Balancing Act Atoms are not CREATED or DESTROYED

Balancing Act

Atoms are not CREATED or DESTROYED during a chemical reaction. This is the LAW OF CONSERVATION OF MASS. There must be the same number of atoms on each side of the equation. massbegin = massend http: //www. neok 12. com/php/watch. php? v=z. X 567 d 4 e 457 d 737 d 75667 b 0 a&t=Law-of-Conservation

Law of Conservation of Mass In other words, the number and type of atoms going INTO a reaction must be the same as the number and type of atoms coming OUT. If an equation obeys the Law of Conservation, it is balanced.
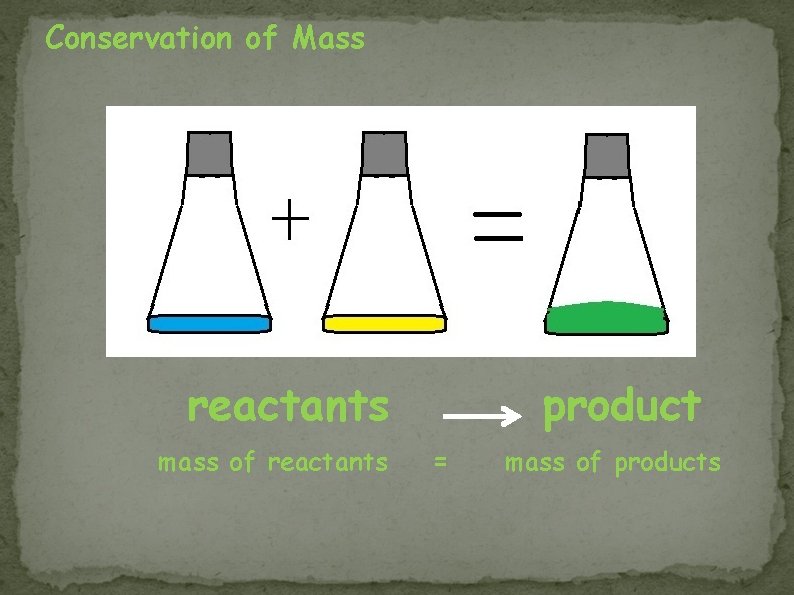
Conservation of Mass reactants mass of reactants product = mass of products
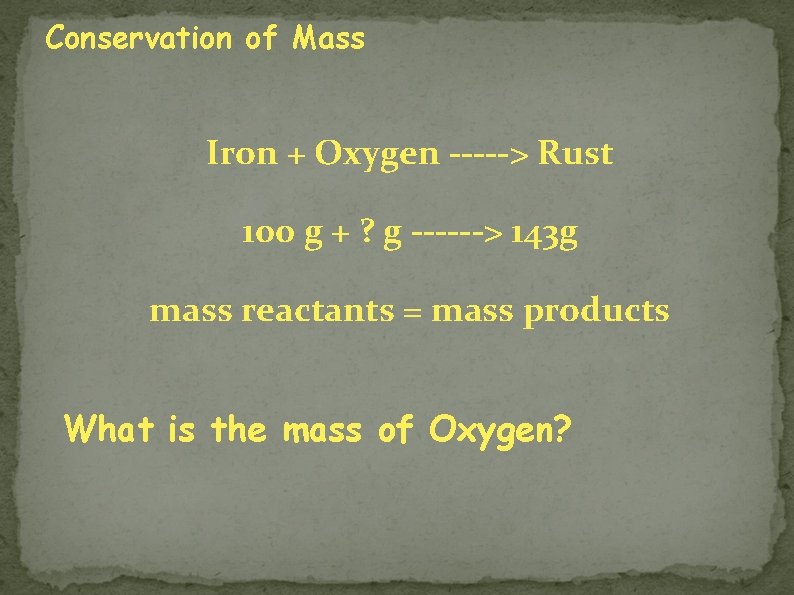
Conservation of Mass Iron + Oxygen -----> Rust 100 g + ? g ------> 143 g mass reactants = mass products What is the mass of Oxygen?
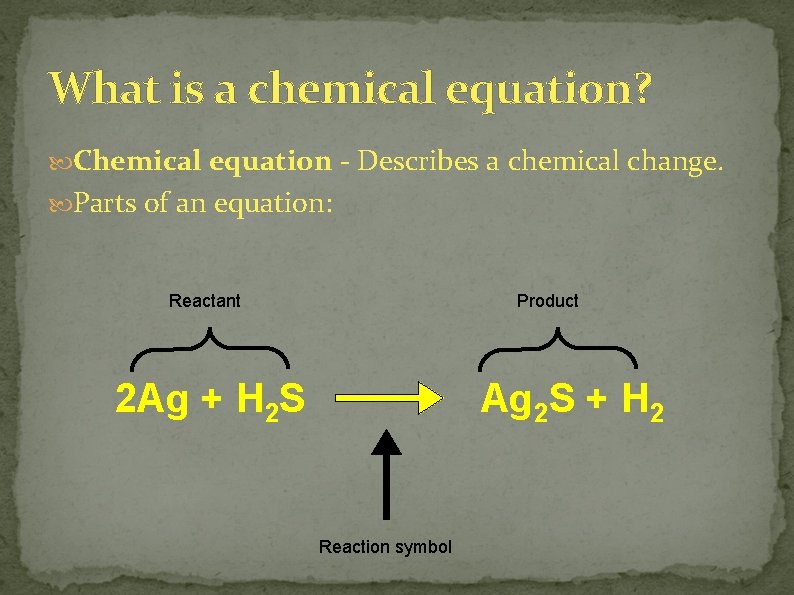
What is a chemical equation? Chemical equation - Describes a chemical change. Parts of an equation: Reactant Product 2 Ag + H 2 S Ag 2 S + H 2 Reaction symbol

Reactants and Products Reactant - The chemical(s) you start with before the reaction. Product - The new chemical(s) formed by the reaction.

To balance the chemical equation, you must place COEFFICIENTS in front of the chemical formulas in the equation. A coefficient is a number placed in front of a chemical formula in an equation that shows how many atoms are involved in a reaction. (Multiplier) Example: 2 Mg. O (2 is the coefficient) atoms There are 2 Magnesium and 2 Oxygen

Subscripts A subscript is a number written to the right of a chemical symbol and slightly below it. It tells how many atoms of that element. Example: H 2 O (2 is the subscript) This molecule has 2 hydrogen and 1 oxygen. You cannot ADD or CHANGE subscripts!

Subscripts and Coefficients Subscript - shows how many atoms of an element are in a molecule. EX: H 2 O 2 atoms of hydrogen (H) 1 atom of oxygen (O) Coefficient - shows how many molecules there are of a particular chemical. EX: 3 H 2 O Means there are 3 water molecules.
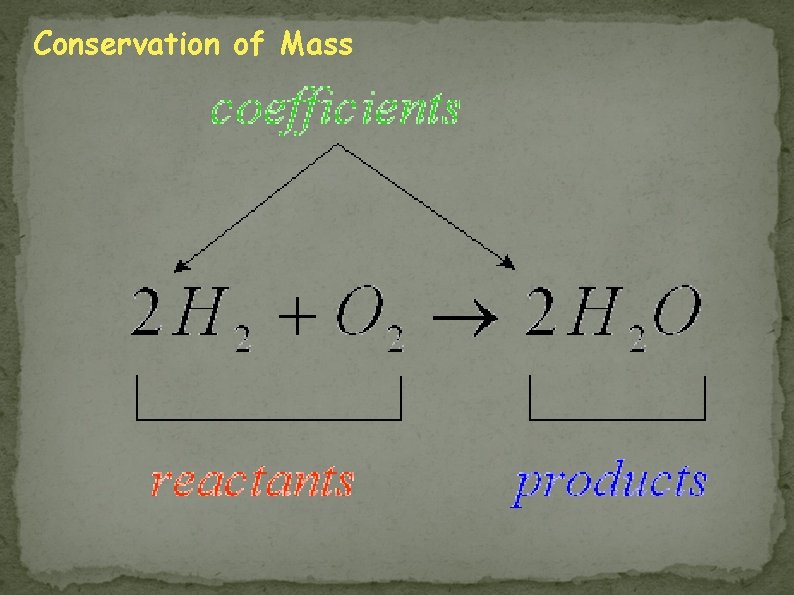
Conservation of Mass

A Chemical Reaction 2 H 2 + O 2 2 H 2 O
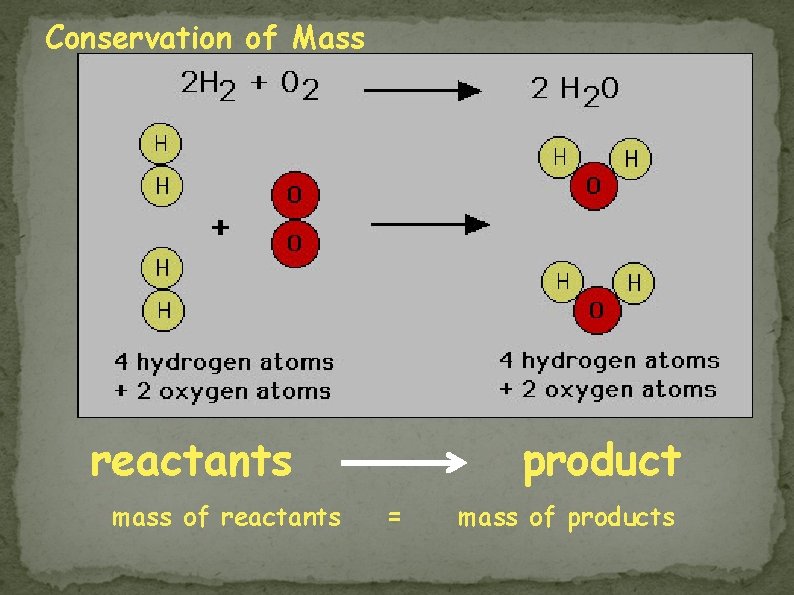
Conservation of Mass reactants mass of reactants product = mass of products

An Unbalanced Equation CH 4 + O 2 CO 2 + H 2 O Reactant Side 1 carbon atom 4 hydrogen atoms 2 oxygen atoms Product Side 1 carbon atom 2 hydrogen atoms 3 oxygen atoms
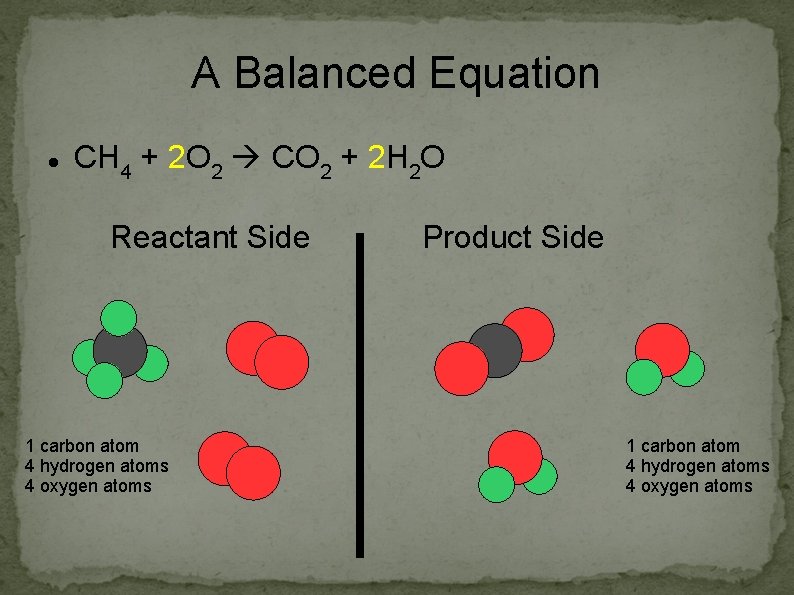
A Balanced Equation CH 4 + 2 O 2 CO 2 + 2 H 2 O Reactant Side 1 carbon atom 4 hydrogen atoms 4 oxygen atoms Product Side 1 carbon atom 4 hydrogen atoms 4 oxygen atoms
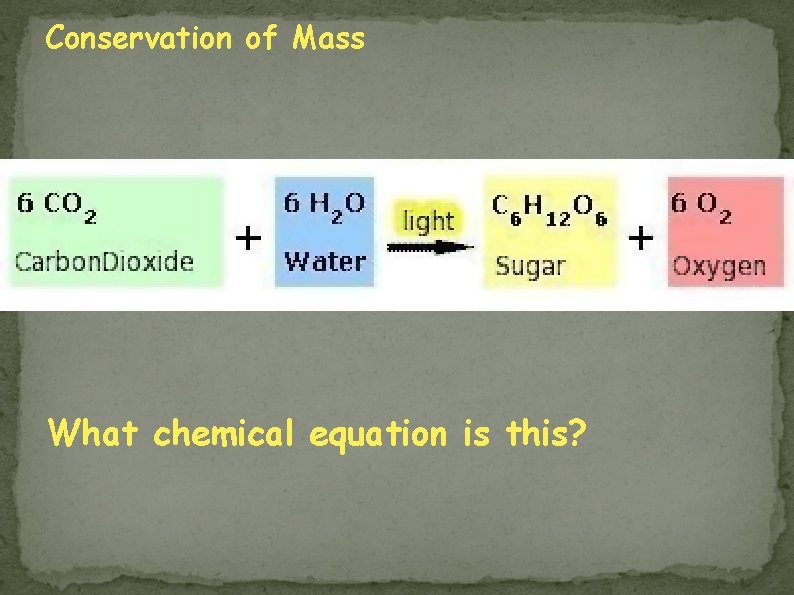
Conservation of Mass What chemical equation is this?

BALANCING EQUATIONS STEPS 1. Determine the number of atoms for each element. 2. Pick an element that is not equal on both sides of the equation. 3. Place a coefficient in front of the formula with that element and adjust your counts. 4. Continue adding coefficients to get the same number of atoms of each element on each side.

Rules of the Game 1. Matter cannot be created or destroyed. 2. Subscripts cannot be added, removed, or changed. 3. You can only change coefficients. 4. Coefficients can only go in front of chem. formulas. . . NEVER in the middle of a formula. A few extra tips: Try balancing big formulas first; save free elements for last. If the same polyatomic ion appears on both sides of the equation, it’s usually okay to treat it as one unit. There is no one particular way to balance equations. Some equations are harder to balance than others and might require some creativity to solve.

Balancing Equations Balance the following equation by adjusting coefficients. N 2 + 3 H 2 2 NH 3 reactants products N 2 21 H 6 2 63
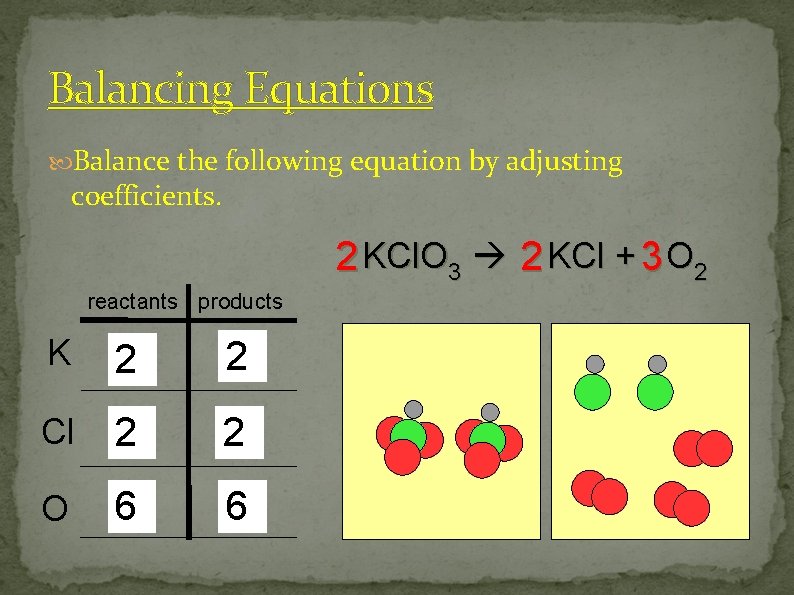
Balancing Equations Balance the following equation by adjusting coefficients. 2 KCl. O 3 2 KCl + 3 O 2 reactants products K 1 2 12 Cl 1 2 O 3 6 2 6

Balancing Chemical Equations Practice

Example 1

Example 2
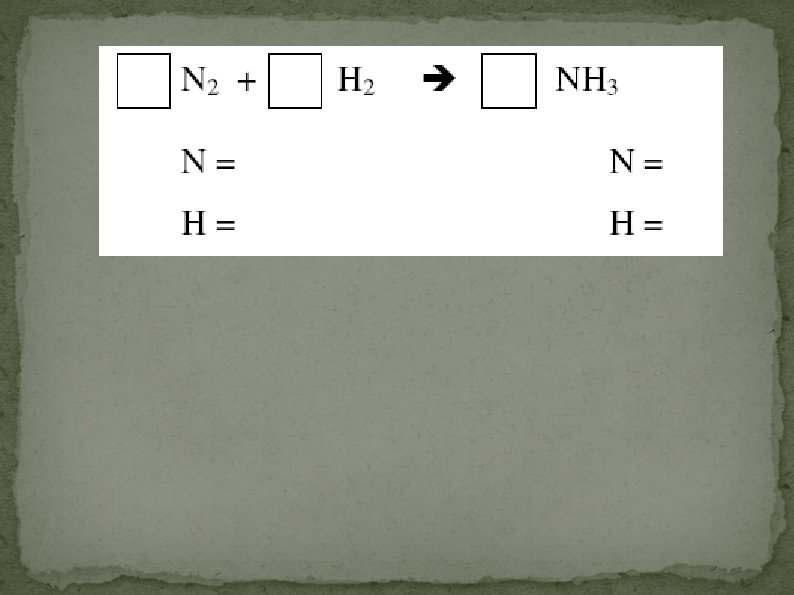



Balancing Equations Balance the following equation: 2 C 2 H 6 + 7 O 2 4 CO 2 + 6 H 2 O

Balancing Equations Balance the following equation: 4 Fe +3 O 2 2 Fe 2 O 3

Conservation of Mass Videos http: //www. neok 12. com/php/watch. php? v=z. X 567 d 4 e 457 d 737 d 75667 b 0 a &t=Law-of-Conservation http: //www. neok 12. com/php/watch. php? v=z. X 780 d 5 e 7806764057657 b 45 &t=Law-of-Conservation Brain Pop http: //www. brainpop. com Conservation of Mass

Balancing Act Homework

Balancing Act Homework ANSWERS

Balancing Act Homework ANSWERS

Balancing Act Homework

Balancing Act Homework ANSWERS

Balancing Act Homework ANSWERS

Balancing Act Homework ANSWERS
- Slides: 36