Molecular Orbital Theory Hybridization 1 Dr Seemal Jelani

Molecular Orbital Theory Hybridization 1 Dr Seemal Jelani Chem-160 1/2/2022

2 – Molecular orbital theory is concerned with the combination of atomic orbitals to form new molecular orbitals – New orbitals arise from the linear combination of atomic orbitals to form bonding and antibonding orbitals – Bonding orbitals ---------- lower energy Antibonding orbitals-------- High energy Dr Seemal Jelani -160 Chem 1/2/2022

3 – The valence bond theory is an extension of the Lewis structures that considers the overlapping of orbitals to create bonds. – The valence bond theory is only limited in its use because it does not explain the molecular geometry of molecules very well. – This is where hybridization and the molecular orbital theory comes into place Dr Seemal Jelani -160 Chem 1/2/2022

4 Hybridization – It is a simple model that deals with mixing orbitals to from new, hybridized, orbitals. – This is part of the valence bond theory and helps explain bonds formed, the length of bonds, and bond energies; however, this does not explain molecular geometry very well. Dr Seemal Jelani -160 Chem 1/2/2022

5 – sp An example of this is acetylene (C 2 H 2). This combines one s orbital with one p orbital. This means that the s and p characteristics are equal. – sp 2 An example of this is ethylene (C 2 H 4). This is the combination of one s orbital and two p orbitals. – sp 3 An example of this is methane (CH 4). This is the combination of one s orbital and three p orbitals. Dr Seemal Jelani -160 Chem 1/2/2022

6 Antibonding vs. Bonding Orbitals – Electrons that spend most of their time between the nuclei of two atoms are placed into the bonding orbitals – Electrons that spend most of their time outside the nuclei of two atoms are placed into antibonding orbitals. – Reason This is because there is an increasing in electron density between the nuclei in bonding orbitals, and a decreasing in electron density in antibonding orbitals Placing an electron in the bonding orbital stabilizes the molecule because it is in between the two nuclei Dr Seemal Jelani -160 Chem 1/2/2022

7 – Conversely, placing electrons into the antibonding orbitals will decrease the stability of the molecule. – Electrons will fill according to the energy levels of the orbitals. – They will first fill the lower energy orbitals, and then they will fill the higher energy orbitals. – If a bond order of zero is obtained, that means that the molecule is too unstable and so it will not exist. Dr Seemal Jelani -160 Chem 1/2/2022
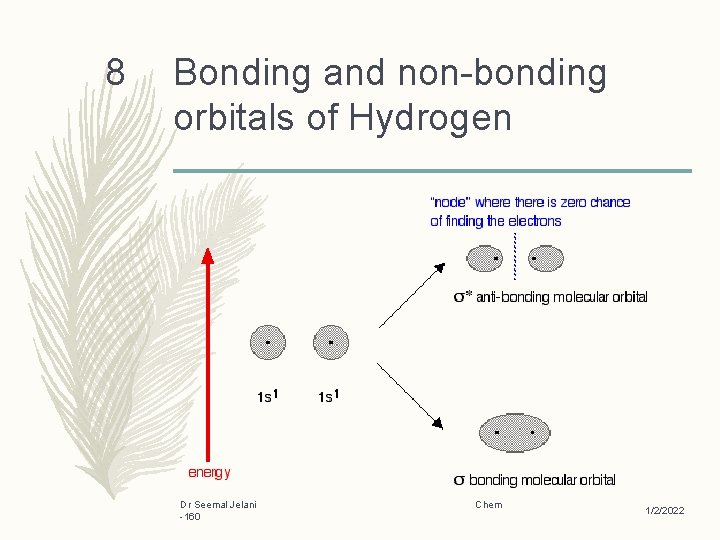
8 Bonding and non-bonding orbitals of Hydrogen Dr Seemal Jelani -160 Chem 1/2/2022

9 Oxygen Example (homonuclear): – Homonuclear species, are molecules composed of only one type of element Dr Seemal Jelani -160 Chem 1/2/2022
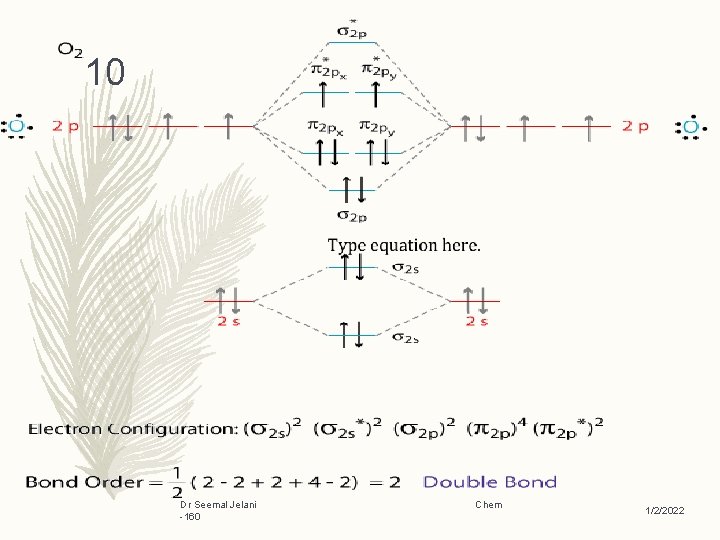
10 Dr Seemal Jelani -160 Chem 1/2/2022
- Slides: 10