Balancing Chemical Equations Chemical Equations A shorthand way

Balancing Chemical Equations

Chemical Equations A shorthand way to describe a chemical reaction using chemical symbols and formulae It’s like a recipe for chemists.

Conserving matter Law of Conservation of Matter matter is neither created nor destroyed in a chemical reaction, it is conserved if you begin a reaction with 5 atoms of Hydrogen, you must end up with 5 atoms of Hydrogen

Parts of the Chemical Equation Three Parts to a Chemical Equation Reactants substances present before the reaction Yield Arrow indicates the direction of a reaction some reactions are reversible [double-sided arrow] Products substances present after the reaction

Chem. Equations – cont’d Chemical symbols give a “before-and-after” picture of a chemical reaction Reactants Mg. O + yields C magnesium oxide to form reacts with carbon Products CO + Mg carbon monoxide and magnesium

Chemical Equations – cont’d Coefficient – (large) # in front of an element or molecule. Ex: 4 C 4 carbon atoms ex: 5 KI 5 potassium atoms & 5 iodine atoms ex: 4 SO 4 4 Sulfur atoms & 16 Oxygen atoms (4 x 4)

Chemical Equations – cont’d Subscript – (small) # to the lower right of an element applies only to the element it is attached to if outside of parentheses [(XY)3] applies to everything inside of parentheses ex: H 2 2 hydrogen atoms ex: CO 2 1 carbon atom, 2 oxygen atoms ex: (SO 4)3 3 Sulfur atoms (1 x 3), 12 Oxygen atoms (4 x 3)

Chemical Equations - cont’d Try this: Al 2(SO 4)3 Aluminum? 2 atoms Sulfur? 3 atoms Oxygen? 12 atoms (4 x 3)

Building a molecule Lets actually build H 2 CO 2 Al 2(SO 4)3 What happens if I put a coefficient in front? Try it!

Balancing Chemical Reactions Remember mass can not disappear so the number of atoms on one side of an equation must match the number of atoms on the other side of an equation. To balance an equation you must: Balance Coefficients. You CAN NOT change a subscript.

Balancing – cont’d Balanced = same # of each element on each side Al + S 2 Al + 3 S Al 2 S 3 Unbalanced Balanced

Balanced or Unbalanced? C 3 H 8 + 5 O 2 Carbon? = 3 vs 3 Hydrogen? = 8 vs 8 Oxygen = 10 vs 10 (4 + 6) 4 H 2 O + 3 CO 2 Is it balanced? Yes.

Balanced or Unbalanced? Fe 3 O 4 + H 2 Iron = 3 vs 1 Oxygen = 4 vs 1 Hydrogen = 2 vs 2 Fe + H 2 O Is it balanced? No. So, now what?

Balancing a Chemical Reaction Set up two columns: labeled “reactants” &“products. ” Identify & record reacting elements on the left. Record product elements in the SAME ORDER on the right. Count & Record # of each reacting element. Count & Record # of each element produced.

Fe 3 O 4 + H 2 Fe + H 2 O

Balancing Chemical Reactions To balance an equation you must: Balance Coefficients. You CAN NOT change a subscript. There are different ways to solve equations. Do what works best for you. Way 1 change your greatest inequality first leave solitary elements for last Way 2 Simply work left to right, guess and check style.
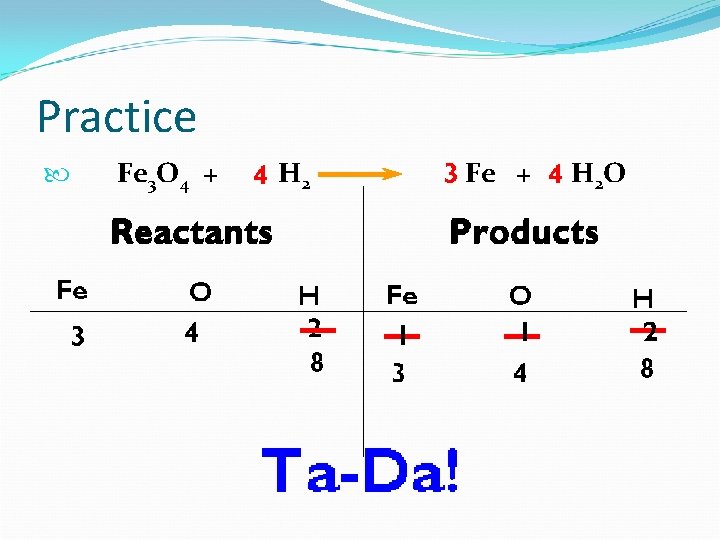
Practice Fe 3 O 4 + H 2 Fe + H 2 O

Practice On your worksheet, complete the two problems on the front of your sheet. 2 2 + ____O 2 2 O ____H 1 2 + ____Na. Br 2 1 2 ____Cl ____Br 2 + ____Na. Cl
- Slides: 18