Chapter 17 1 Gibbs Free Energy For a

Chapter 17 1

Gibbs Free Energy For a constant temperature and constant pressure process: -T Suniv = Hsys - T Ssys Gibbs free energy ( G)- Can be used to predict spontaneity. G < 0 The reaction is spontaneous in the forward direction. G = -T(+ Suniv) G > 0 Suniv > 0 The reaction is nonspontaneous as written. The reaction is spontaneous in the reverse direction. + G = -T(- Suniv) S < 0 univ G = 0 The reaction is at equilibrium. G = -T( Suniv) = 0 Suniv = 0 2

Gibbs Free Energy G = H - T S • If you know G for reactants and products then you can calculate if a reaction is spontaneous. • If you know G for two reaction then you can calculate if the sum is spontaneous. • If you know S, H and T then you can calculate spontaneity. • Can predict the temperature when a reaction becomes spontaneous. • If you have Hvap or Hfus and S you can predict boiling and freezing points. • If you have Hvap or Hfus and T you can predict the entropy change during a phase change. • Can predict equilibrium shifts. 3

Chapter 17 4

Free Energy and Equilibrium 0 ) is the free-energy The standard free-energy of reaction (DGrxn change for a reaction when it occurs under standard-state conditions. a. A + b. B c. C + d. D 0 Grxn = S n G 0 f (products) - S m G 0 f (reactants) G° < 0 favors products spontaneously G° > 0 favors reactants spontaneously Does not tell you it will go to completion! a. A + b. B G° fwd G° rev c. C + d. D G° fwd = - G° rev The value of G° calculated under the standard conditions characterizes the “driving force” of the reaction towards equilibrium. 5

Free Energy and Equilibrium G° = -R T ln. K standard free-energy (k. J/mol) gas constant (8. 314 J/K mol) equilibrium constant (Kp, Kc, Ka, Ksp, etc. ) temperature (K) • Arguably most important equation in chemical thermodynamics! • It allows us to calculate the extent of a chemical reaction if its enthalpy and entropy changes are known. • The changes in enthalpy and entropy can be evaluated by measuring the variation of the equilibrium constant with temperature. • This relationship is only valid for the standard conditions, i. e. when the activities of all reactants and products are equal to 1. 6
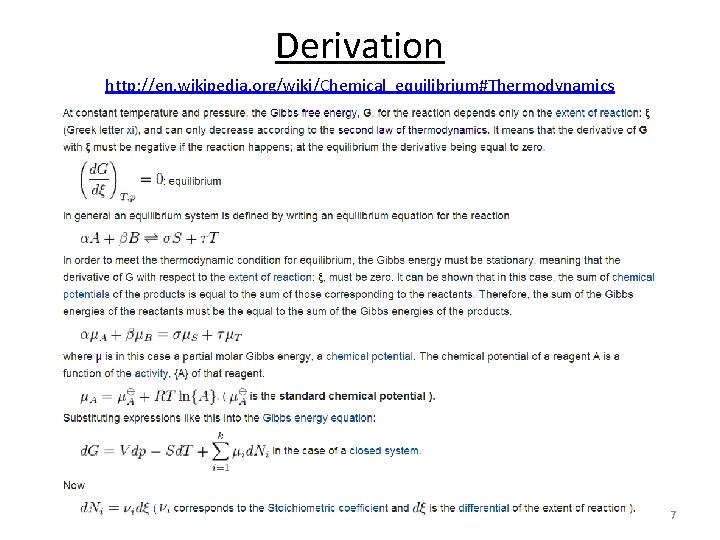
Derivation http: //en. wikipedia. org/wiki/Chemical_equilibrium#Thermodynamics 7

8

Free Energy and Equilibrium G° = -R T ln. K R is constant so at a given temperature: 0 DG (k. J) K 100 3 x 10 -18 50 2 x 10 -9 10 2 x 10 -2 1 7 x 10 -1 0 1 -1 1. 5 -10 5 x 101 -50 6 x 108 -100 3 x 1017 -200 1 x 1035 Essentially no forward reaction; reverse reaction goes to completion Forward and reverse reactions proceed to same extent Forward reaction goes to completion; essentially no reverse reaction REVERSE REACTION 9 x 10 -36 FORWARD REACTION 200 Significance 9

Using G° and K Using the table of standard free energies, calculate the equilibrium constant, KP, for the following reaction at 25 C. 2 HCl(g) H 2(g) + Cl 2(g) G 0 rxn = S n G 0 (products) - S m G 0 f (reactants) f G 0 rxn = [1(0) + 1(0)] - [2( 95. 3 k. J/mol)] G 0 rxn = 190. 6 k. J/mol Non-spontaneous! Favors reactants! G° = -R T ln K From appendix 3: H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol 10

Using G° and K Using the table of standard free energies, calculate the equilibrium constant, KP, for the following reaction at 25 C. 2 HCl(g) H 2(g) + Cl 2(g) G 0 rxn = S n G 0 (products) - S m G 0 f (reactants) f G 0 rxn = [1(0) + 1(0)] - [2( 95. 3 k. J/mol)] G 0 rxn = 190. 6 k. J/mol Non-spontaneous! Favors reactants! G° = -R T ln K 190. 6 k. J/mol = (8. 314 J/K·mol)(25 C) ln KP correct units 190. 6 k. J/mol = (8. 314 x 10 3 k. J/K·mol)(298 K) ln KP KP = 3. 98 x 10 34 Favors reactants! 11

Example 17. 6 Using data listed in Appendix 3, calculate the equilibrium constant (KP) for the following reaction at 25°C: 2 H 2 O(l) 2 H 2(g) + O 2(g) ΔG°rxn = [2ΔG°f(H 2) + ΔG°f(O 2)] - [2ΔG°f(H 2 O)] = [(2)(0 k. J/mol) + (0 k. J/mol)] - [(2)(-237. 2 k. J/mol)] = 474. 4 k. J/mol Bonus: What is the Kp for the reverse reaction? 2 H 2(g) + O 2(g) 2 H 2 O(l) ΔG°rxn = -474. 4 k. J/mol or 1/Kp(fwd) = Kp(rev) ln. Kp(fwd) = -ln. Kp(rev) 12

Example 17. 7 In Chapter 16 we discussed the solubility product of slightly soluble substances. Using the solubility product of silver chloride at 25°C (1. 6 x 10 -10), calculate ΔG° for the process Ag. Cl(s) Ag+(aq) + Cl-(aq) Ksp = [Ag+][Cl-] = 1. 6 x 10 -10 G° = -R T ln. K ΔG° = -(8. 314 J/K·mol) (298 K) ln (1. 6 x 10 -10) = 5. 6 x 104 J/mol = 56 k. J/mol Favors reactants. Not very soluble! 13

Free Energy and Equilibrium G° = -R T ln. K G° Rearrange: ln K = R T Substitution: ln K = - H - T S G = H - T S R T H T S Rearrange: ln K = + R T H 1 ln K = R T () + S R 14
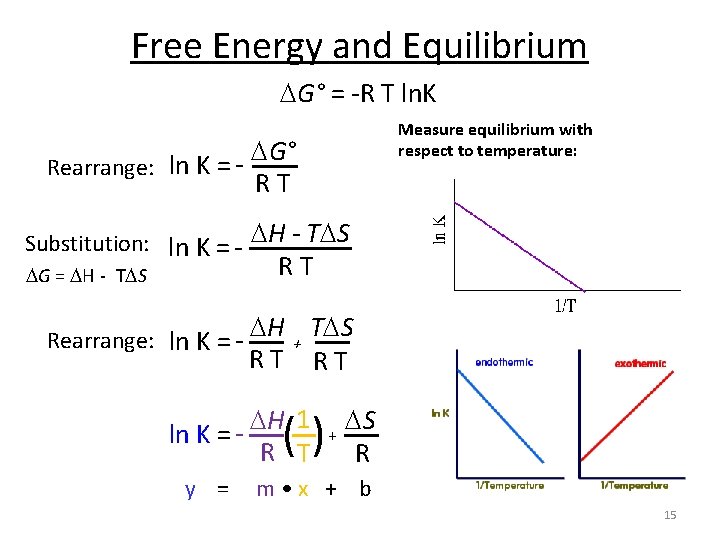
Free Energy and Equilibrium G° = -R T ln. K Measure equilibrium with respect to temperature: G° Rearrange: ln K = R T Substitution: ln K = - H - T S G = H - T S R T H T S Rearrange: ln K = + R T H 1 ln K = R T () + S R y = m • x + b 15

Free Energy and Equilibrium Find the S and H of the following: kobs kf A B rate. A B = kobs [A] rate. B A = kf [B] At equilibrium: rate. A B = rate. B A kobs [A] = kf [B] kobs kf = [B] [A] = Ku 16
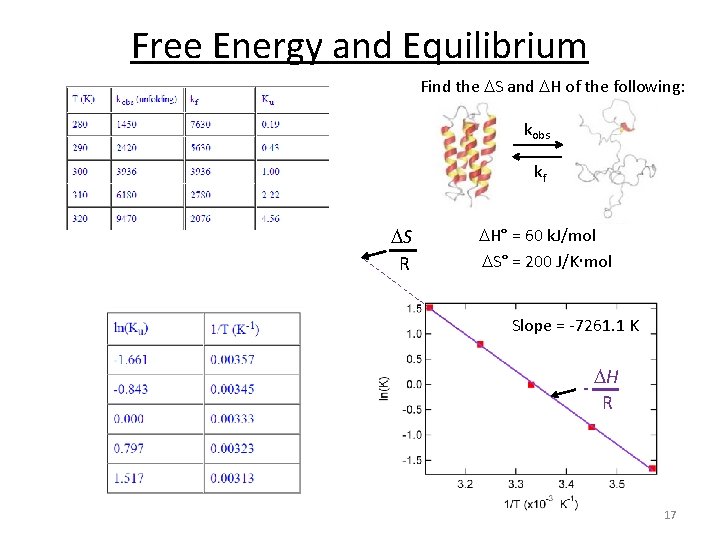
Free Energy and Equilibrium Find the S and H of the following: kobs kf S R H° = 60 k. J/mol S° = 200 J/K mol Slope = -7261. 1 K - H R 17

G° vs G You have already delta ΔG°, ΔS° and ΔH° in which the ° indicates that all components are in their standard states. Definition of “standard”: • Even if we start a reaction at standard conditions (1 M) the reaction will quickly deviate from standard. • G° indicates whether reactants or products are favored at equilibrium. • G at any give time is used to predict the direction shift to reach equilibrium. • If a mixture is not at equilibrium, the liberation of the excess Gibbs free energy ( G) is the “driving force” for the composition of the mixture to *There is no "standard temperature", change until equilibrium is reached. 18 but we usually use 298. 15 K (25° C).

Free Energy and Equilibrium At equilibrium: At any time: G° = -R T ln K G = G° + R T ln Q reaction quotient temperature standard free-energy (K) non-standard free-energy gas constant (k. J/mol) (8. 314 J/K mol) The sign of G tells us that the reaction would have to shift to the left to reach equilibrium. G < 0, reaction will shift right G > 0, reaction will shift left G = 0, the reaction is at equilibrium The magnitude of G tells us how far it has to go to reach equilibrium. 19

Free Energy and Equilibrium G = G° + R T ln Q reaction quotient temperature standard free-energy (K) non-standard free-energy gas constant (k. J/mol) (8. 314 J/K mol) If Q/K < 1, then ln Q/K < 0; the reaction proceeds to the right ( G < 0) If Q/K > 1, then ln Q/K > 0; the reaction proceeds to the left ( G > 0) If Q/K = 1, then ln Q/K = 0; the reaction is at equilibrium ( G = 0) 20

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: From appendix 3: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q calculate constant calculate given 21

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm From appendix 3: H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q G° = [2( 95. 27 k. J/mol)] [0 + 0] = 190. 54 k. J/mol 22

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: From appendix 3: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q G° = 190. 54 k. J/mol constant Q = 0. 80 given G = 190, 540 J/mol + (8. 314 J/K·mol)(298 K) ln (0. 80) G = 191. 09 k. J/mol Because ΔG < 0, the net reaction proceeds from left to right to reach equilibrium. 23

Example 17. 8 The equilibrium constant (KP) for the reaction N 2 O 4(g) 2 NO 2(g) is 0. 113 at 298 K, which corresponds to a standard free-energy change of 5. 40 k. J/mol. In a certain experiment, the initial pressures are PNO 2 = 0. 122 atm and PN 2 O 4 = 0. 453 atm. Calculate ΔG for the reaction at these pressures and predict the direction of the net reaction toward equilibrium. Because ΔG < 0, the net reaction proceeds from left to right to reach equilibrium. 24
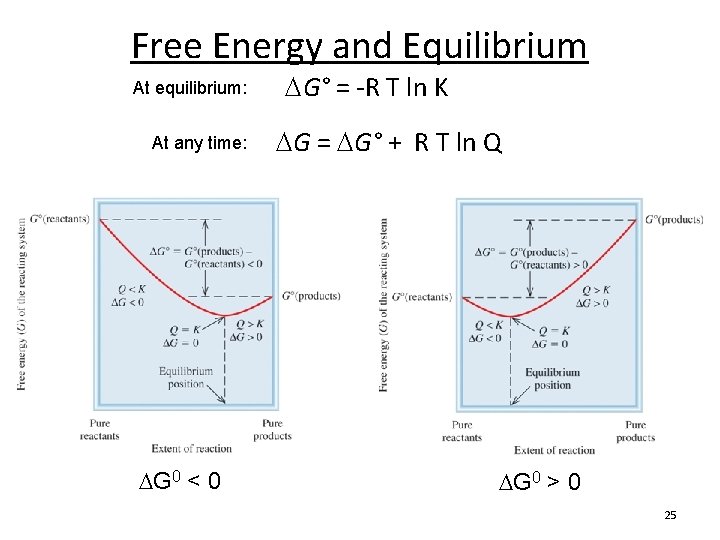
Free Energy and Equilibrium At equilibrium: At any time: G 0 < 0 G° = -R T ln K G = G° + R T ln Q G 0 > 0 25

Chapter 17 26
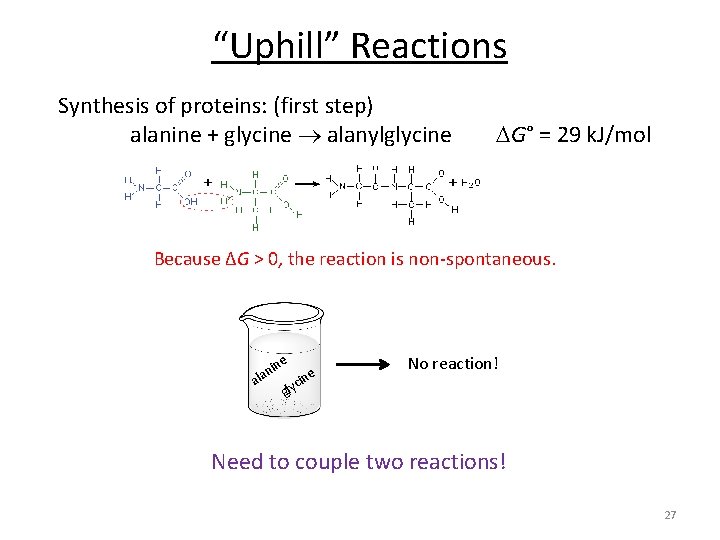
“Uphill” Reactions Synthesis of proteins: (first step) alanine + glycine alanylglycine G° = 29 k. J/mol Because ΔG > 0, the reaction is non-spontaneous. e in lan a e cin No reaction! gly Need to couple two reactions! 27

Coupled Reactions- using a thermodynamically favorable ( G° < 0) reaction ( G° < 0) to drive an unfavorable one ( G° > 0). Example: Industrial ore separation- Zinc Metal Sphalerite ore Major applications in the US 1) Galvanizing (55%) 2) Alloys (21%) 3) Brass and bronze (16%) 4) Miscellaneous (8%) White pigment (Zn. O) Fire retardant (Zn. Cl 2) Vitamin supplement (Zn 2+) Reducing agent (Zn(s)) We need 2000 tones of the zinc metal per year!28

Coupled Reactions- using a thermodynamically favorable ( G° < 0) reaction ( G° < 0) to drive an unfavorable one ( G° > 0). Example: Industrial ore separation- Unfavorable reaction ( G° > 0) 95 % of Zinc is produced by this method 29
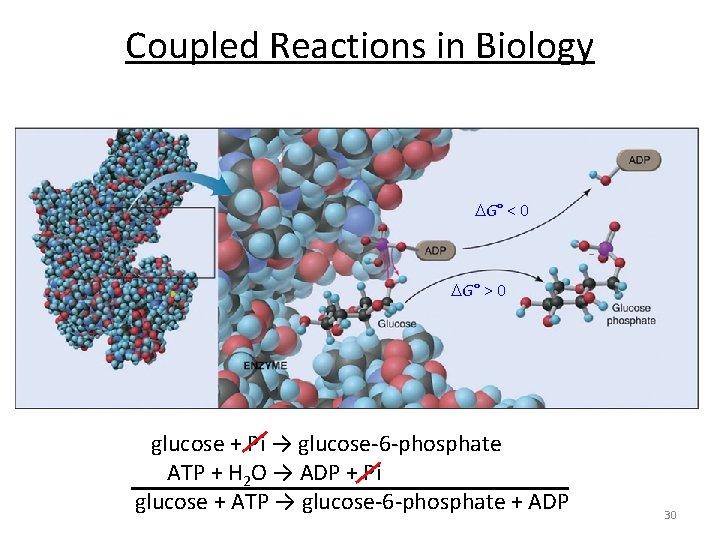
Coupled Reactions in Biology G° < 0 G° > 0 glucose + Pi → glucose-6 -phosphate ATP + H 2 O → ADP + Pi glucose + ATP → glucose-6 -phosphate + ADP 30
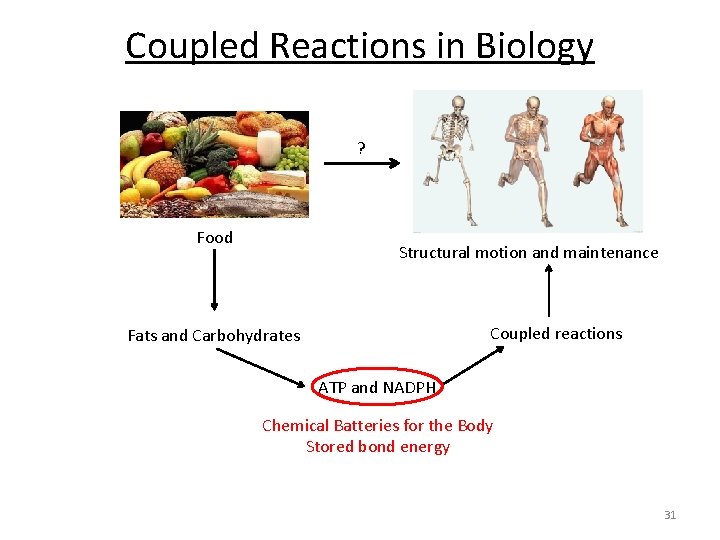
Coupled Reactions in Biology ? Food Structural motion and maintenance Coupled reactions Fats and Carbohydrates ATP and NADPH Chemical Batteries for the Body Stored bond energy 31
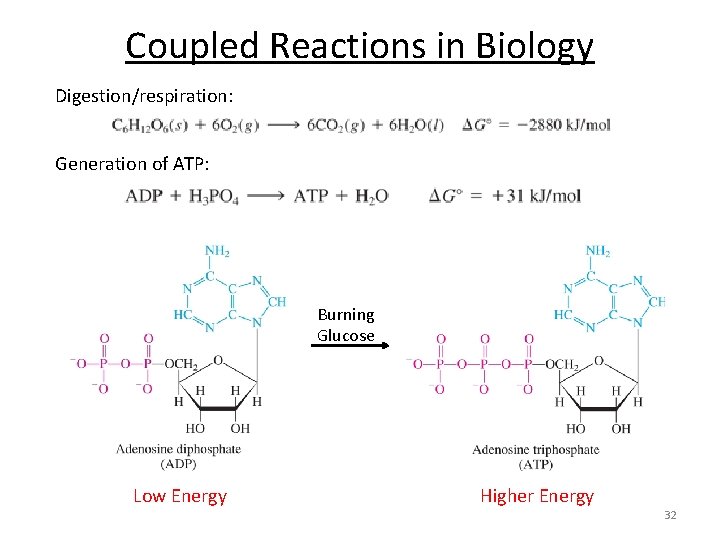
Coupled Reactions in Biology Digestion/respiration: Generation of ATP: Burning Glucose Low Energy Higher Energy 32
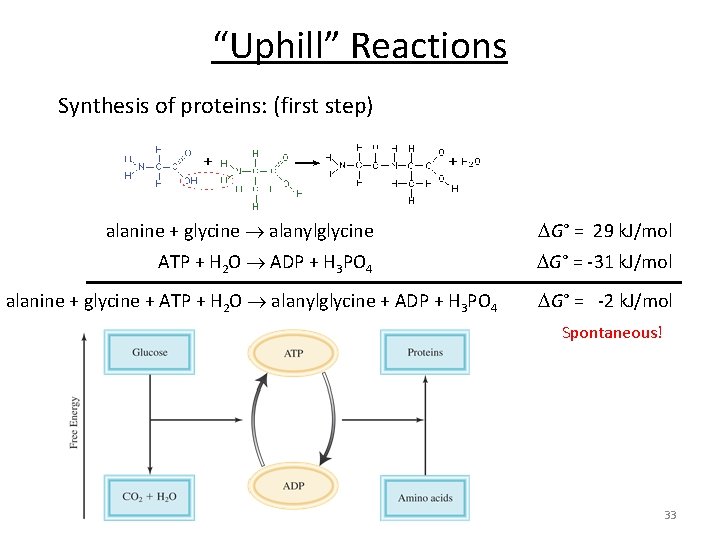
“Uphill” Reactions Synthesis of proteins: (first step) alanine + glycine alanylglycine G° = 29 k. J/mol ATP + H 2 O ADP + H 3 PO 4 G° = -31 k. J/mol alanine + glycine + ATP + H 2 O alanylglycine + ADP + H 3 PO 4 G° = -2 k. J/mol Spontaneous! 33
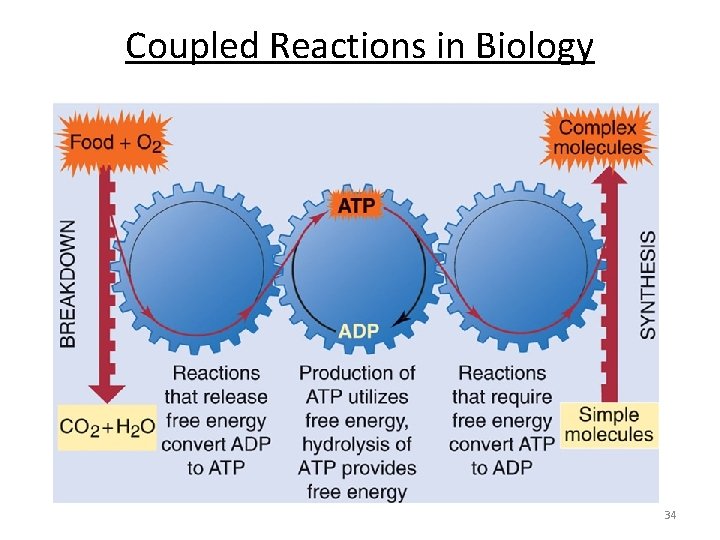
Coupled Reactions in Biology 34
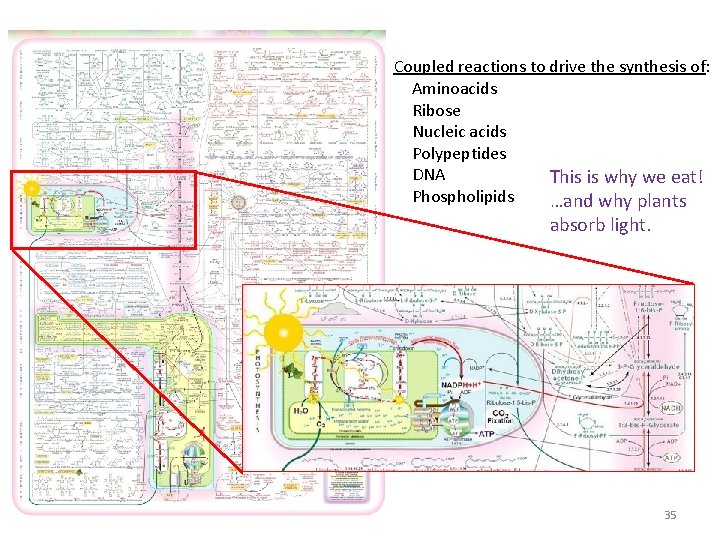
Coupled reactions to drive the synthesis of: Aminoacids Ribose Nucleic acids Polypeptides DNA This is why we eat! Phospholipids …and why plants absorb light. 35

Chapter 17 36
- Slides: 36