TOPIC 5 ENERGETICSTHERMOCHEMISTRY 5 1 MEASURING ENERGY CHANGES



































































- Slides: 73

TOPIC 5 ENERGETICS/THERMOCHEMISTRY 5. 1 MEASURING ENERGY CHANGES

ESSENTIAL IDEA The enthalpy changes from chemical reactions can be calculated from their effect on the temperature of their surroundings. NATURE OF SCIENCE (2. 6) Fundamental principal – conservation of energy is a fundamental principle of science. NATURE OF SCIENCE (3. 1) Making careful observations – measurable energy transfers between systems and surroundings.

INTERNATIONALMINDEDNESS The SI unit of temperature is the Kelvin (K), but the Celsius scale (◦C), which has the same incremental scaling, is commonly used in most countries. The exception if the USA which continues to use the Fahrenheit scale (◦F) for all nonscientific communication.

THEORY OF KNOWLEDGE What criteria do we use in judging discrepancies between experimental and theoretical values? Which ways of knowing do we use when assessing experimental limitations and theoretical assumptions?

UNDERSTANDING/KEY IDEA 5. 1. A Heat is a form of energy.

ENERGY • All chemical reactions are accompanied by energy changes. • Energy is a measure of the ability to do work, that is to move an object against an opposing force. • Examples: heat, light, sound, electricity and chemical energy which is the energy released or absorbed during chemical reactions.

HEAT • Heat is a form of energy which is transferred as a result of a difference in temperature and produces an increase in disorder of the behavior of particles. • Heat increases the average kinetic energy of the molecules in a disordered fashion.

Needed definitions • Enthalpy • Heat content of a substance (at constant pressure) • Enthalpy is also the internal energy stored in the reactants. • The absolute value for the enthalpy of reactants and products cannot be known, but the what can be measured is the difference between the two.

Needed definitions • System – area of interest (Example: beaker and its contents) • Surroundings – everything else in the universe

• An “open” system can exchange matter and energy with the surroundings. • A “closed” system can only exchange energy, not matter, with the surroundings.

• The joule (J) is the SI unit for energy and work.

UNDERSTANDING/KEY IDEA 5. 1. B Temperature is a measure of the average kinetic energy of the particles.

Temperature • Def – measure of the average kinetic energy of the particles • Temperature increase depends upon: • Mass of the object • Amount of heat added • Nature of the substance • Different substances need different amounts of heat to increase the temp of a unit mass by 1 K or 1ºC.

UNDERSTANDING/KEY IDEA 5. 1. C Total energy is conserved in chemical reactions.

UNDERSTANDING/KEY IDEA 5. 1. D Chemical reactions that involve transfer of heat between the system and the surroundings are described as endothermic or exothermic.

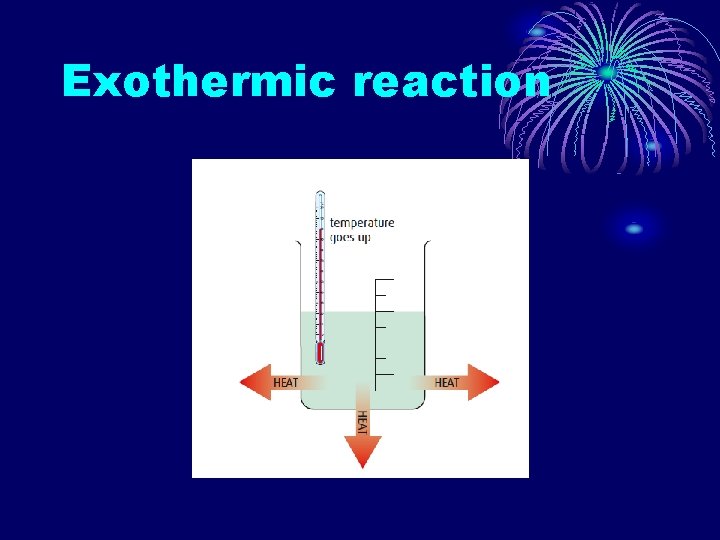
EXOTHERMIC REACTIONS A reaction which results in a transfer of energy from the system to the surroundings. • Heat is given off or produced. • Products have less energy or heat content than the reactants. • ΔH is negative. • The bonds in the products are stronger than the bonds in the reactants.

Exothermic reaction

Exothermic reaction Enthalpy diagram reactants enthalpy H ΔH = negative products extent of reaction

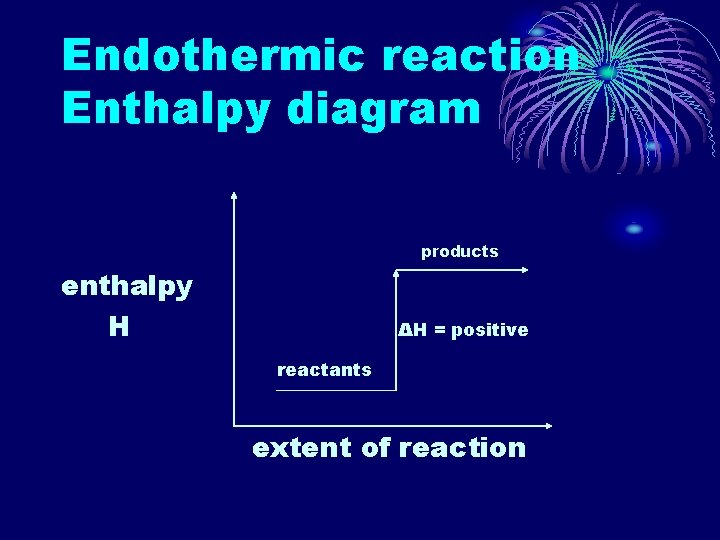
ENDOTHERMIC REACTIONS • A reaction which results in a transfer of energy from the surroundings to the system. • Heat is absorbed. • Reactants have less energy than the products. • ΔH is positive. • The bonds in the reactants are stronger than those in the products.

Endothermic reaction

Endothermic reaction Enthalpy diagram products enthalpy H ΔH = positive reactants extent of reaction

UNDERSTANDING/KEY IDEA 5. 1. E The enthalpy change (ΔH) for chemical reactions is indicated in k. J mol-1

UNDERSTANDING/KEY IDEA 5. 1. F ΔH values are usually expressed under standard conditions, given by ΔH◦, including the standard states.

GUIDANCE Standard state refers to the normal, most pure stable state of a substance measured at 100 k. Pa. Temperature is not part of the definition of standard state, but 298 K is commonly given as the temperature of interest.

GUIDANCE Be familiar with the enthalpy change of combustion (ΔHc ◦) and the enthalpy change of formation (ΔHf ◦).

APPLICATION/SKILLS Be able to calculate the heat change when the temperature of a pure substance is changed using q = mcΔT.

GUIDANCE The specific heat capacity of water is provided in the data booklet in section 2.

APPLICATION/SKILLS Be able to evaluate calorimetry experiments for the enthalpy of a reaction.

GUIDANCE Be familiar with reactions occurring in aqueous solutions and combustion reactions.

GUIDANCE Students can assume the specific heat and density of aqueous solutions are equal to those of water, but be aware of this limitation.

GUIDANCE Heat losses to the environment and the heat capacity of the calorimeter in experiments should be considered, but the use of a bomb calorimeter is not required.

THERMOCHEMICAL EQUATION EXAMPLES Combustion of Methane CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) ΔH = -890 k. J mol-1 Photosynthesis 6 CO 2(g) + 6 H 2 O(l) C 6 H 12 O 6(aq) + 6 O 2(g) ΔH = +2802. 5 k. J mol-1 Thermite Reaction 2 Al(s) + Fe 2 O 3(s) Al 2 O 3(s) + 2 Fe(s) ΔH = -841 k. J mol-1

• You must give the “state” symbols such as (s), (g), (l), (aq) in thermochemical equations because energy changes depend upon the state of the reactants and the products.

• Standard enthalpy change of reaction ΔHº • This is the heat or enthalpy change of a reaction when carried out at standard conditions. • Temperature = 298 K or 25ºC • Pressure = 101. 3 k. Pa (1 atm) • Solution concentration = 1 mol dm-3 (1 M) • All substances are in their standard states (how they are found in nature)

• The actual amount of heat absorbed or produced in a chemical reaction depends upon several factors: • Nature of the reactants and products • The amount or concentration of the reactants. (The greater the amount that reacts, the greater the heat change. ) • The states of the reactants and products. Changing states involves an enthalpy change so this will affect the total heat change. • The temperature of the reaction.

• All combustion reactions are exothermic processes. • All neutralization reactions are exothermic processes.

• Apply the relationship between temperature change, enthalpy change and be able to classify the reaction as endothermic or exothermic. • This will be determined by calorimetry.

Specific Heat Capacity • The following relationship allows the heat change in a material to be calculated from the temperature change. q = mcΔT heat = mass x specific heat x ΔT


• Specific heat capacity (c) is the heat needed to increase the temperature of a unit mass (usually 1 g) by 1 K or 1ºC. • Specific heat of water c = 4. 18 J K-1 g-1

• Be able to deduce from an enthalpy level diagram, the relative stabilities of reactants and products and the sign of the enthalpy change for the reaction.

• There is a natural direction for change. • The direction of change is in the direction of lower stored energy. • We generally expect a reaction to occur if ΔH is negative (exothermic), but some endothermic reactions do occur. These occur when the entropy (S) of the system is large.

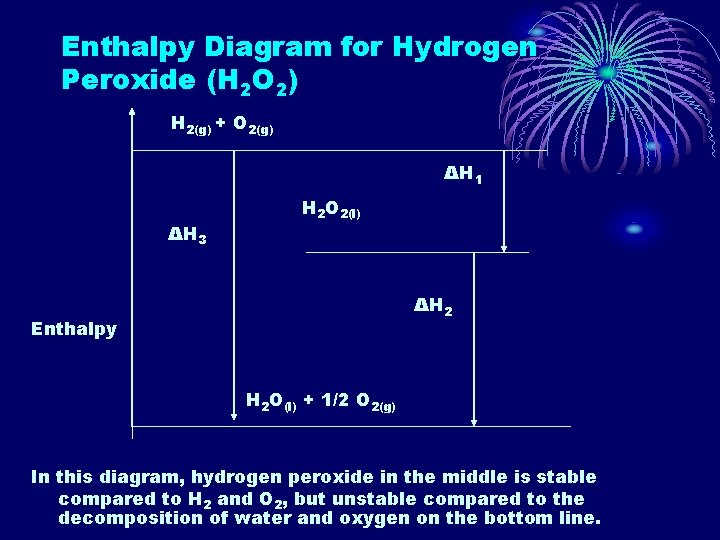
Enthalpy Diagram for Hydrogen Peroxide (H 2 O 2) H 2(g) + O 2(g) ΔH 1 ΔH 3 H 2 O 2(l) ΔH 2 Enthalpy H 2 O(l) + 1/2 O 2(g) In this diagram, hydrogen peroxide in the middle is stable compared to H 2 and O 2, but unstable compared to the decomposition of water and oxygen on the bottom line.

• The heat produced when one mole of a substance is burned in excess oxygen is called the enthalpy of combustion. • ΔHc is exothermic and always negative.

• Calculate the heat energy change when the temperature of a pure substance is changed.

How to calculate heat changes from temp changes. • When heat released by an exothermic reaction is absorbed by water, the temperature of the water increases. • The heat produced by the reaction can be calculated if it is assumed that all the heat is absorbed by the water. heat change of rxn = -heat change of water = - m. H 2 O x c. H 2 O x ΔTH 2 O As the water has gained the heat produced by the reaction, the heat change of the reaction is negative when the temperature of the water increases.

Example Problem 1 • Calculate the enthalpy of combustion of ethanol from the following data. Assume all heat from the reaction is absorbed by the water. Compare your value with the IB Data booklet value and suggest reasons for any differences.

• Mass of water in Cu Calorimeter = 200. 00 g • Temperature increase in water = 13. 00ºC • Mass of ethanol burned = 0. 45 g heat change of rxn = - m. H 2 O x c. H 2 O x ΔTH 2 O = -(200. 00 g)(4. 18 Jg-1ºC-1)(13. 00ºC) = -10868 J The heat calculated above is the heat gained by the water which is also the heat lost by the combustion of the ethanol. Since ΔHc is per mole of substance combusted, you have to find moles of ethanol burned and divide the heat it lost by the moles. To find moles, divide mass by molar mass: 0. 45 g / 46. 08 g mol-1 =. 0098 mol ΔHc = -10868 J /. 0098 mol = -1112883 J/mol = -1112. 883 k. J/mol Using sig figs, the answer is limited by the mass = -1100 k. J/mol

• The IB Data booklet value is -1367 k. J/mol. • What are some reasons for the difference in values? • Not all the heat produced by the combustion is transferred to the water. • Some is needed to heat the Cu calorimeter and some has passed to the surroundings. • The combustion of ethanol is unlikely to be complete due to the limited oxygen available.

Heat of Combustion Set up

Example Problem 2 • When 0. 824 g of ethanol was burned, it produced a temperature rise of 15. 7 K in 275 g of water. • a. Draw the calorimetry set up. • b. Write the ΔHc reaction. • c. Calculate the ΔH for the reaction. • d. The Data Book value is -1367 k. J/mol. Include three reasons for the discrepancy.

• It is important to record qualitative as well as quantitative data. • When asked to evaluate experiments and suggest improvements, avoid giving trivial answers such as incorrect measurements. • Incomplete combustion can be reduced by burning the fuel in oxygen and heat loss can be reduced by insulating the apparatus.

• Design suitable experimental procedures for measuring the heat energy change of reactions.

ΔH of reactions in solution • The enthalpy changes of reaction in solution can be calculated by carrying out the reaction in an insulated system such as a polystyrene cup or Styrofoam calorimeter. • The heat released or absorbed by the reaction can be measured by the change in temp of the water. • The largest source of error in this type of experiment is heat loss to the environment.

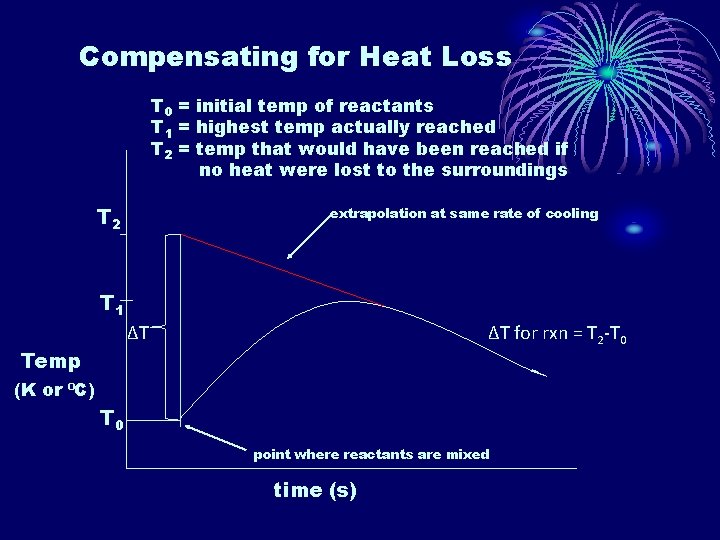
• To compensate for the heat lost by the water in exothermic reactions to the surroundings as the reaction proceeds, a plot of temperature vs time can be drawn. • We want the highest temperature that would have been reached had the reaction been instantaneous. • If we graph the temp vs time, we can extrapolate back to determine the highest temperature.

Compensating for Heat Loss T 0 = initial temp of reactants T 1 = highest temp actually reached T 2 = temp that would have been reached if no heat were lost to the surroundings T 2 extrapolation at same rate of cooling T 1 ∆T ∆T for rxn = T 2 -T 0 Temp (K or ⁰C) T 0 point where reactants are mixed time (s)

• Calculate the enthalpy change for a reaction using experimental data on temperature changes, quantities of reactants and mass of water.

Example problem 3 • 50. 0 cm 3 of 1. 00 mol/dm 3 hydrochloric acid was added to 50. 0 cm 3 of 1. 00 mol/dm 3 sodium hydroxide solution in a polystyrene beaker. The initial temperature of both solutions was 16. 7ºC. After stirring and accounting for the heat loss, the highest temperature reached was 23. 5ºC. Calculate the enthalpy change for the reaction.

• Step 1 – write the equation HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) • Step 2 – calculate molar quantities Anytime you are given a volume and a molarity, multiply them together (watch your units) to get moles. Convert cm 3 to dm 3 first. 50. 0 cm 3 x 1 dm 3/1000 cm 3 = 0. 0500 dm 3 Then multiply the volume by the molarity. 0. 0500 dm 3 x 1. 00 mol/dm 3 = 0. 0500 mol

• Step 3 – calculate the heat evolved • Assumptions: • The solution has the same density as water which is 1. 00 g/cm 3. • The solution has the same specific heat as water which is 4. 18 J/g. K. • Total volume of solution 50. 0 cm 3 + 50. 0 cm 3 = 100. 0 cm 3 • Using the density of water, calculate the mass of the solution 100. 0 cm 3 x 1. 00 g/cm 3 = 100. 0 g • Temperature change 23. 5ºC – 16. 7ºC = 6. 8 K • The heat evolved from the reaction = mcΔT. q = (100. g)(4. 18 J/g. K)(6. 8 K) = 2840 J = -28. 4 k. J • The ΔH is per mole so ΔH = -28. 4 k. J/. 0500 mol = -56. 8 k. J/mol

• Evaluate the results of experiments to determine enthalpy changes.

Example Problem to evaluate the results of an experiment. • The experiment involves the neutralization of sodium hydroxide with sulfuric acid to form water and sodium sulfate. • Assumptions: • The Na. OH and H 2 SO 4 are 1 M (1 mol dm-3). • The total volume is kept constant at 120 cm 3 with varying volumes of Na. OH and H 2 SO 4. • No heat is lost from the system. • All heat is transferred to the water. • Assume 120 cm 3 of solution contains 120 cm 3 of water.

• Your task is to evaluate the following when the temperature changes are measured when different volumes of Na. OH and H 2 SO 4 are mixed: • Determine the volumes of the solutions which produce the largest increase in temperature. • Calculate the heat produced by the reaction when the maximum temperature was produced. • Calculate the heat produced for one mole of Na. OH. • Calculate the %error and suggest a reason for the discrepancy between the experimental and literature values. • The literature value at standard conditions is -57. 5 k. J/mol-1.

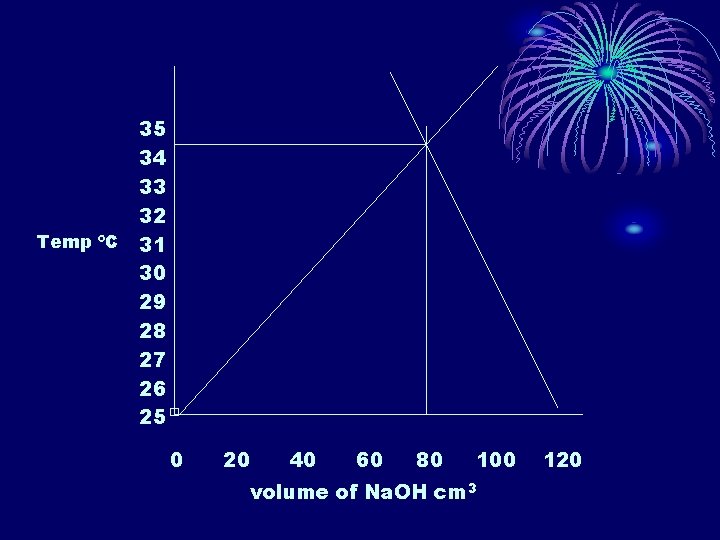
35 34 33 32 Temp ºC 31 30 29 28 27 26 25 □ 0 20 40 60 80 100 volume of Na. OH cm 3 120

• The highest increase in temp is produced at 80 cm 3 of Na. OH which means that there is 120 -80 = 40 cm 3 of H 2 SO 4. • The maximum temp where the lines cross on the graph was 33. 5ºC. q = -mcΔT = -(120. 0 g)(4. 18 J/gºC)(33. 5ºC-25. 0ºC) = -4264 J • To calculate the heat produced per mole: 80. 0 cm 3 x 1 dm 3/1000 cm 3 x 1 mol/dm 3 =. 0800 mol ΔH = -4264 J/. 0800 mol = -53300 Jmol-1 = -53. 3 k. Jmol-1 • Percent Error %error = (-57. 5 --53. 3)/-57. 5 x 100% = 7% • There are uncertainties in temp, vol and concentration measurements and it is assumed all heat is transferred to the water and no heat is lost from the system.

Another fine example • 50. 0 cm 3 of 0. 200 mol dm-3 copper II sulfate was placed in a polystyrene cup. After 2 min, 1. 20 g of powdered zinc was added. The temperature was taken every 30 seconds and the following graph was obtained. Calculate the enthalpy change for the reaction taking place.

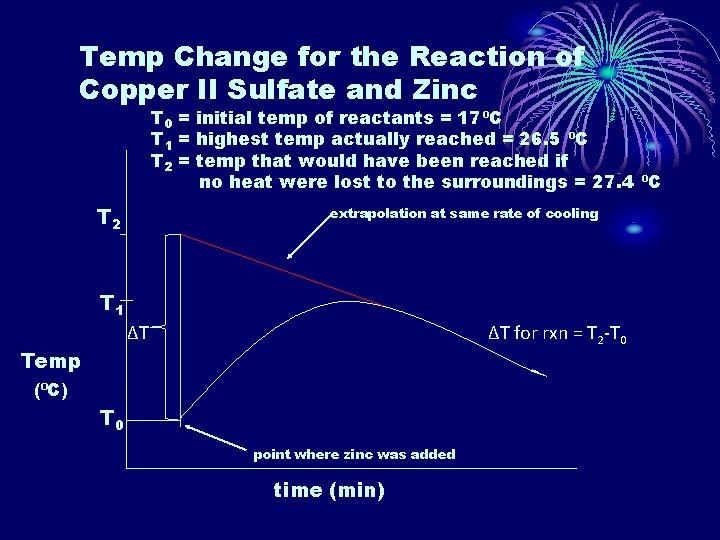
Temp Change for the Reaction of Copper II Sulfate and Zinc T 0 = initial temp of reactants = 17⁰C T 1 = highest temp actually reached = 26. 5 ⁰C T 2 = temp that would have been reached if no heat were lost to the surroundings = 27. 4 ⁰C T 2 extrapolation at same rate of cooling T 1 ∆T ∆T for rxn = T 2 -T 0 Temp (⁰C) T 0 point where zinc was added time (min)

• From the graph, we are able to extrapolate back and determine that T 2 would have been 27. 4⁰C. • That gives us a ∆T of 27. 4⁰C - 17. 0⁰C = 10. 4⁰C • Next we should recognize that we are mixing two given amounts which gives us a limiting reactant problem. Cu. SO 4(aq) + Zn(s) Cu(s) + Zn. SO 4(aq) 1. 20 g Zn x mol/65. 4 g = 0. 0183 mol Zn given 50. 0 cm 3 x 1 dm 3/1000 cm 3 x 0. 200 mol/dm 3 = 0. 0100 mol Cu. SO 4 given 0. 0183 mol Zn x 1/1 = 0. 0183 mol Cu. SO 4 needed 0. 0100 mol Cu. SO 4 x 1/1 = 0. 0100 mol Zn needed We need 0. 0183 mol Cu. SO 4 and are only given 0. 0100 mol so Cu. SO 4 is the limiting reactant.

• We know that the reaction will stop after the 0. 0100 mol of Cu. SO 4 is used up so the heat evolved is per 0. 0100 mol of Cu. SO 4. • q = mc∆T = (50. 0 cm 3 x 1. 00 g/cm 3)(4. 18 J/g⁰C)(10. 4⁰C) = 2170 J = 2. 17 k. J • ∆H = -2. 17 k. J / 0. 0100 mol = -217 k. J mol-1

Comments on problem • Why was there a wait time at the beginning? • The highest temp was reached 5 min after the zinc had been added. What exactly was happening at this point? • Copper II sulfate solution is blue. Zinc sulfate solution is colorless. Zinc is a silver – grey metal and copper is a reddish brown metal. State what you would think you would observe as the reaction proceeds. • What are three assumptions you have made in arriving at your answer? • The literature value is -218 k. J/mol. Comment on the validity of your assumptions you stated.

Final comments on calculating ∆H • There are 4 types of problems that you should be familiar with. • Combustion calorimetry • Neutralization rxns • Extrapolation for heat loss • Finding answers from a graph when 2 solutions are mixed and volumes are kept constant.

• Make sure you always record qualitative as well as quantitative results. • Be sure to state the assumptions you are making which are often that no heat is lost from the system, all heat is transferred to the water and that the mass of the solution is assumed to be water so you are using the specific heat of water in your calculations.

Citations International Baccalaureate Organization. Chemistry Guide, First assessment 2016. Updated 2015. Brown, Catrin, and Mike Ford. Higher Level Chemistry. 2 nd ed. N. p. : Pearson Baccalaureate, 2014. Print. Most of the information found in this power point comes directly from this textbook. The power point has been made to directly complement the Higher Level Chemistry textbook by Catrin and Brown and is used for direct instructional purposes only.