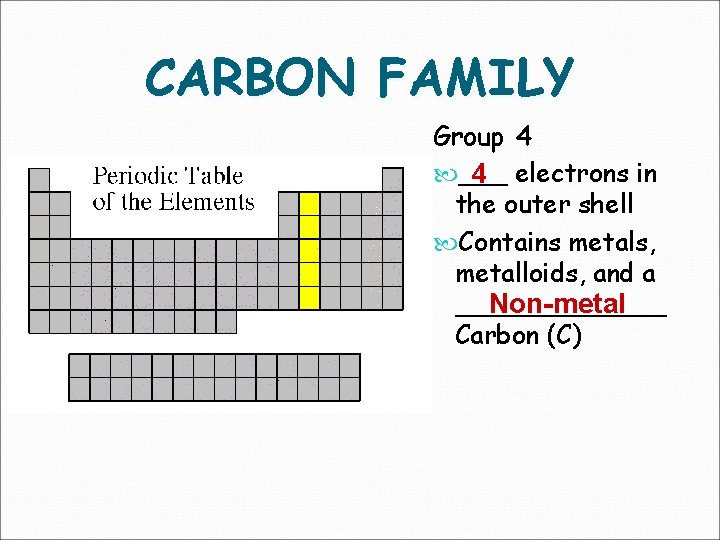
CARBON FAMILY Group 4 ___ 4 electrons in the outer shell Contains metals, metalloids, and a _______ Non-metal Carbon (C)
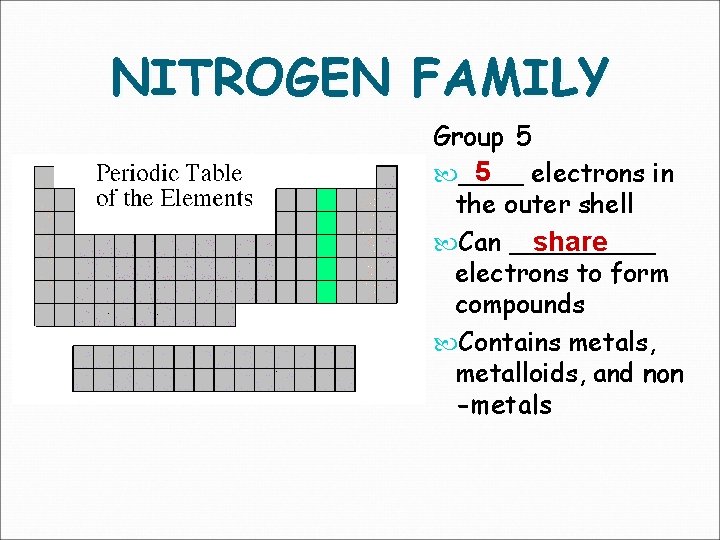
NITROGEN FAMILY Group 5 5 electrons in ____ the outer shell share Can _____ electrons to form compounds Contains metals, metalloids, and non -metals
OXYGEN FAMILY Group 6 6 electrons in ____ the outer shell Contains metals, metalloids, and non-metals Reactive _______
Halogens Group 7 ____ 7 electrons in the outer shell ALL are non _____ metals VERY ____________REACTIVE are often bonded with elements from Group 1
Noble Gases Group 8 Exist as gases Non-metals 8 electrons in ____ the outer shell = FULL _____ Helium (He) has only 2 electrons in ____ the outer shell = Full ____ NOT ______ REACTIVE with other elements
Rare Earth Metals (Special rows on the Periodic Table) Some are radioactive _______ The rare earths are silver, silverywhite, or gray metals. Conduct electricity Actinides Lanthanides
- Slides: 17