Thermodynamics Energy is neither created or destroyed during

















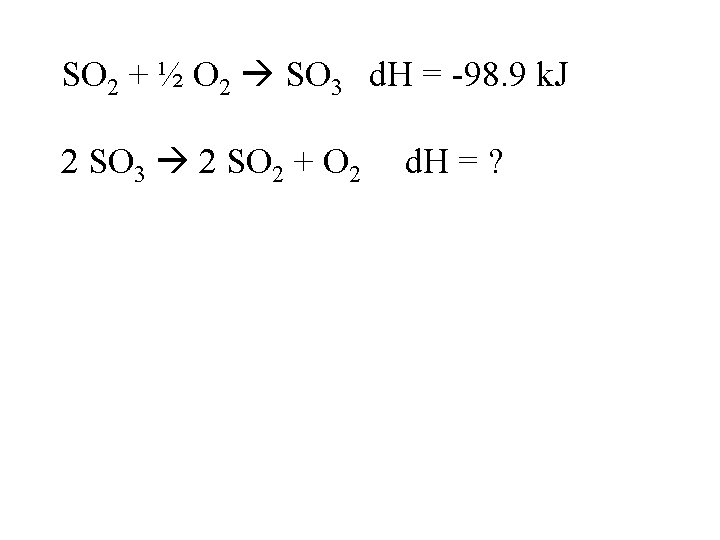





- Slides: 39

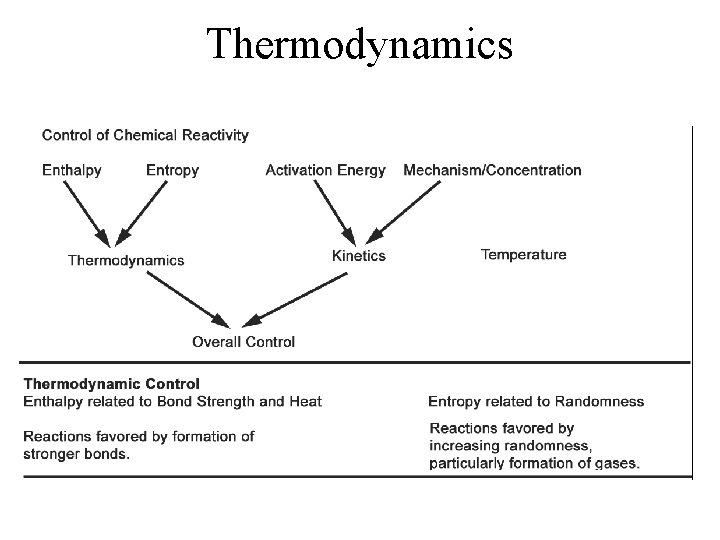
Thermodynamics

Energy is neither created or destroyed during chemical or physical changes, but it is transformed from one form to another. Euniverse = 0

TYPES of ENERGY Kinetic Mechanical Thermal Electrical Radiant Energy Conversion Examples: 1. dropping a rock 2. using a flashlight 3. driving a car Potential Gravitational Electrostatic Chemical


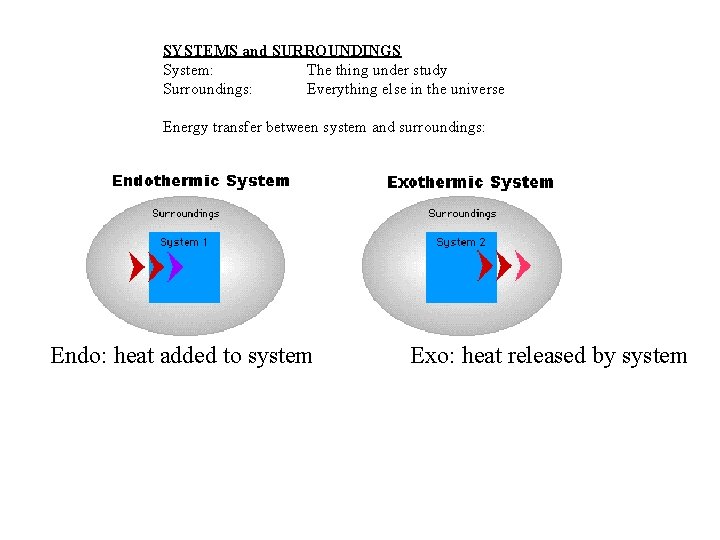
SYSTEMS and SURROUNDINGS System: The thing under study Surroundings: Everything else in the universe Energy transfer between system and surroundings: Endo: heat added to system Exo: heat released by system

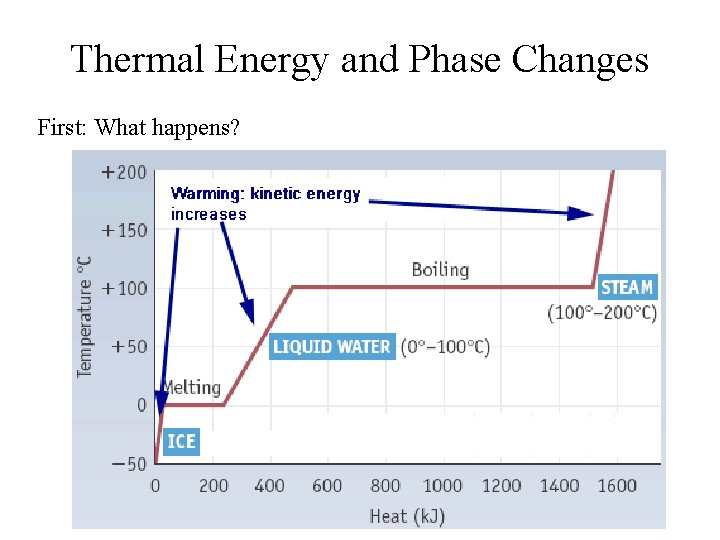
HEAT: What happens to thermal (heat) energy? Three possibilities: • Warms another object • Causes a change of state • Is used in an endothermic reaction

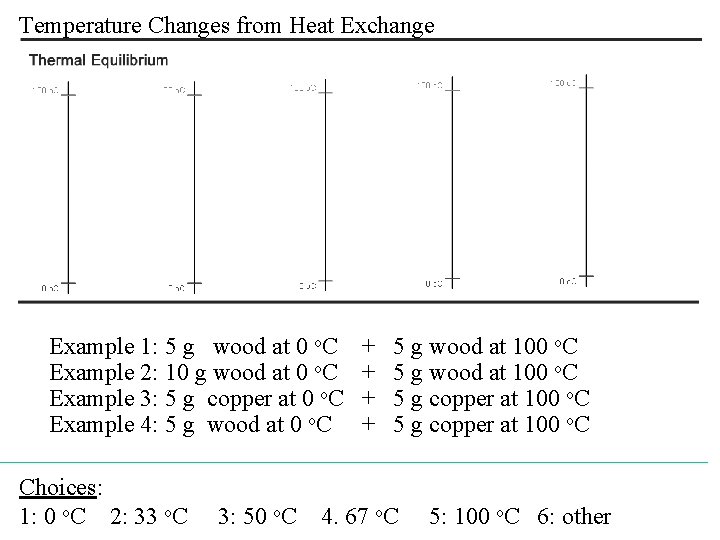
Temperature Changes from Heat Exchange Example 1: 5 g wood at 0 o. C Example 2: 10 g wood at 0 o. C Example 3: 5 g copper at 0 o. C Example 4: 5 g wood at 0 o. C Choices: 1: 0 o. C 2: 33 o. C 3: 50 o. C + + 5 g wood at 100 o. C 5 g copper at 100 o. C 4. 67 o. C 5: 100 o. C 6: other

What happens to thermal (heat) energy? When objects of different temperature meet: • Warmer object cools • Cooler object warms • Thermal energy is transferred • qwarmer = -qcooler

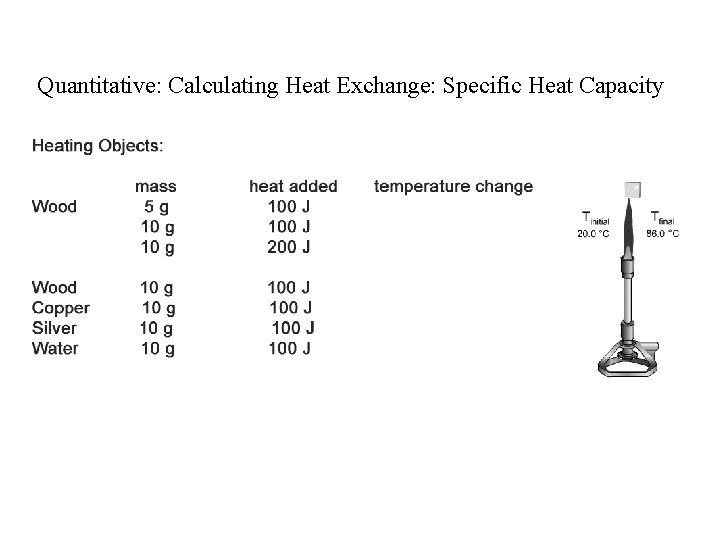
Quantitative: Calculating Heat Exchange: Specific Heat Capacity

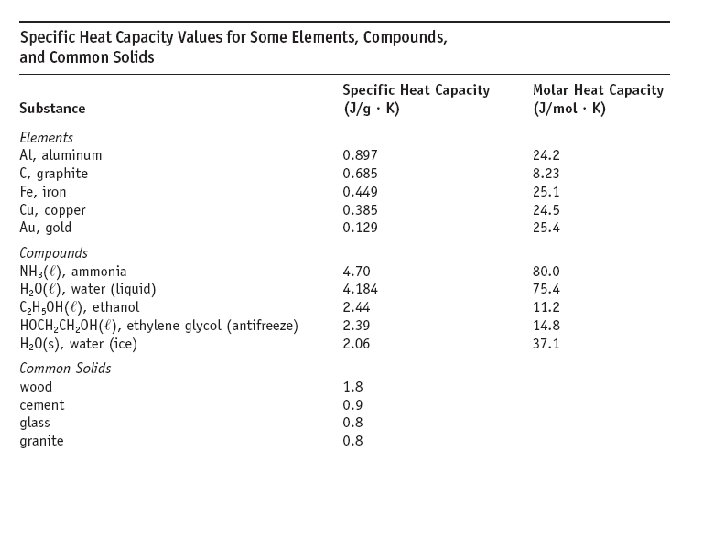
Specific Heat Capacity The energy required to heat one gram of a substance by 1 o. C. Usefulness: #J transferred = S. H. x #g x T How much energy is used to heat 250 g water from 17 o. C to 100 o. C?


What happens to thermal (heat) energy? When objects of different temperature meet: • Warmer object cools • Cooler object warms • Thermal energy is transferred • qwarmer = -qcooler specific heat x mass x T = specific heat x mass x T warmer object cooler object

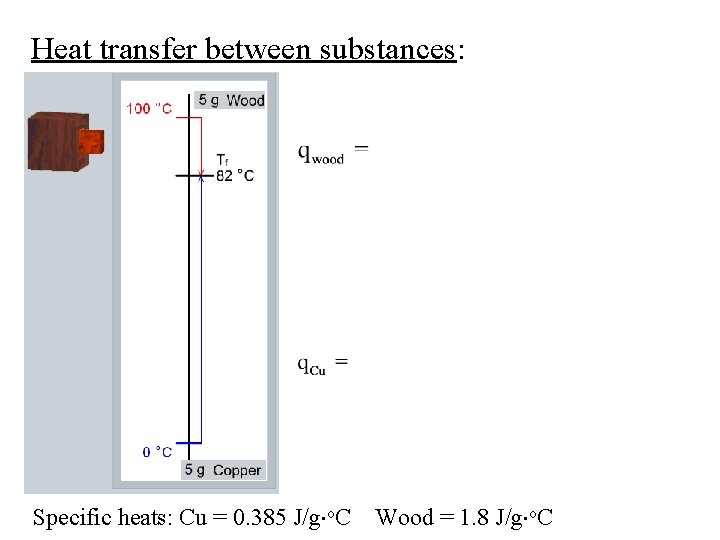
Heat transfer between substances: Specific heats: Cu = 0. 385 J/g o. C Wood = 1. 8 J/g o. C

Conceptually Easy Example with Annoying Algebra: If we mix 250 g H 2 O at 95 o. C with 50 g H 2 O at 5 o. C, what will the final temperature be?

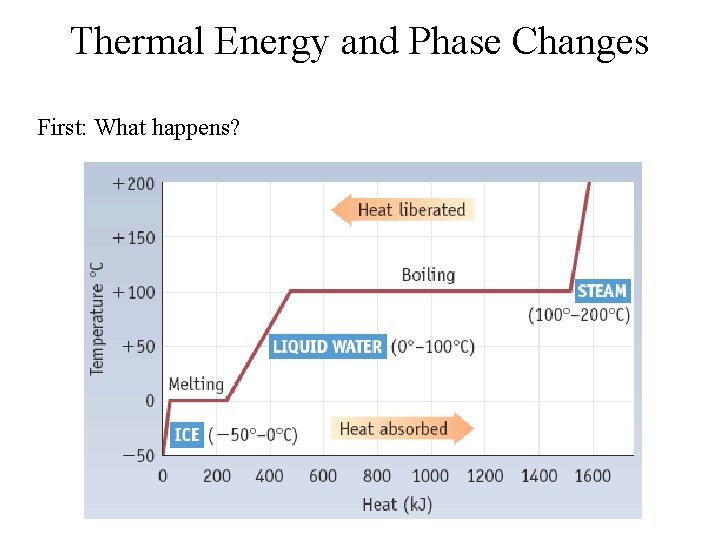
Thermal Energy and Phase Changes First: What happens?

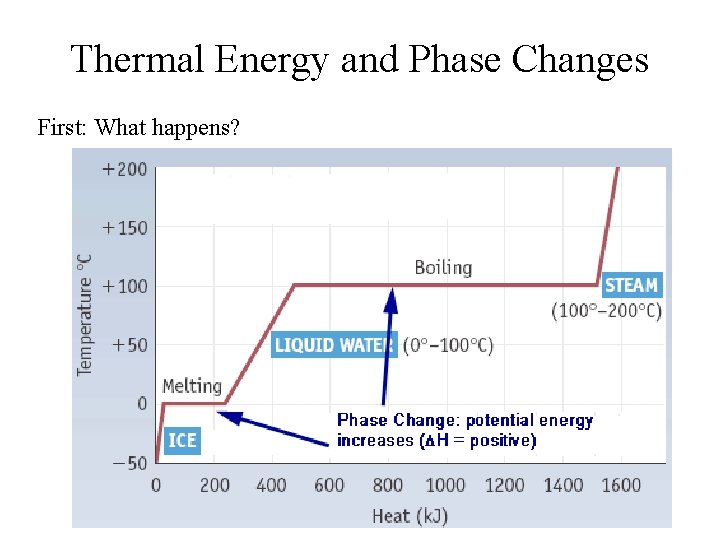
Thermal Energy and Phase Changes First: What happens?

Thermal Energy and Phase Changes First: What happens?

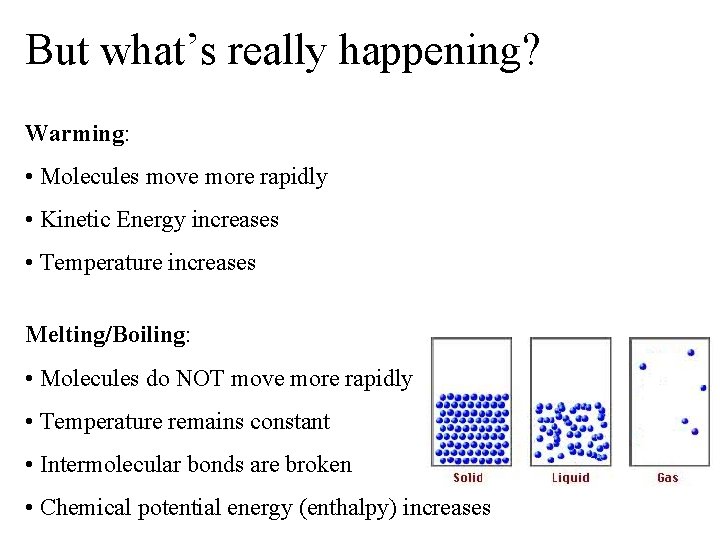
But what’s really happening? Warming: • Molecules move more rapidly • Kinetic Energy increases • Temperature increases Melting/Boiling: • Molecules do NOT move more rapidly • Temperature remains constant • Intermolecular bonds are broken • Chemical potential energy (enthalpy) increases

Energy and Phase Changes: Quantitative Treatment Melting: Heat of Fusion (DHfus) for Water: 333 J/g Boiling: Heat of Vaporization (DHvap) for Water: 2256 J/g

Total Quantitative Analysis Convert 40. 0 g of ice at – 30 o. C to steam at 125 o. C Warm ice: (Specific heat = 2. 06 J/g-o. C) Melt ice: Warm water (s. h. = 4. 18 J/g-o. C)

Total Quantitative Analysis Convert 40. 0 g of ice at – 30 o. C to steam at 125 o. C Boil water: Warm steam (s. h. = 1. 92 J/g-o. C)

Energy and Chemical Reactions Lots of different types of energy. We use Enthalpy: Heat exchanged under constant pressure.

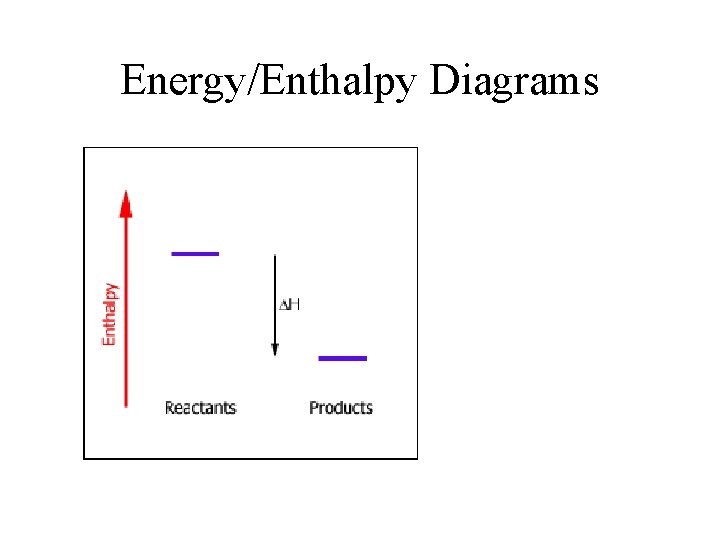
Energy/Enthalpy Diagrams

Some Examples of Enthalpy Change 2 C(s) + 2 H 2(g) C 2 H 4(g) H = +52 k. J

Enthalpy Change and Chemical Reactions H is usually more complicated, due to solvent and solid interactions. So, we measure H experimentally. Calorimetry Run reaction in a way that the heat exchanged can be measured. Use a “calorimeter. ”


Bomb Calorimetry Experiment N 2 H 4 + 3 O 2 2 NO 2 + 2 H 2 O Energy released = E absorbed by water + E absorbed by calorimeter Ewater = Ecalorimeter = Total E = H = energy/moles = 0. 500 g N 2 H 4 600 g water 420 J/o. C

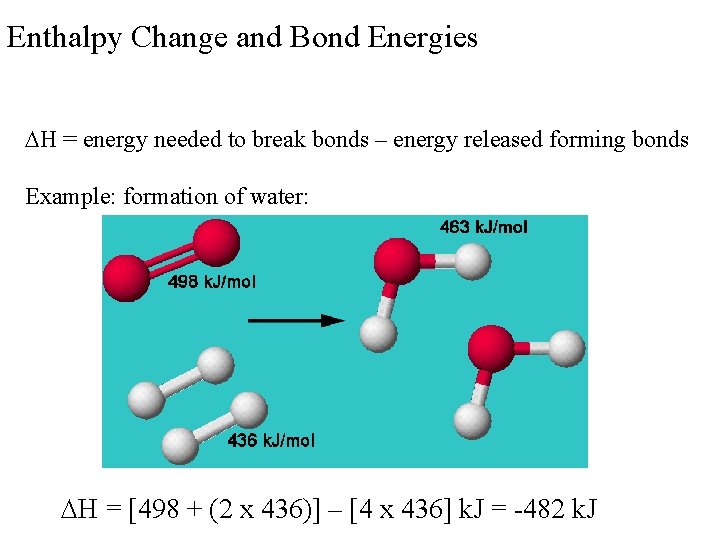
Enthalpy Change and Bond Energies H = energy needed to break bonds – energy released forming bonds Example: formation of water: H = [498 + (2 x 436)] – [4 x 436] k. J = -482 k. J

General Rule:

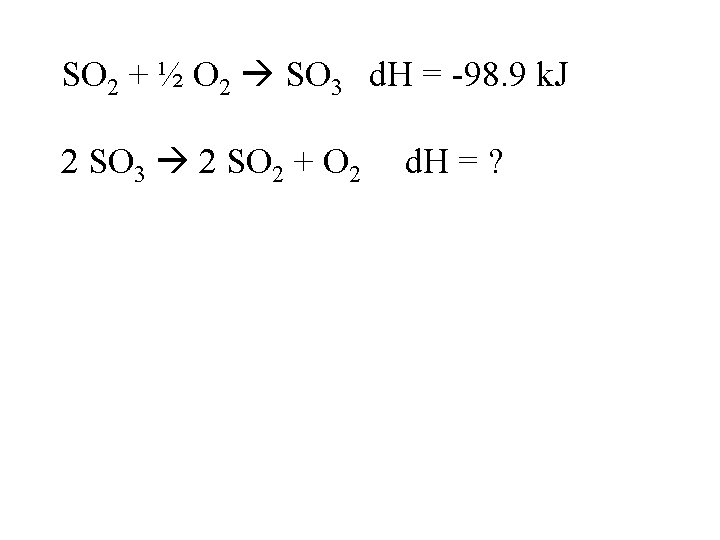
SO 2 + ½ O 2 SO 3 d. H = -98. 9 k. J 2 SO 3 2 SO 2 + O 2 d. H = ?

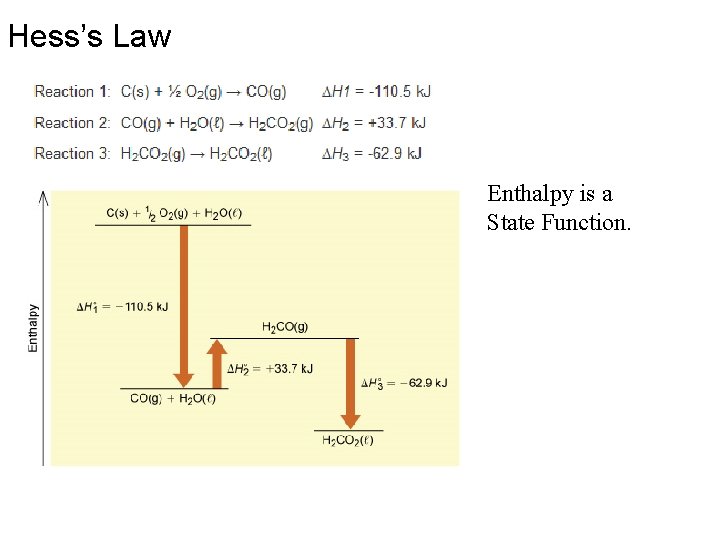
Hess’s Law Enthalpy is a State Function.

Thermochemistry Lab Calculations Goal: What is H for the formation of Mg. O from Mg(s) and O 2(g)? Mg(s) + ½ O 2(g) Mg. O(s) Data: From lab measurements: H = ? k. J/mol Mg(s) + 2 H+(aq) Mg 2+(aq) + H 2(g) H 1 = ______ k. J/mol Mg. O(s) + 2 H+(aq) Mg 2+(aq) + H 2 O(l) H 2 = ______ k. J/mol From a table: H 2(g) + ½ O 2(g) H 2 O(l) H 3 = -285. 8 k. J/mol Task: Find a way to add these three reactions to get the desired reaction. Manipulate the H values as needed, and add them.

Calculating Heat Production

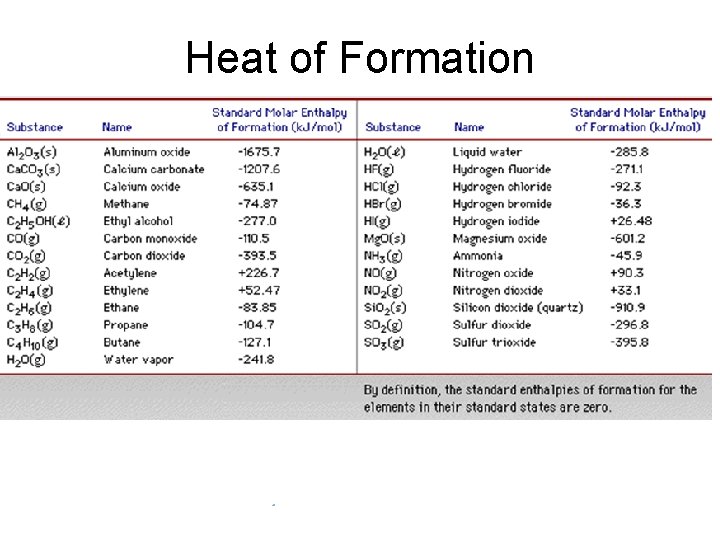
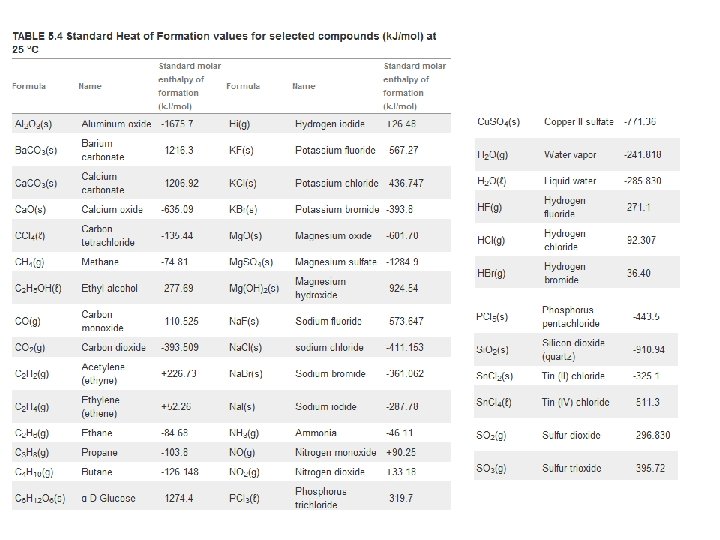
Heat of Formation


Heat of Formation: The general idea


Find the enthalpy change for burning ethyl alcohol