First Law of Thermodynamics energy cannot be created

First Law of Thermodynamics • energy cannot be created or destroyed. • Therefore, the total energy of the universe is a constant. • Energy can, however, be converted from one form to another or transferred from a system to the surroundings or vice versa. 1 אנרגיה חופשית וספונטניות 14 -

Enthalpy/Entropy • Enthalpy is the heat absorbed by a system during a constant-pressure process. • Entropy is a measure of the randomness in a system. • Both play a role in determining whether a process is spontaneous. 2 אנרגיה חופשית וספונטניות 14 -

Spontaneous Processes • Spontaneous processes proceed without any outside assistance. • The gas in vessel A will spontaneously effuse into vessel B, but it will not spontaneously return to vessel A. • Processes that are spontaneous in one direction are nonspontaneous in the reverse direction. 3 אנרגיה חופשית וספונטניות 14 -

Experimental Factors Affect Spontaneous Processes • Temperature and pressure can affect spontaneity. • An example of how temperature affects spontaneity is ice melting or freezing. 4 אנרגיה חופשית וספונטניות 14 -
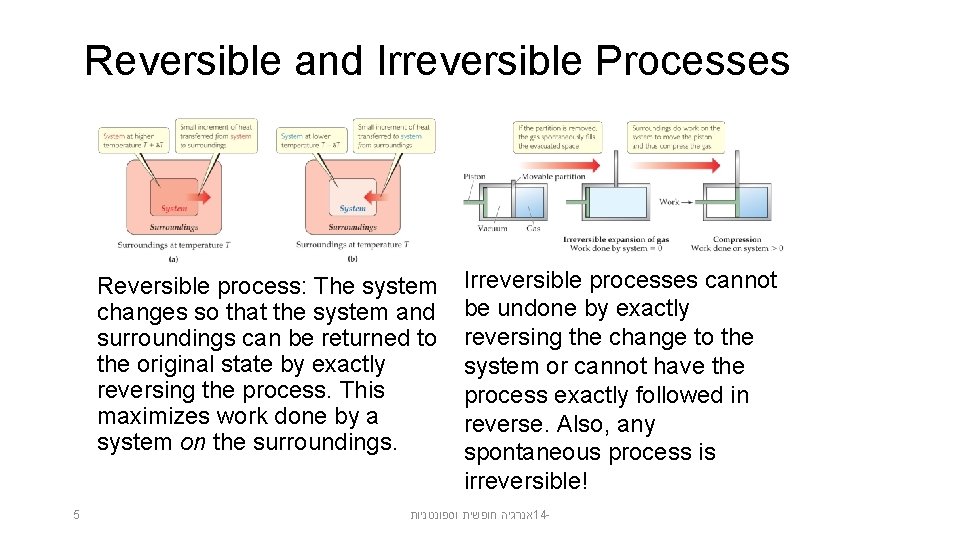
Reversible and Irreversible Processes Reversible process: The system changes so that the system and surroundings can be returned to the original state by exactly reversing the process. This maximizes work done by a system on the surroundings. 5 Irreversible processes cannot be undone by exactly reversing the change to the system or cannot have the process exactly followed in reverse. Also, any spontaneous process is irreversible! אנרגיה חופשית וספונטניות 14 -

Entropy • Entropy can be thought of as a measure of the randomness of a system. • It is a state function: • It can be found by heat transfer from surroundings at a given temperature: 6 אנרגיה חופשית וספונטניות 14 -

Second Law of Thermodynamics Ø The entropy of the universe increases in any spontaneous processes. Ø This results in the following relationships: 7 אנרגיה חופשית וספונטניות 14 -

Entropy on the Molecular Scale • Boltzmann described entropy on the molecular level. • Gas molecule expansion: Two molecules are in the apparatus above; both start in one side. What is the likelihood they both will end up there? (1/2)2 6. 02× 1023 • If one mole is used? (1/2) ! (No chance!) • Gases spontaneously expand to fill the volume given. • Most probable arrangement of molecules: approximately equal molecules in each side 8 אנרגיה חופשית וספונטניות 14 -

Statistical Thermodynamics • Thermodynamics looks at bulk properties of substances (the big picture). • We have seen what happens on the molecular scale. • How do they relate? • We use statistics (probability) to relate them. The field is called statistical thermodynamics. • Microstate: A single possible arrangement of position and kinetic energy of molecules 9 אנרגיה חופשית וספונטניות 14 -

Boltzmann’s Use of Microstates • Because there are so many possible microstates, we can’t look at every picture. • W represents the number of microstates. • Entropy is a measure of how many microstates are associated with a particular macroscopic state. • The connection between the number of microstates and the entropy of the system is: k = Boltzmann constant = 1. 38 × 10− 23 J/K 10 אנרגיה חופשית וספונטניות 14 -

Entropy Change • Since entropy is a state function, the final value minus the initial value will give the overall change. • In this case, an increase in the number of microstates results in a positive entropy change (more disorder). 11 אנרגיה חופשית וספונטניות 14 -
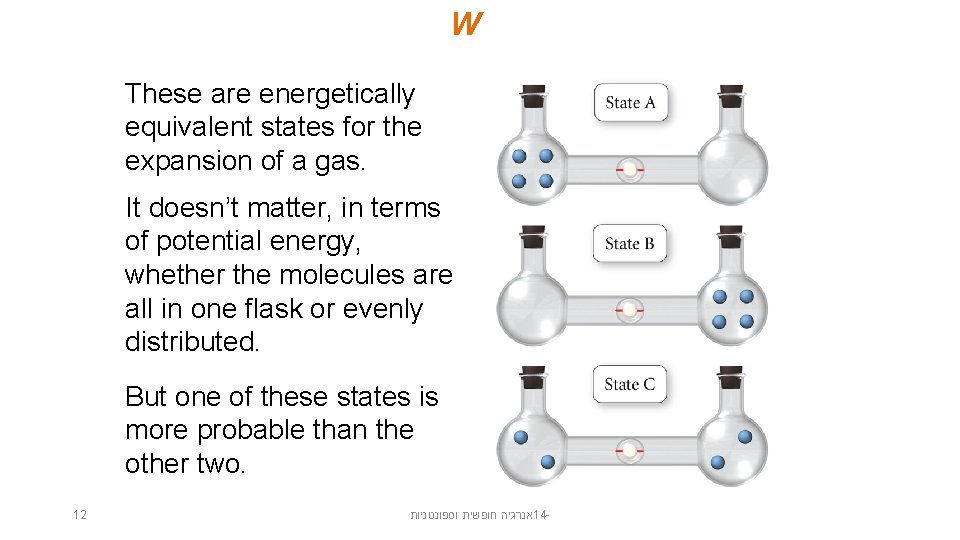
W These are energetically equivalent states for the expansion of a gas. It doesn’t matter, in terms of potential energy, whether the molecules are all in one flask or evenly distributed. But one of these states is more probable than the other two. 12 אנרגיה חופשית וספונטניות 14 -

Macrostates → Microstates This macrostate can be achieved through several different arrangements of the particles. 13 אנרגיה חופשית וספונטניות 14 -
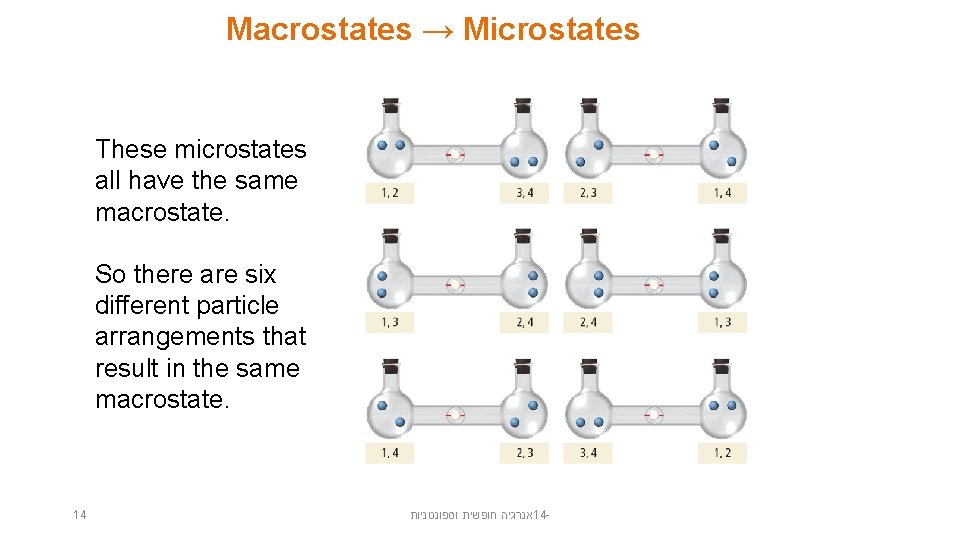
Macrostates → Microstates These microstates all have the same macrostate. So there are six different particle arrangements that result in the same macrostate. 14 אנרגיה חופשית וספונטניות 14 -
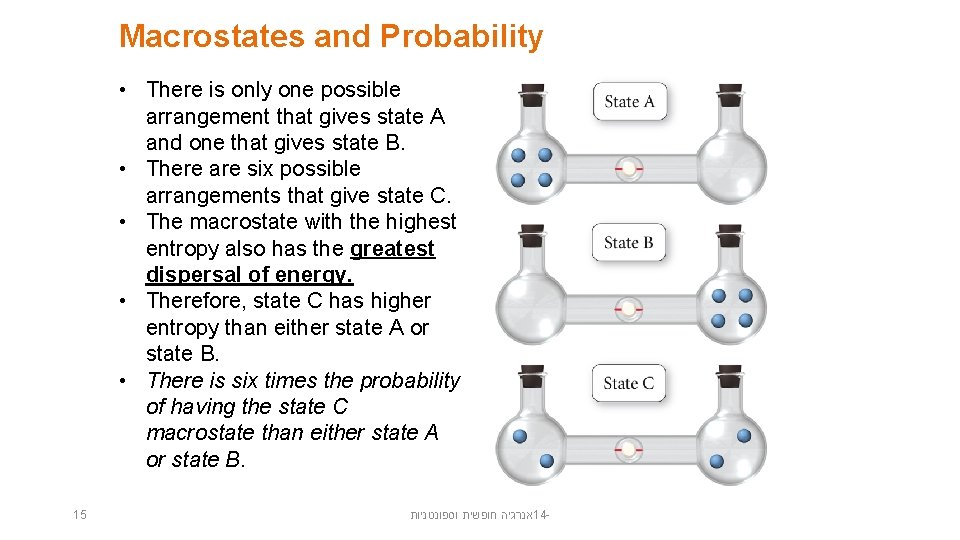
Macrostates and Probability • There is only one possible arrangement that gives state A and one that gives state B. • There are six possible arrangements that give state C. • The macrostate with the highest entropy also has the greatest dispersal of energy. • Therefore, state C has higher entropy than either state A or state B. • There is six times the probability of having the state C macrostate than either state A or state B. 15 אנרגיה חופשית וספונטניות 14 -

Effect of Volume and Temperature Change on the System • If we increase volume, there are more positions possible for the molecules. This results in more microstates, so increased entropy. • If we increase temperature, the average kinetic energy increases. This results in a greater distribution of molecular speeds. Therefore, there are more possible kinetic energy values, resulting in more microstates, increasing entropy. 16 אנרגיה חופשית וספונטניות 14 -
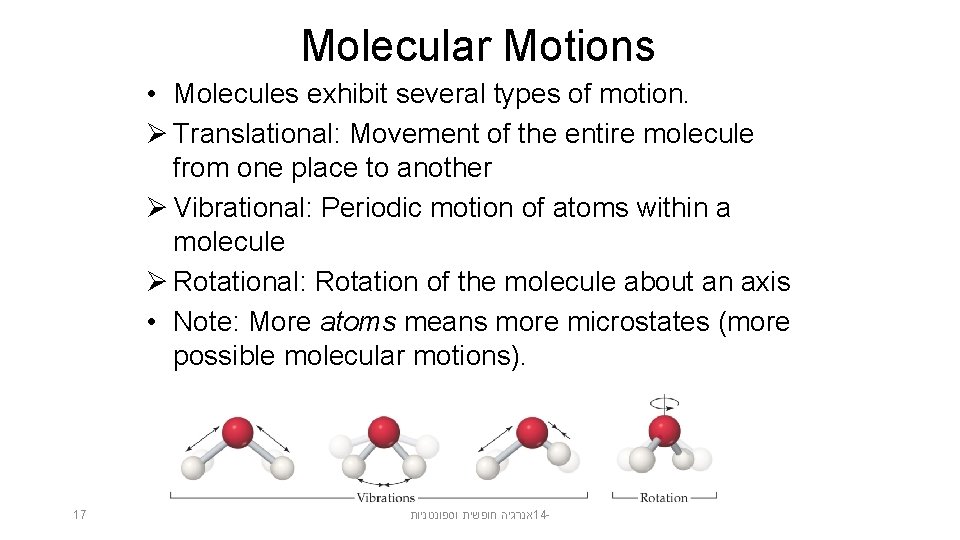
Molecular Motions • Molecules exhibit several types of motion. Ø Translational: Movement of the entire molecule from one place to another Ø Vibrational: Periodic motion of atoms within a molecule Ø Rotational: Rotation of the molecule about an axis • Note: More atoms means more microstates (more possible molecular motions). 17 אנרגיה חופשית וספונטניות 14 -

Entropy on the Molecular Scale • The number of microstates and, therefore, the entropy tend to increase with increases in Ø temperature. Ø volume. Ø the number of independently moving molecules. 18 אנרגיה חופשית וספונטניות 14 -

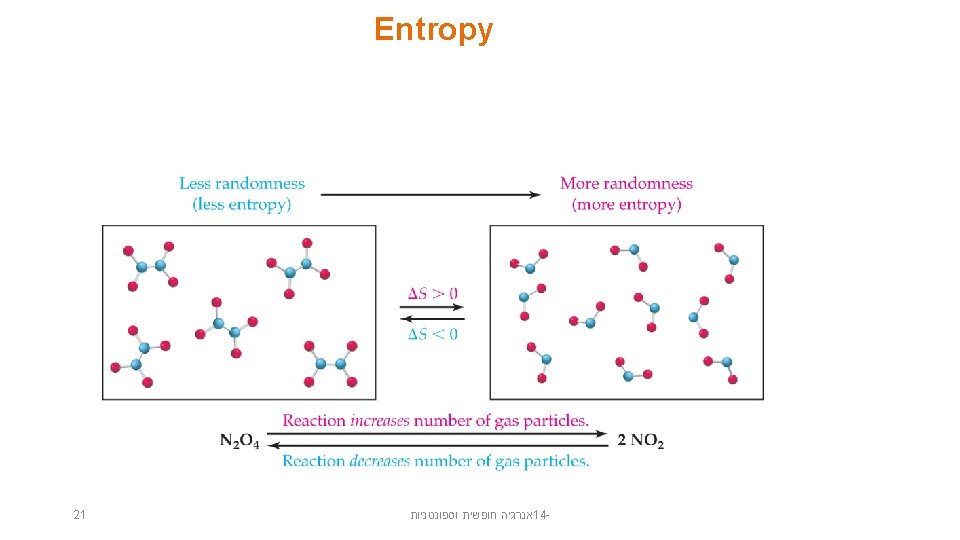
Entropy and Physical States • • • Ø Ø Ø 19 Entropy increases with the freedom of motion of molecules. S(g) > S(l) > S(s) Entropy of a system increases for processes where gases form from either solids or liquids or solutions form from solids. the number of gas molecules increases during a chemical reaction. אנרגיה חופשית וספונטניות 14 -
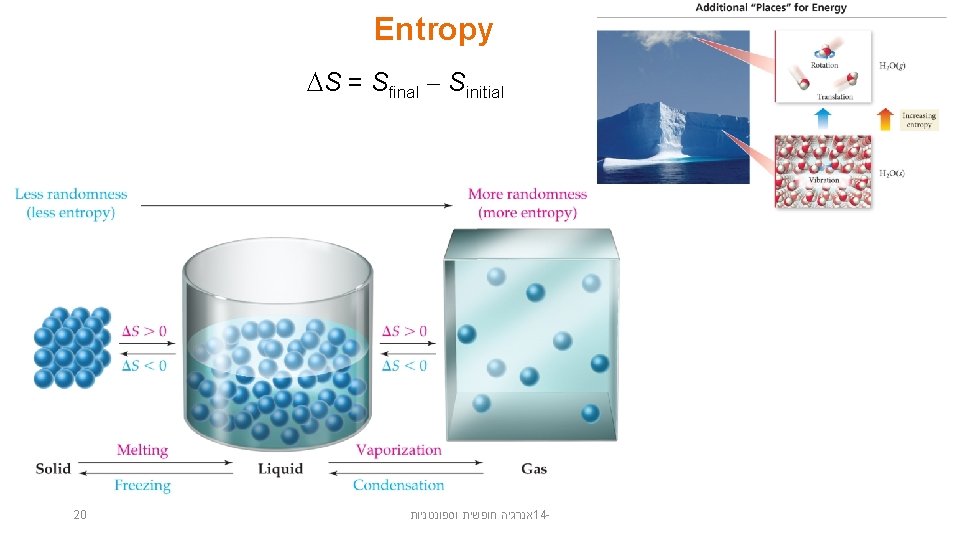
Entropy S = Sfinal Sinitial 20 אנרגיה חופשית וספונטניות 14 -
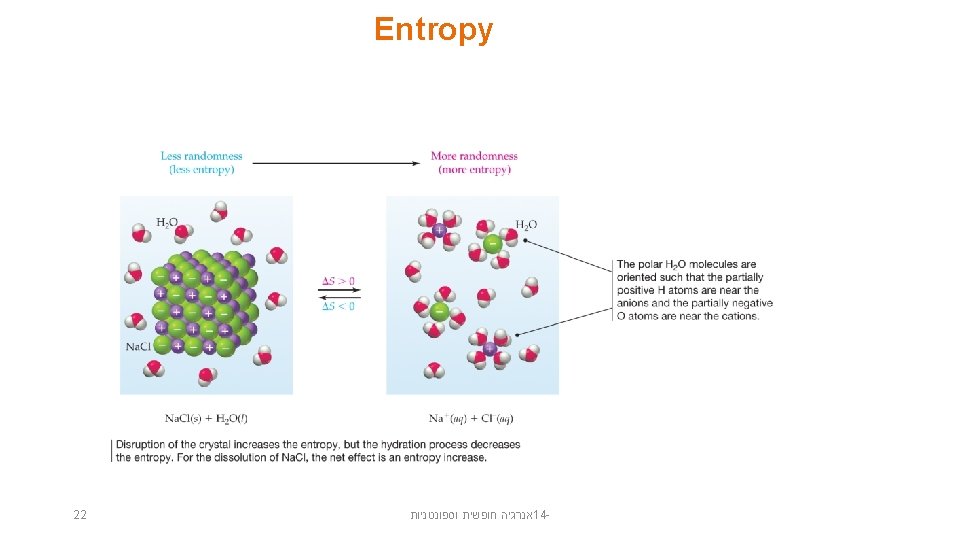

Entropy Changes in Surroundings • Heat that flows into or out of the system changes the entropy of the surroundings. • For an isothermal process • At constant pressure, qsys is simply H° for the system. 23 אנרגיה חופשית וספונטניות 14 -

Measuring Dispersal of E: Entropy Calculate Δfus. S when 25. 0 g of Al(s) melts at its normal melting point (660. 3 °C; 1 atm). Δfus. H° (Al)=10. 7 k. J mol-1 qrev Δfus. H° qp = ΔH Δfus. S = = Reversible if T T normal melting T = 660. 3 °C = 933. 5 K 25. 0 g n. Al = = 0. 9266 mol -1 26. 98 g mol 0. 9266 mol(10. 7 k. J mol-1) = 0. 01062 k. J K-1 Δfus. S = 933. 5 K 24 Δfus. S = 10. 6 J K-1 (system) אנרגיה חופשית וספונטניות 14 -

The Third Law of Thermodynamics: Absolute Entropy A perfect crystal, at absolute zero (0 K) has: • Minimum molecular motion (min. E dispersal). S=0 (perfect crystal at 0 K) (The 3 rd law of thermodynamics) Measure the heat to change from 0 K room-T (reversibly). Gives ΔS and the absolute S at room T. qrev ΔS = Sfinal – Sinitial = T Sroom T = ΔS – Sinitial = ΔS 25 אנרגיה חופשית וספונטניות 14 -

Absolute Entropy Values Standard molar entropies (S°), are measured by heating 1 mol of substance from 0 K to: • A specified T (often 25 °C = 298. 15 K) • At constant P = 1 bar S° has J K-1 mol-1 units. S° is always positive, for all materials. 26 אנרגיה חופשית וספונטניות 14 -
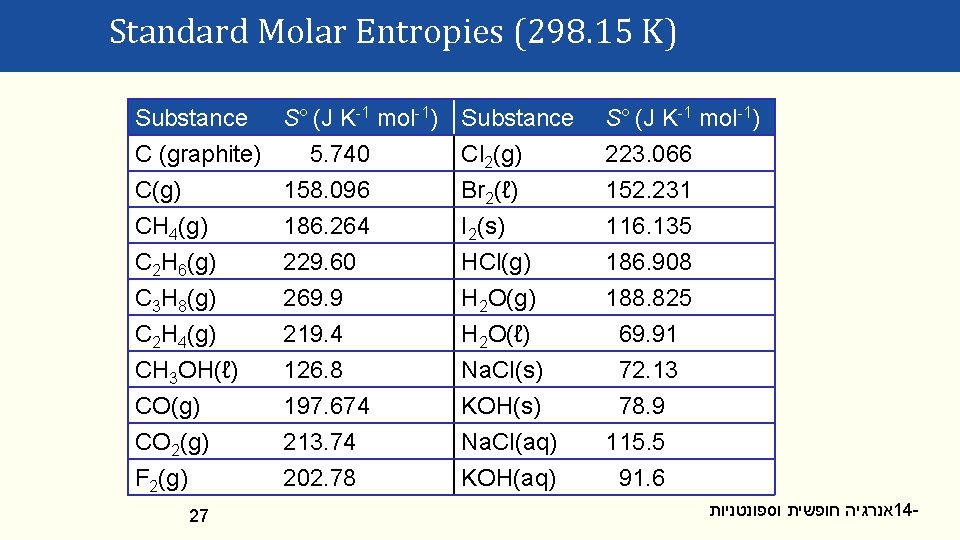
Standard Molar Entropies (298. 15 K) Substance S° (J K-1 mol-1) C (graphite) 5. 740 C(g) 158. 096 CH 4(g) 186. 264 Substance Cl 2(g) Br 2(ℓ) I 2(s) S° (J K-1 mol-1) 223. 066 152. 231 116. 135 C 2 H 6(g) C 3 H 8(g) C 2 H 4(g) CH 3 OH(ℓ) CO(g) CO 2(g) F 2(g) HCl(g) H 2 O(ℓ) Na. Cl(s) KOH(s) Na. Cl(aq) KOH(aq) 186. 908 188. 825 69. 91 72. 13 78. 9 115. 5 91. 6 27 229. 60 269. 9 219. 4 126. 8 197. 674 213. 74 202. 78 אנרגיה חופשית וספונטניות 14 -

Qualitative Guidelines for Entropy Sgas >> Sliquid > Ssolid Gas molecules have few restrictions on motion. Liquid molecules can slide past each other. Solid molecules vibrate about fixed lattice points. אנרגיה חופשית וספונטניות 14 - S[Cl 2(g)] = 223 S[Br 2(ℓ)] = 152 S[I 2(s)] = 116 J K-1 mol-1 28
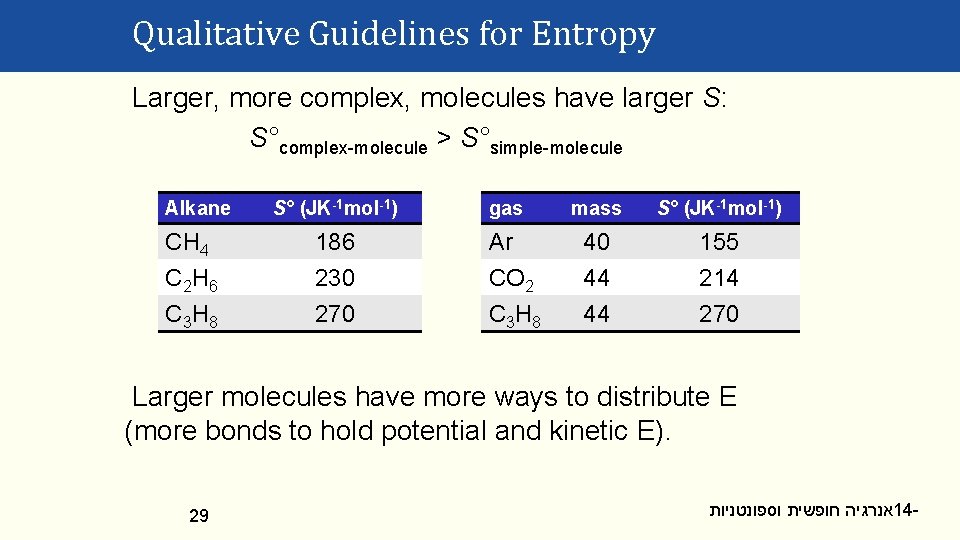
Qualitative Guidelines for Entropy Larger, more complex, molecules have larger S: S°complex-molecule > S°simple-molecule Alkane CH 4 C 2 H 6 C 3 H 8 S° (JK-1 mol-1) 186 230 270 gas Ar CO 2 C 3 H 8 mass S° (JK-1 mol-1) 40 44 44 155 214 270 Larger molecules have more ways to distribute E (more bonds to hold potential and kinetic E). 29 אנרגיה חופשית וספונטניות 14 -
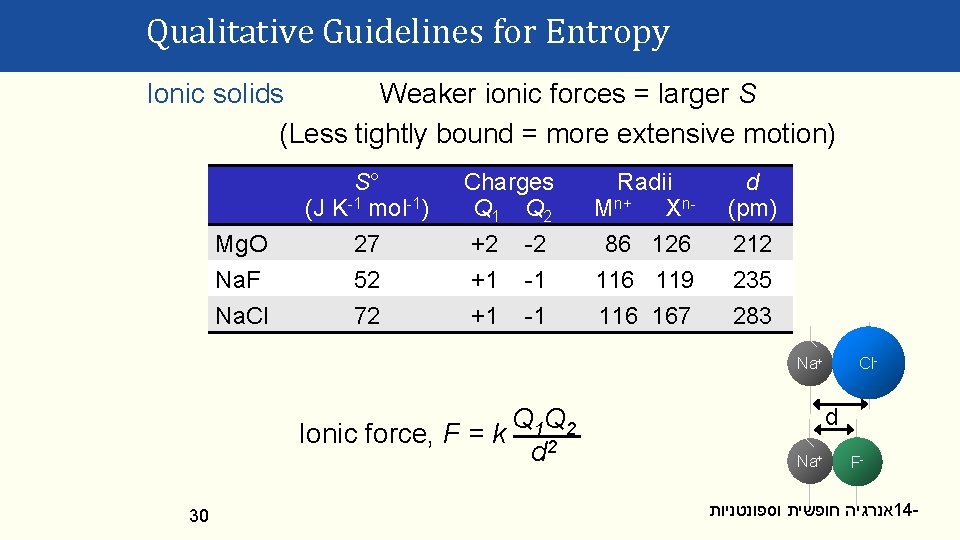
Qualitative Guidelines for Entropy Ionic solids Weaker ionic forces = larger S (Less tightly bound = more extensive motion) Mg. O Na. F Na. Cl S° (J K-1 mol-1) 27 52 72 Charges Q 1 Q 2 +2 -2 +1 -1 Radii Mn+ Xn 86 126 119 116 167 d (pm) 212 235 283 Cl- Na+ Q 1 Q 2 Ionic force, F = k 2 d 30 d Na+ F- אנרגיה חופשית וספונטניות 14 -

Qualitative Guidelines for Entropy Solid or liquid dissolving Larger matter dispersal. S usually* increases: molecule S°(pure) CH 3 COOH(ℓ) 160 NH 4 NO 3(s) 151 S°(aq) 179 260 ΔS° +19 +109 J K-1 mol-1 units *Strong solvation may increase order. ΔS < 0 is possible. 31 אנרגיה חופשית וספונטניות 14 -

Qualitative Guidelines for Entropy Gas dissolving Gas-molecule motion becomes restricted. ΔS < 0. Gas CO 2 H 2 CH 3 OH 32 S°(pure) 214 131 240 S°(aq) 118 58 133 ΔS° -96 -73 -107 אנרגיה חופשית וספונטניות 14 -

Predicting Entropy Changes Ca. CO 3(s) → Ca. O(s) + CO 2(g) ΔS = 161 J K-1 mol-1 Large entropy increase: • Solid → gas, ΔS > 0. • 1 molecule → 2 molecules, ΔS > 0. H 2(g) + F 2(g) → 2 HF(g) ΔS = 14 J K-1 mol-1 • All gases, S ≈ constant. • 2 reactant gases, 2 product gases, S ≈ constant. Actual ΔS shows a small increase. 33 אנרגיה חופשית וספונטניות 14 -

Predicting Entropy Changes Predict whether S will increase or decrease for: 2 CO(g) + O 2(g) → 2 CO 2(g) Decrease 3 gas molecules → 2 gas molecules. Na. Cl(s) → Na+(aq) + Cl-(aq) Increase Ions locked in a crystal → ions dispersed in water. H 2 O(ℓ) → H 2 O(s) Decrease Liquid molecules less restricted than solid molecules. אנרגיה חופשית וספונטניות 1434

Calculating Entropy Changes Entropy is a state function (like H). For a reaction: Δr. S° = (nproduct. S°product) – (nreactant. S°reactant) Example Methanol is a common fuel additive. Calculate Δr. S° at 298 K for its combustion: 2 CH 3 OH(ℓ) + 3 O 2(g) → 2 CO 2(g) + 4 H 2 O(ℓ) Look up S° values (J K-1 mol-1): CH 3 OH(ℓ) = 126. 8 O 2(g) = 205. 138 CO 2(g) = 213. 74 H 2 O(ℓ) = 69. 91 35 אנרגיה חופשית וספונטניות 14 -

Calculating Entropy Changes Example: Δr. S° at 298 K for: 2 CH 3 OH(ℓ) + 3 O 2(g) → 2 CO 2(g) + 4 H 2 O(ℓ) Δr. S° = (nproduct. S°product) – (nreactant. S°reactant) = (2 S°CO 2 + 4 S°H 2 O) – (2 S°CH 3 OH + 3 S°O 2) = [2(213. 74) + 4(69. 91)] – [2(126. 8) + 3(205. 14)] = 707. 12 – 869. 02 = – 161. 90 J K-1 mol-1 S decreases as expected: 3 gas + 2 ℓ → 2 gas + 4 ℓ 36 אנרגיה חופשית וספונטניות 14 -

Entropy & the 2 nd Law of Thermodynamics E dispersal is accompanied by an increase in disorder of a system: Increased disorder = increased S Second Law of Thermodynamics “The total entropy of the universe is continuously increasing. ” • The universe is slowly becoming more disordered. 37 אנרגיה חופשית וספונטניות 14 -

Entropy & the 2 nd Law of Thermodynamics The entropy of a system can increase or decrease. • Suniverse always goes up. • Suniverse = Ssystem + Ssurr (No subscript? S = Ssystem ). • If Ssystem falls, Ssurr must increase by a larger amount. qrev Δr. S°surr = T 38 = Δr. Hsurr T = -Δr. Hsys T אנרגיה חופשית וספונטניות 14 -

Entropy & the 2 nd Law of Thermodynamics Calculate Δr. S°univ for the combustion of octane in air: 2 C 8 H 18(g) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(ℓ) For the reaction Δr. H° = -11, 024 k. J mol-1 and Δr. S° = -1383. 9 J mol-1 K-1 at 298. 15 K. ° = Δr. S°sys + Δr. S°surr Δr. Suniv given -ΔHsys / T ° = -1383. 9 J/K + (+11, 024 x 103 J)/298. 15 K Δr. Suniv = -1383. 9 J/K + 36. 975 k. J/K 39 = +35, 591 J/K אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy Neither entropy (S), nor enthalpy (H ), alone can predict whether a reaction is product favored. Spontaneous (product favored) reactions can: • Be exothermic or endothermic. • Increase or decrease Ssystem. The Gibbs free energy (G), combines H and S. Δr. G does predict if a reaction is product favored or not. 40 אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy For a constant T change, ΔG is equal to: Δr. G = Δr. H - TΔr. S Josiah Willard Gibbs If G: Decreases (Δr. G < 0) a reaction is product favored Increases (Δr. G > 0) a reaction is reactant favored (at constant P and T). 41 אנרגיה חופשית וספונטניות 14 -
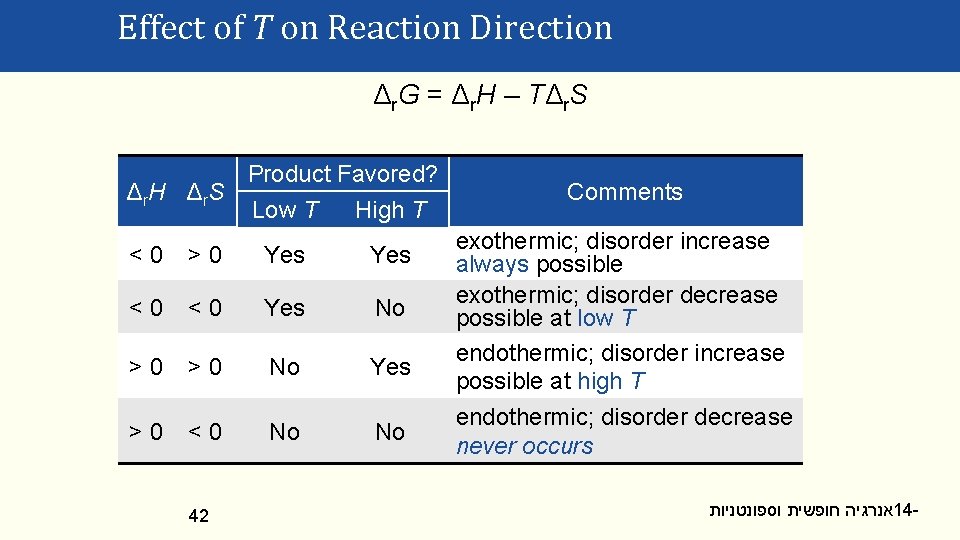
Effect of T on Reaction Direction Δ r. G = Δ r. H – T Δ r. S Product Favored? Δ r. H Δ r. S Low T High T <0 >0 Yes <0 <0 Yes No >0 >0 No Yes >0 <0 No No 42 Comments exothermic; disorder increase always possible exothermic; disorder decrease possible at low T endothermic; disorder increase possible at high T endothermic; disorder decrease never occurs אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy Δr. G° can be calculated from Δr. H° and Δr. S° values… … or from Gibbs free energies of formation (Δf. G°). Δr. G° = (nproductΔf. G°product) – (nreactantΔf. G°reactant) Δf. G° equals: • Δr. G to make 1 mol of compound from its elements. • 0 for an element in its most stable state (like Δf. H°) 43 אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy Calculate Δr. G° for the following reaction at 25°C: CO(g) + 2 H 2(g) → CH 3 OH(ℓ) Δf. G° values: CO(g) = -137. 2 k. J/mol CH 3 OH(ℓ) = -166. 3 k. J/mol ΔG° = Δf. G°CH 3 OH – (2Δf. G°H 2 + 1Δf. G°CO) element in its standard state = -166. 3 k. J/mol – [2(0) + 1(-137. 2 k. J/mol)] = -29. 1 k. J/mol Product favored 44 אנרגיה חופשית וספונטניות 14 -

Effect of T on Reaction Direction A reactions will change from reactant to productfavored (or visa versa) when Δr. G° = 0. Δ r. G° = 0 = Δ r. H° − TΔ r. S° Δr. H° and Δr. S° typically remain ≈constant over large T ranges and thus the “switchover” T can be calculated: 0 = Δ r. H° − TΔ r. S° Δ r. H° = TΔ r. S° Δr. H° T= Δr. S° 45 אנרגיה חופשית וספונטניות 14 -
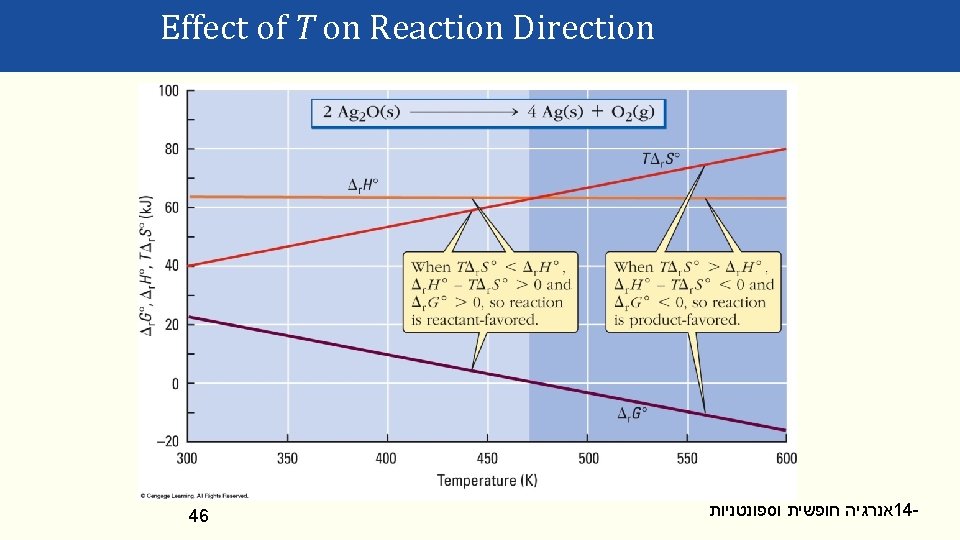
Effect of T on Reaction Direction 46 אנרגיה חופשית וספונטניות 14 -

Effect of T on Reaction Direction At what T will the following reaction become product favored? CH 4(g) + H 2 O(g) CO(g) + 3 H 2(g) Δf. H° (k. J/mol) − 74. 81 S° (J K-1 mol-1) 186. 26 − 241. 82 188. 83 − 110. 53 197. 67 0 130. 68 Δr. S°=[197. 67 +3(130. 68)] − [186. 26 + 188. 83] J K-1 mol-1 = 214. 62 J K-1 mol-1 47 Δr. H° = [-110. 53 + 3(0)] − [-74. 81 + -241. 82] k. J mol-1 = 206. 10 k. J mol-1 It will switch direction at: 2. 0610 x 105 J mol-1 Δ r. H T= = = 960. 30 K -1 -1 214. 62 J K mol Δ r. S אנרגיה חופשית וספונטניות 14 -

Variation of G During a Reaction Δr. G° equals the change in G which occurs when pure reactants are completely converted to pure products. How does G vary during a reaction? Consider: Na. OH(s) + CO 2(g) Na. HCO 3(s) Δr. G°= − 77. 2 k. J/mol The extent of reaction (x) equals the fraction of reactants converted to products. 48 אנרגיה חופשית וספונטניות 14 -
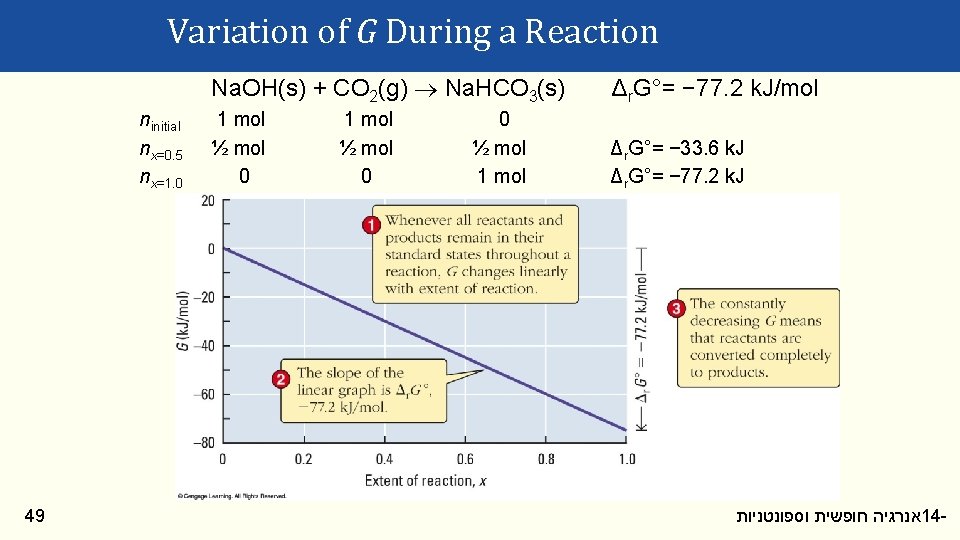
Variation of G During a Reaction ninitial nx=0. 5 nx=1. 0 49 Na. OH(s) + CO 2(g) Na. HCO 3(s) Δr. G°= − 77. 2 k. J/mol 1 mol ½ mol 0 Δr. G°= − 33. 6 k. J Δr. G°= − 77. 2 k. J 1 mol ½ mol 0 0 ½ mol 1 mol אנרגיה חופשית וספונטניות 14 -
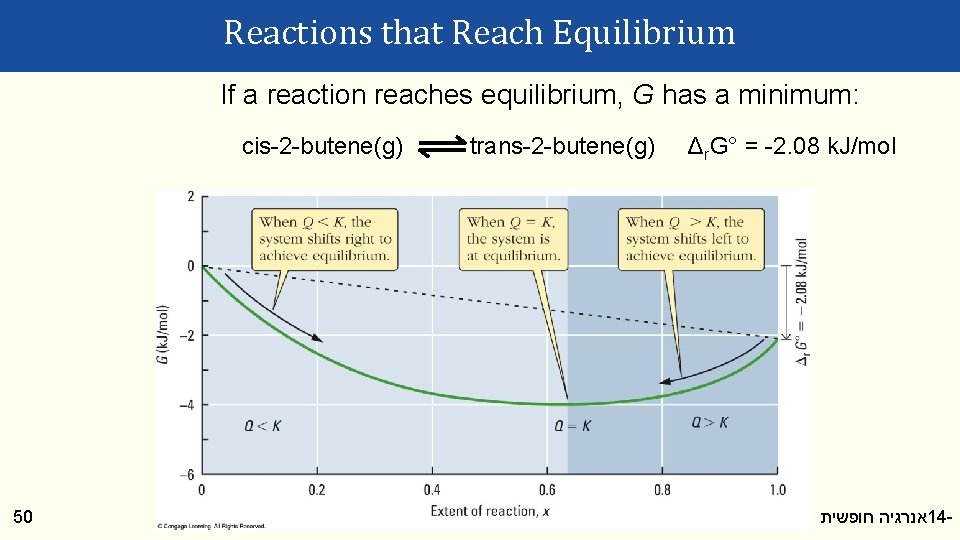
Reactions that Reach Equilibrium If a reaction reaches equilibrium, G has a minimum: cis-2 -butene(g) 50 trans-2 -butene(g) Δr. G° = -2. 08 k. J/mol אנרגיה חופשית וספונטניות 14 -

Free Energy and Equilibrium Under any conditions, standard or nonstandard, the free energy change can be found this way: G = G° + RT ln Q (Under standard conditions, concentrations are 1 M, so Q = 1 and ln Q = 0; the last term drops out. ) 51 אנרגיה חופשית וספונטניות 14 -

Free Energy and Equilibrium • At equilibrium, Q = K, and G = 0. • The equation becomes 0 = G° + RT ln K • Rearranging, this becomes G° = RT ln K or G /RT K=e 52 אנרגיה חופשית וספונטניות 14 -

Reactions that Reach Equilibrium Gibbs free energy G and the equilibrium constant K both show if a reaction is product-favored and are related. Δr. G° = −RT ln K° K° must be unitless. Each quantity in the K° expression is divided by its standard state value: • concentrations by 1 mol L-1 (solids and liquids). • pressures by 1 bar (gas reactions). 53 אנרגיה חופשית וספונטניות 14 -

Reactions that Reach Equilibrium Evaluate K° (298 K) for the reaction: 2 NO 2(g) N 2 O 4(g) Δr. G° = Δf. G°(N 2 O 4) - 2Δf. G°(NO 2) = 97. 89 – 2(51. 31) k. J/mol = -4. 73 k. J/mol 3 J/mol Δr. G° -4. 73 x 10 ln K° = – =– = 1. 91 -1 -1 RT (8. 314 J K mol )(298 K) K° = 6. 75 54 אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy & Maximum Work Δr. G = maximum useful work that can be done by a reaction on its surroundings (constant T and P). Δr. G = wsystem = - wmax “free” energy = available energy For: 2 H 2(g) + O 2(g) 2 H 2 O(g) Δr. G°= -474. 3 k. J/mol Every 2 moles of H 2 consumed can do up to 474. 3 k. J of work. 55 אנרגיה חופשית וספונטניות 14 -

Free Energy and Reversible Reactions • The change in free energy is a theoretical limit as to the amount of work that can be done. • If the reaction achieves its theoretical limit, it is a reversible reaction. 56 אנרגיה חופשית וספונטניות 14 -

Gibbs Free Energy & Maximum Work Δr. G also equals the minimum work required to cause a reactant-favored process to occur. 2 H 2 O(g) 2 H 2(g) + O 2(g) Δr. G°= 474. 3 k. J/mol A minimum of 474. 3 k. J of work must be used to produce 2 mol of H 2 from liquid water. 57 אנרגיה חופשית וספונטניות 14 -
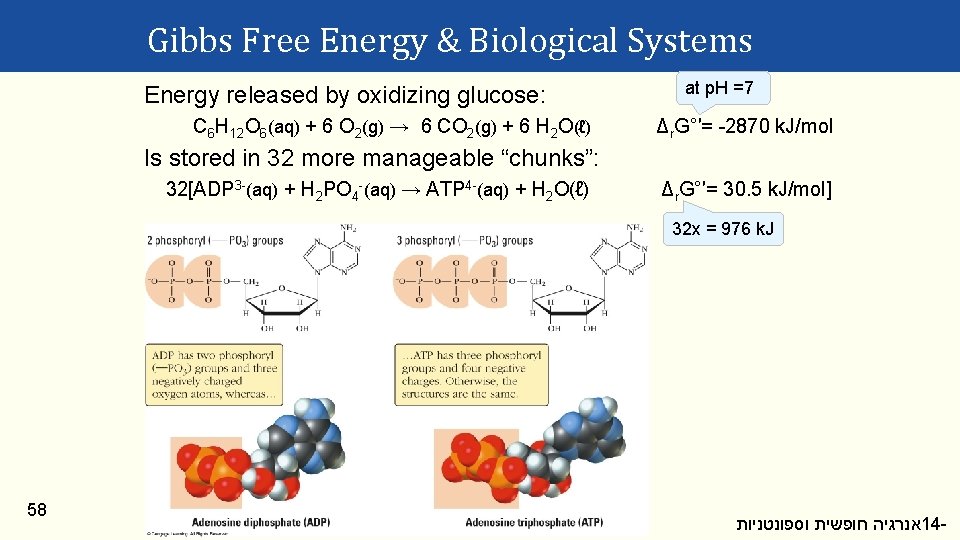
Gibbs Free Energy & Biological Systems Energy released by oxidizing glucose: C 6 H 12 O 6(aq) + 6 O 2(g) → 6 CO 2(g) + 6 H 2 O(ℓ) at p. H =7 Δr. G°′= -2870 k. J/mol Is stored in 32 more manageable “chunks”: 32[ADP 3 -(aq) + H 2 PO 4 -(aq) → ATP 4 -(aq) + H 2 O(ℓ) Δr. G°′= 30. 5 k. J/mol] 32 x = 976 k. J 58 אנרגיה חופשית וספונטניות 14 -
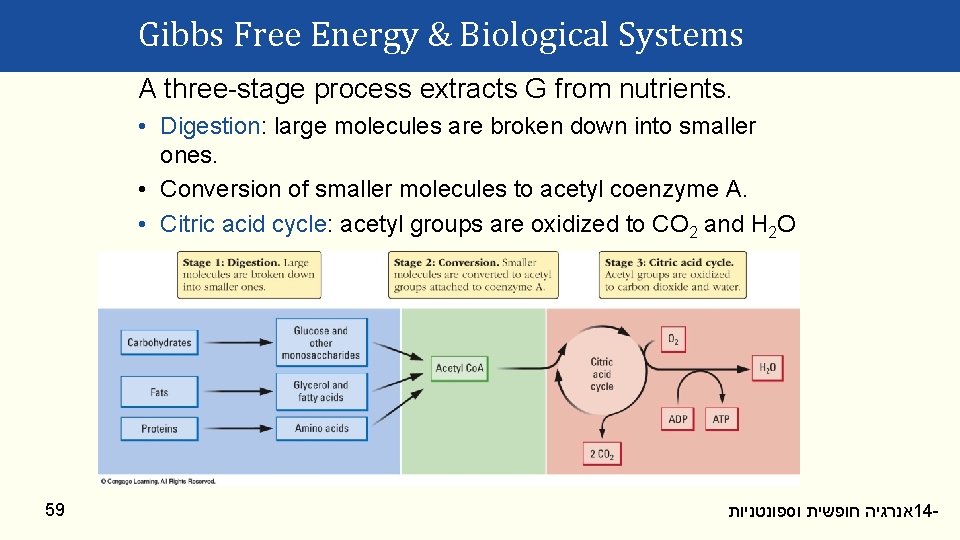
Gibbs Free Energy & Biological Systems A three-stage process extracts G from nutrients. • Digestion: large molecules are broken down into smaller ones. • Conversion of smaller molecules to acetyl coenzyme A. • Citric acid cycle: acetyl groups are oxidized to CO 2 and H 2 O 59 אנרגיה חופשית וספונטניות 14 -
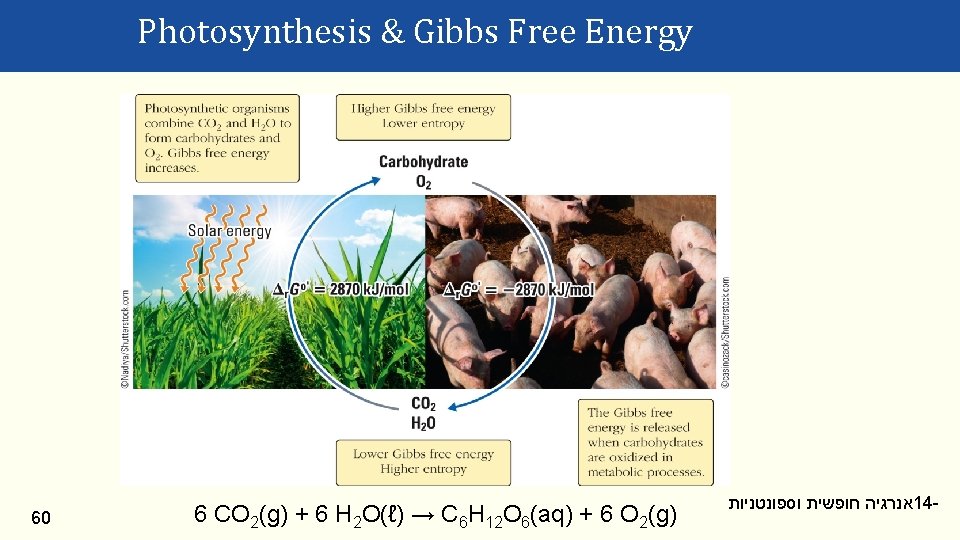
Photosynthesis & Gibbs Free Energy 60 6 CO 2(g) + 6 H 2 O(ℓ) → C 6 H 12 O 6(aq) + 6 O 2(g) אנרגיה חופשית וספונטניות 14 -
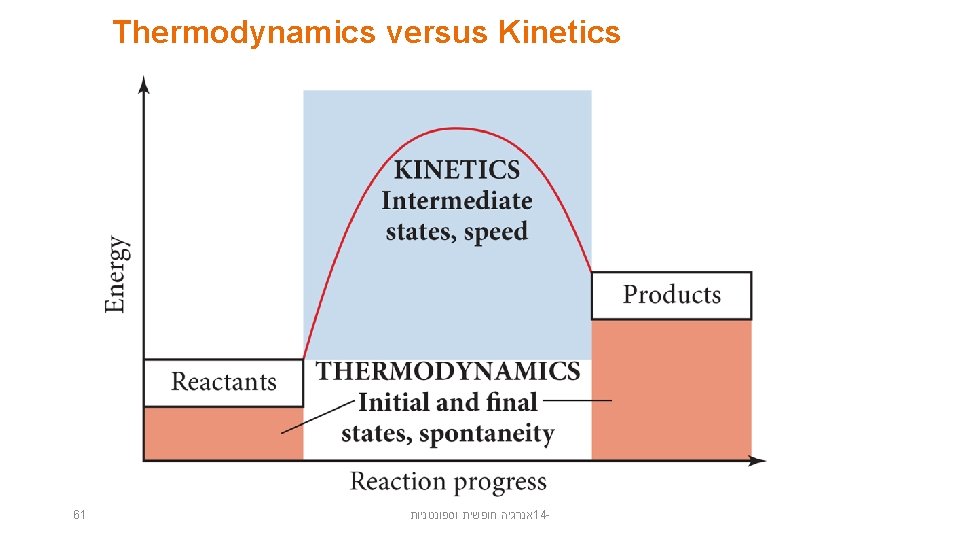
Thermodynamics versus Kinetics 61 אנרגיה חופשית וספונטניות 14 -

Thermodynamic & Kinetic Stability Thermodynamically stable: process is reactant-favored. • Thermodynamically stable: Pd + O 2 → Pd. O 2 Δr. G° = +326 k. J/mol • Thermodynamically unstable: 4 Al + 3 O 2 → 2 Al 2 O 3 Δr. G° = -3164. 6 k. J/mol 62 Kinetically stable: product-favored, but too slow to be important. • Diamond is thermodynamically unstable Cdiamond → Cgraphite Δr. G° = -2. 9 k. J/mol • but it is kinetically stable • Ea is too large אנרגיה חופשית וספונטניות 14 -
- Slides: 62