Types of Chemical Reactions 12192021 Dr Seemal Jelani




















- Slides: 25

Types of Chemical Reactions 12/19/2021 Dr Seemal Jelani Chem-100 1

Learning Outcomes Identify, give evidence for, predict products of, and classify the following types of chemical reactions: 1. Synthesis (combination) 2. Decomposition 3. Single Replacement 4. Double Replacement 5. Neutralization (acid/base) 6. Combustion 12/19/2021 Dr Seemal Jelani Chem-100 2

Vocabulary q Synthesis q Decomposition q Neutralization q Single displacement q Double displacement q Combustion 12/19/2021 Dr Seemal Jelani Chem-100 3

Chemical Reactions A chemical change: any change in which a new substance is formed. Evidence of a Chemical Change: v Release of energy as heat v Release of energy as light v Change in colour v Formation of a gas v Change in odour… 12/19/2021 Dr Seemal Jelani Chem-100 4

Types of Chemical Reactions 1. Neutralization: Acid(H) + Base(OH) salt + H(OH) 2. Combustion: AB + oxygen CO 2 + H 2 O 3. Synthesis: A + B AB 4. Decomposition: AB A + B 5. Single displacement: A + BC AC + B 6. Double displacement: AB + CD AD + CB 12/19/2021 Dr Seemal Jelani Chem-100 5

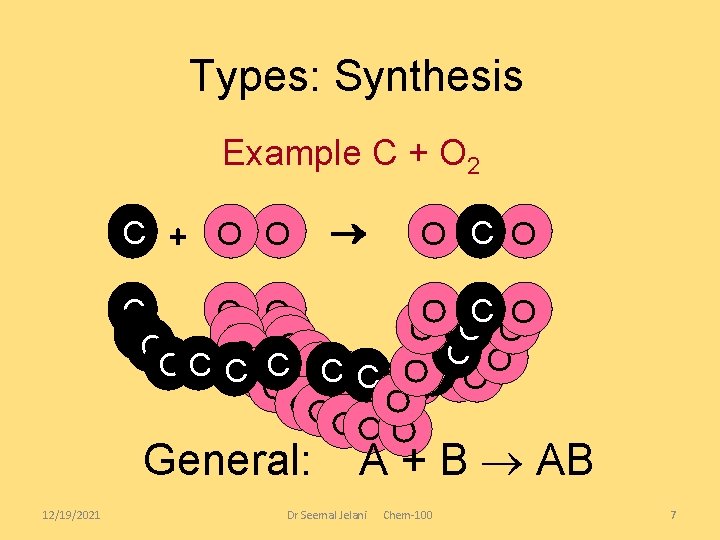
Synthesis Reactions Synthesis reactions are also known as FORMATION reactions. Two or more reactants (usually elements) join to form a compound. – A + B AB where A and B represent elements – The elements may form ionic compounds, like… – Sodium metal and chlorine gas combine to form sodium chloride. – 2 Na + Cl 2 2 Na. Cl Sodium added to chlorine gas See pages 258 - 259 Dr Seemal Jelani 12/19/2021 Chem-100 6

Types: Synthesis Example C + O 2 C + O O O C O CC O O O C C C OC OOC C OCO OO CO O O OO OO OOO General: 12/19/2021 A + B AB Dr Seemal Jelani Chem-100 7

Synthesis Reactions OTHER EXAMPLES… 1. Elements that form ionic compounds: Magnesium metal reacts with oxygen gas to form magnesium oxide. • 2 Mg + O 2 2 Mg. O 2. Elements that form covalent compounds: Nitrogen gas and oxygen gas join to form dinitrogen monoxide. • 2 N 2 + O 2 2 N 2 O SYNTHESIS REACTION (iron + sulphur): http: //www. youtube. com/watch? v=A 5 H 6 DVe 5 FAI Dr Seemal Jelani 12/19/2021 Chem-100 See pages 258 - 259 8

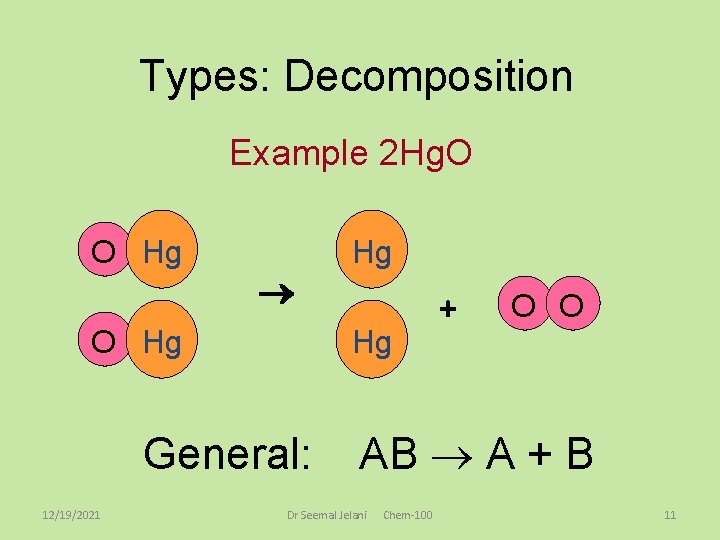
Decomposition Reactions • Decomposition reactions are the opposite of synthesis reactions. – A compounds breaks down into two or more products (often elements). – AB A + B where A and B represent elements 1. Ionic compounds may decompose to produce elements, like the following: • Table salt, sodium chloride, can be broken down into sodium metal and chlorine gas by melting salt at 800ºC and running electricity through it. • 2 Na. Cl 2 Na + Cl 2 See page 260 Dr Seemal Jelani 12/19/2021 Chem-100 9

Types: Decomposition Example: Na. Cl Cl Na General: 12/19/2021 Cl + Na AB A + B Dr Seemal Jelani Chem-100 10

Types: Decomposition Example 2 Hg. O O Hg Hg O Hg General: 12/19/2021 Hg + O O AB A + B Dr Seemal Jelani Chem-100 11

Decomposition Reactions 2. Covalent compounds may decompose into elements, like the following: • By running electricity through water, the water molecules decompose into hydrogen and oxygen gases. • 2 H 2 O 2 H 2 + O 2 DECOMPOSITION REACTION: http: //www. youtube. com/w atch? v=Nddwt. XEA_Ak See page 260 Dr Seemal Jelani 12/19/2021 Chem-100 12

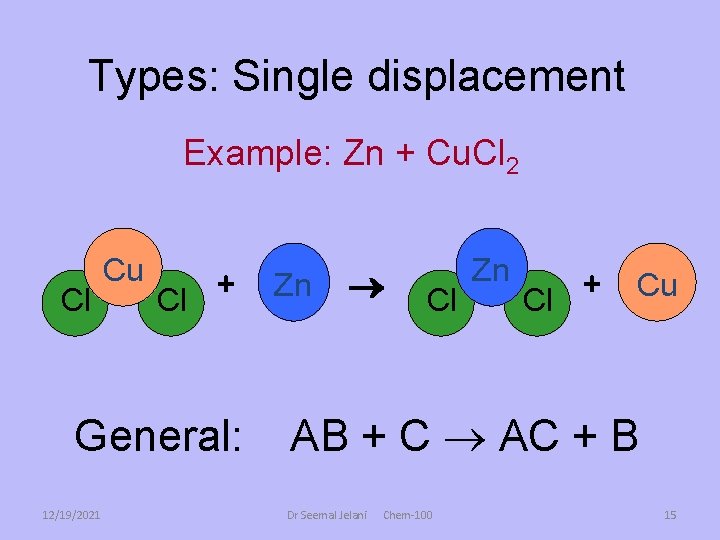
Single Replacement Reactions • Single replacement reactions replace one element from a compound with another element. – A compound an element react, and the element switches places with part of the original compound. • A + BC B + AC where A is a metal, or • A + BC C + BA where A is a non-metal See page 261 Dr Seemal Jelani 12/19/2021 Chem-100 13

Single Replacement Reactions 12/19/2021 Dr Seemal Jelani Chem-100 14

Types: Single displacement Example: Zn + Cu. Cl 2 Cl Cu + Cl General: 12/19/2021 Zn Cl Zn + Cu Cl AB + C AC + B Dr Seemal Jelani Chem-100 15

Single Replacement Reactions 1. When A is a metal: • Aluminum foil in a solution of copper(II) chloride produces solid copper and aluminum chloride. • 2 Al + 3 Cu. Cl 2 3 Cu + 2 Al. Cl 3 2. When A is a non-metal: • When fluorine is bubbled through a sodium iodide solution, iodine and sodium fluoride are produced. • Fl 2 + 2 Na. I I 2 + 2 Na. F 12/19/2021 SINGLE REPLACEMENT: Dr Seemal Jelani Chem-100 http: //www. youtube. com/watch? v=h. Ktynb. Vt. MKc 16

Double Replacement Reactions • Double replacement reactions swap elements between 2 compounds that react together to form two new compounds. – Two compounds react, with elements switching places between the original compounds. AB + CD AD + CB See page 262 Dr Seemal Jelani 12/19/2021 Chem-100 17

Double Replacement Reactions See page 262 Dr Seemal Jelani 12/19/2021 Chem-100 18

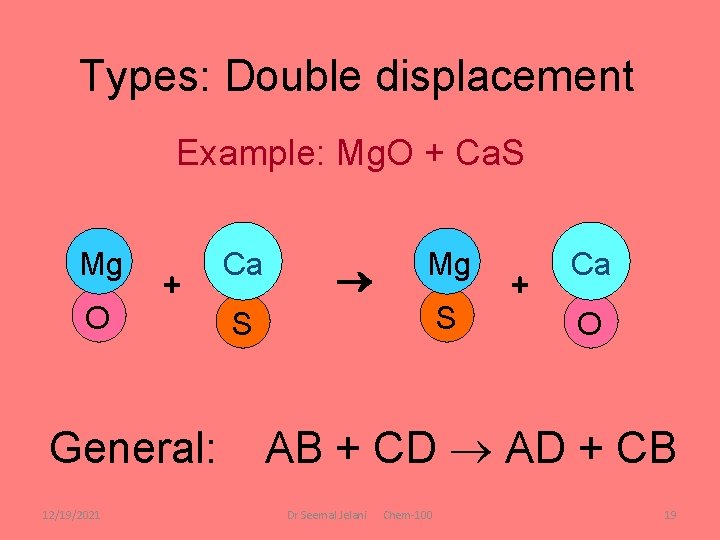
Types: Double displacement Example: Mg. O + Ca. S Mg O + General: 12/19/2021 Ca Mg S S + Ca O AB + CD AD + CB Dr Seemal Jelani Chem-100 19

Double Replacement Reactions Two solutions react to form a precipitate (solid) and another solution. Ionic solution + ionic solid. AB + CD AD + CB – When potassium chromate and silver nitrate react, they form a red precipitate, silver chromate, in a solution of potassium nitrate. – K 2 Cr. O 4 + 2 Ag. NO 3 Ag 2 Cr. O 4 + 2 KNO 3 Dr Seemal Jelani 12/19/2021 silver chromate DOUBLE REPLACEMENT: http: //www. youtube. com/watch? v=op. Y 3 FLr. PTa 4 Chem-100 20

Neutralization Reactions • Neutralization reactions occur when an acid (most compounds starting with H) and a base (most compounds ending in OH) react to form a salt and water. • Neutralization reactions are a type of double replacement. • Acid + base salt + water HX + MOH MX + H 2 O where X and M are elements See page 263 Dr Seemal Jelani 12/19/2021 Chem-100 21

Neutralization Reactions 1. Sulfuric acid is used to neutralize calcium hydroxide: • H 2 SO 4 + Ca(OH) 2 Ca. SO 4 + 2 H 2 O 2. Phosphoric acid helps to neutralize the compounds that cause rust, such as iron(II) hydroxide. • H 3 PO 4 + 3 Fe(OH)2 Fe 3(PO 4)2 + 6 H 2 O http: //www. youtube. com/watch? v=_P 5 h. Gz. A 6 Vb 0 See page 263 Dr Seemal Jelani 12/19/2021 Chem-100 22

Combustion Reactions • Combustion reactions occur when a compound or element react with oxygen to release energy and produce an oxide. – Also sometimes referred to as hydrocarbon combustion. CXHY + O 2 CO 2 + H 2 O where X and Y represent integers METHANOL + oxygen: http: //www. youtube. com/watch? v=98 Ju. J-G 1 q. XY&feature=related See page 264 Dr Seemal Jelani 12/19/2021 Chem-100 23

Combustion Reactions 1. Natural gas (methane) is burned in furnaces to heat homes. » CH 4 + O 2 CO 2 + 2 H 2 O + energy 2. An acetylene torch is used to weld metals together. » 2 C 2 H 2 + 5 O 2 4 CO 2 + 2 H 2 O + energy 3. Carbohydrates like glucose combine with oxygen in our body to release energy. Acetylene torch » C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy See page 264 Dr Seemal Jelani 12/19/2021 Chem-100 24

SUMMARY OF REACTIONS MOVIE TO REVIEW: http: //www. bcscience. com/bc 10/pgs/videos_013_chemical_reactions. html Dr Seemal Jelani 12/19/2021 Chem-100 Take the Section 6. 1 Quiz See page 265 25