The Chemical Context of Life All matter including


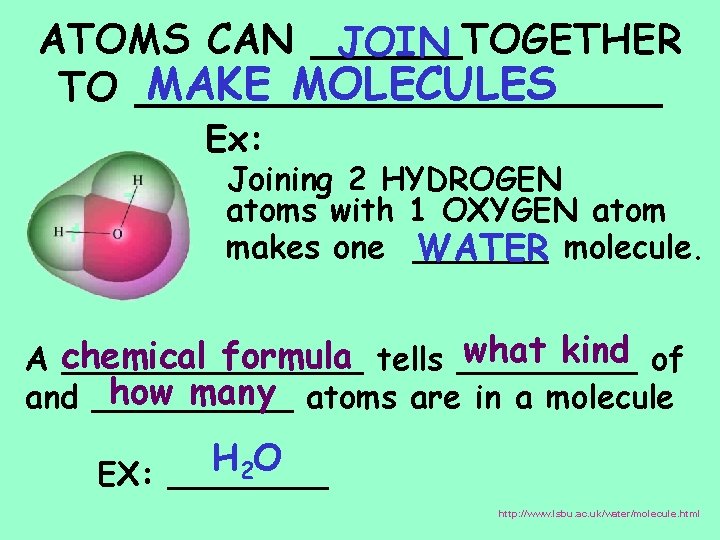

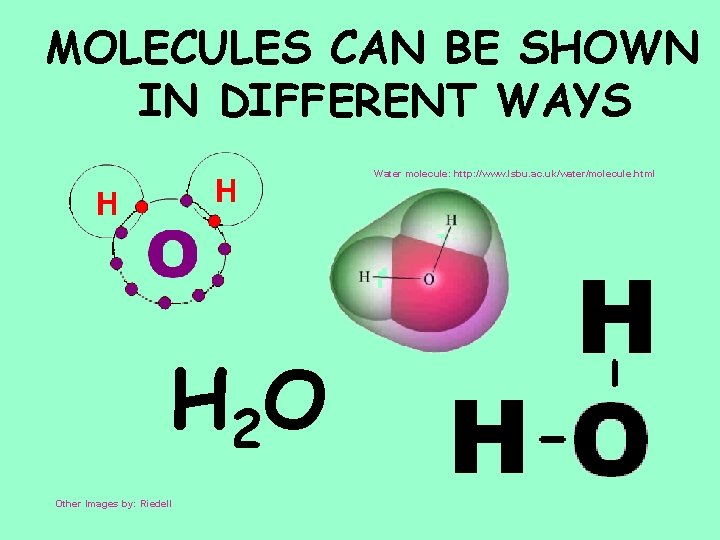












- Slides: 28

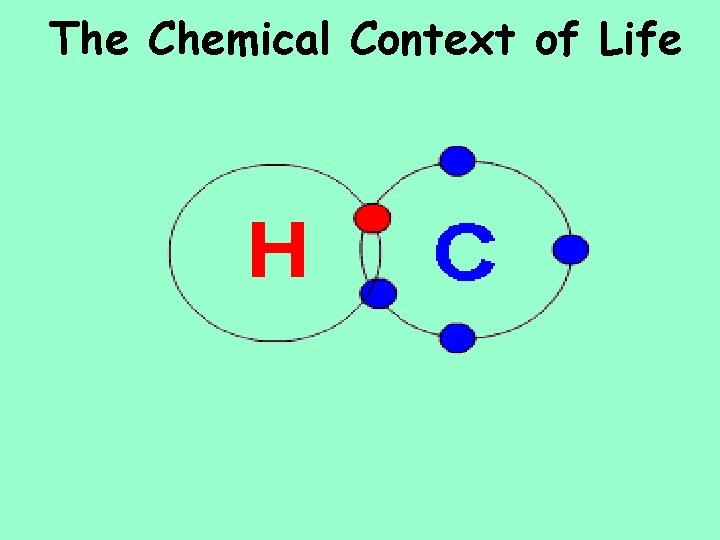
The Chemical Context of Life

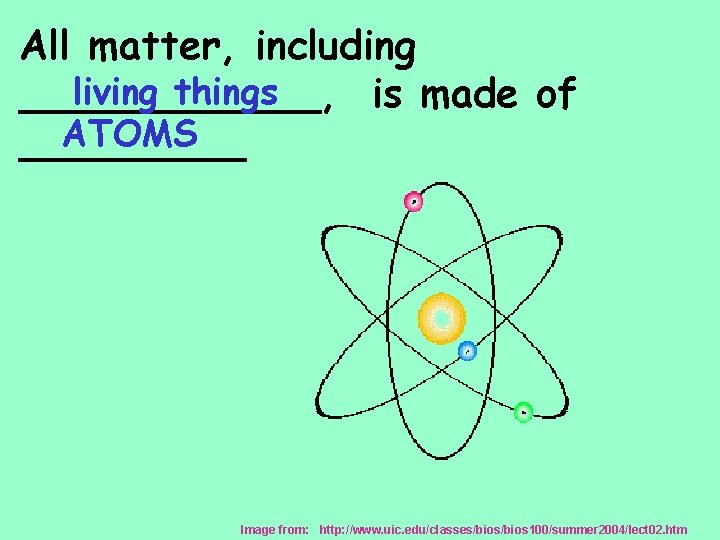
All matter, including living things ______, is made of ATOMS _____ Image from: http: //www. uic. edu/classes/bios 100/summer 2004/lect 02. htm

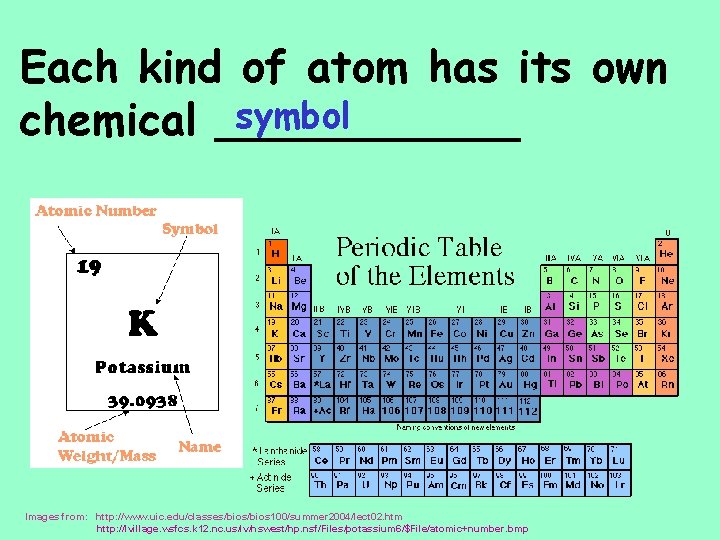
Each kind of atom has its own symbol chemical ______ Images from: http: //www. uic. edu/classes/bios 100/summer 2004/lect 02. htm http: //lvillage. wsfcs. k 12. nc. us/lv/hswest/hp. nsf/Files/potassium 6/$File/atomic+number. bmp

Atoms that have _________ gained or lost electrons have an _______ electric charge and are called IONS _____ They are written with a + or – next to their symbol + Na Cl

Atoms/ions important for living things Used to make bigger molecules 1. ______ Carbon - C 2. ______ Oxygen - O 3. ______ Hydrogen - H 4. ______ Nitrogen - N 5. ______ Phosphorus - P Ions = electrically charged atoms 1. ______ Sodium – Na+ 2. ______ Chloride – Cl+ Potassium – K 3. ______ + Hydrogen – H 4. ______

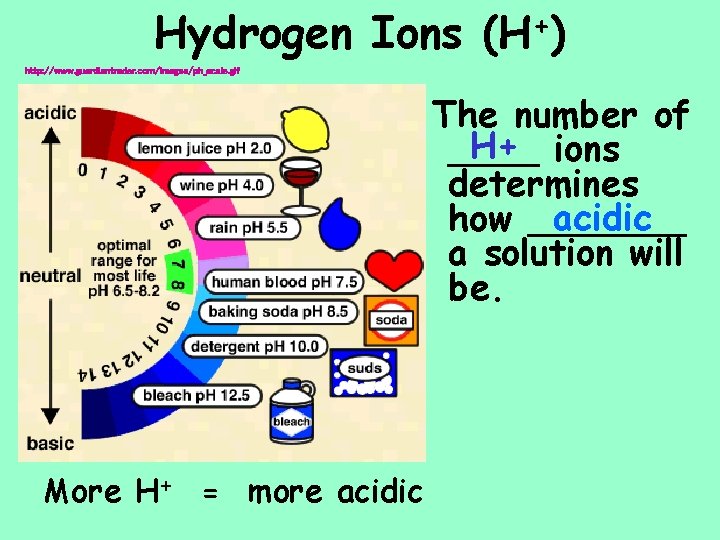
Hydrogen Ions + (H ) http: //www. guardiantrader. com/images/ph_scale. gif The number of H+ ions ____ determines acidic how _______ a solution will be. More H+ = more acidic
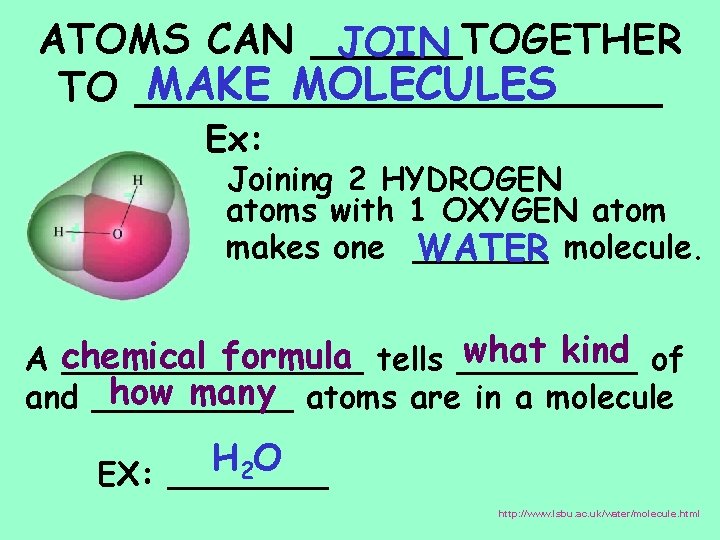
ATOMS CAN ______TOGETHER JOIN MAKE MOLECULES TO ___________ Ex: Joining 2 HYDROGEN atoms with 1 OXYGEN atom makes one ______ WATER molecule. what kind of formula tells _____ A chemical ________ how many atoms are in a molecule and _____ H O 2 EX: ____ http: //www. lsbu. ac. uk/water/molecule. html

VERY, VERY LARGE MOLECULES MACROMOLECULES = __________ EXAMPLE: Insulin = C 254 H 377 N 65 O 76 S 6 Image from: http: //www. biology. arizona. edu/biochemistry/tutorials/chemistry/page 2. html
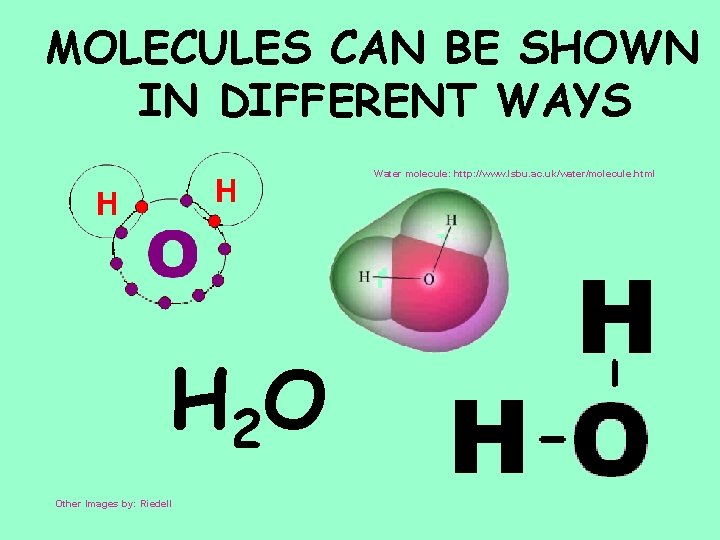
MOLECULES CAN BE SHOWN IN DIFFERENT WAYS Water molecule: http: //www. lsbu. ac. uk/water/molecule. html H 2 O Other Images by: Riedell

Polar Molecules Image from: http: //www. estrellamountain. edu/faculty/farabee/biobk/Bio. Book. CHEM 2. html Because of the location of electrons in molecules, some molecules have an UNEVEN pattern of _______________ electric charge + - More ___ on one side; More ___ on the other EX: water More about this in Chapter 7

EX: PHOSPHOLIPIDS The same molecule can have both POLAR and NON-POLAR parts More about this in Chapter 7 Polar head NON-polar tails Image by Riedell

HYDROGEN BONDS Bonds that form between the positively charged HYDROGEN atom _____________ in one molecule and a negatively charged _____ atom _____ in a nearby molecule are called HYDROGEN BONDS ___________ EX: water molecules are held together by Hydrogen bonds Images from: http: //www. estrellamountain. edu/faculty/farabee/biobk/Bio. Book. CHEM 2. html

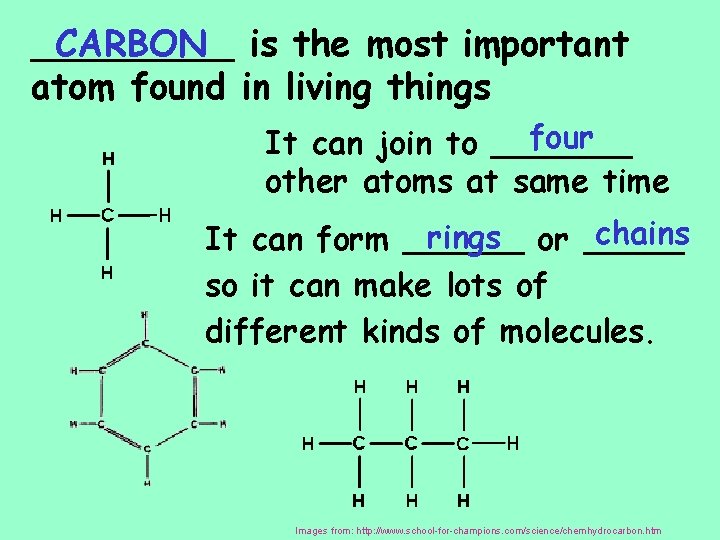
_____ CARBON is the most important atom found in living things four It can join to _______ other atoms at same time chains rings or _____ It can form ______ so it can make lots of different kinds of molecules. Images from: http: //www. school-for-champions. com/science/chemhydrocarbon. htm

“Like dissolves like” HYDROPHILIC __________ means “water loving” _____ groups/molecules try to POLAR _____ and touch water or be near _______ molecules other polar Water makes a great solvent in living things because so many molecules found in living things are polar or have a charge. http: //www. makash. ac. il/h_school/hstsb/chem/luach/dissolve. jpg

http: //egullet. com/imgs/egci/nonstocksauce/nons 1. JPG “Like dissolves like” HYDROPHOBIC _________ means “water fearing” NON-polar groups/molecules try to ______ be near other _____ NON-polar molecules polar away from and _____ molecules Oil based paints dissolve in solvents such as turpentine. . . not water.

http: //mchi. mcallenisd. org/mchi/site/hosting/ipc/ipcch 23 htm/ipcch 23 sec 2. htm “Like dissolves like” SOAP works because it has a ______ grease NON-polar end that dissolves _____________ and a _____ that dissolves in _____ Polar end water to wash away oily dirt.

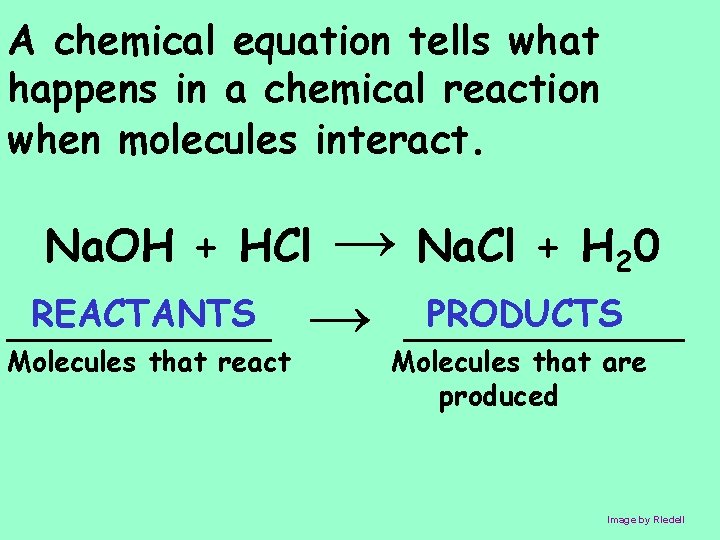
A chemical equation tells what happens in a chemical reaction when molecules interact. → Na. Cl + H 20 PRODUCTS → ________ Na. OH + HCl REACTANTS ________ Molecules that react Molecules that are produced Image by RIedell

REMEMBER: ALL the chemical reactions that happen in cells = _______ METABOLISM join Chemical reactions can _____ molecules together. break Chemical reactions can ______ molecules apart.

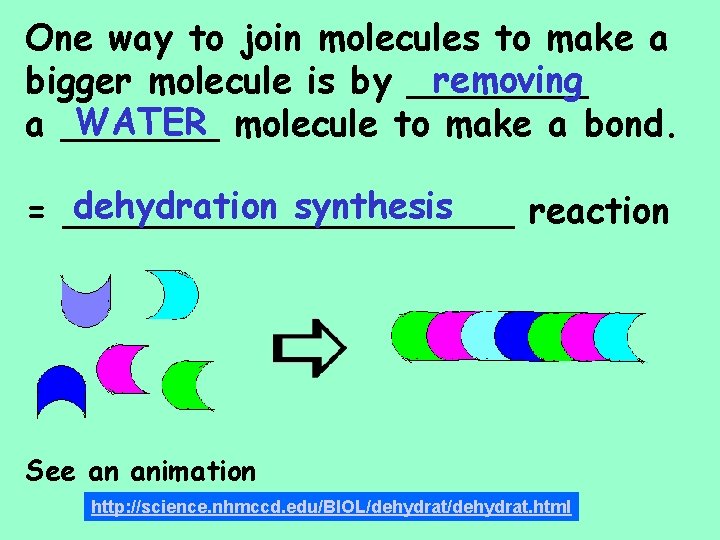
One way to join molecules to make a removing bigger molecule is by ____ WATER molecule to make a bond. a _______ dehydration synthesis = __________ reaction See an animation http: //science. nhmccd. edu/BIOL/dehydrat. html

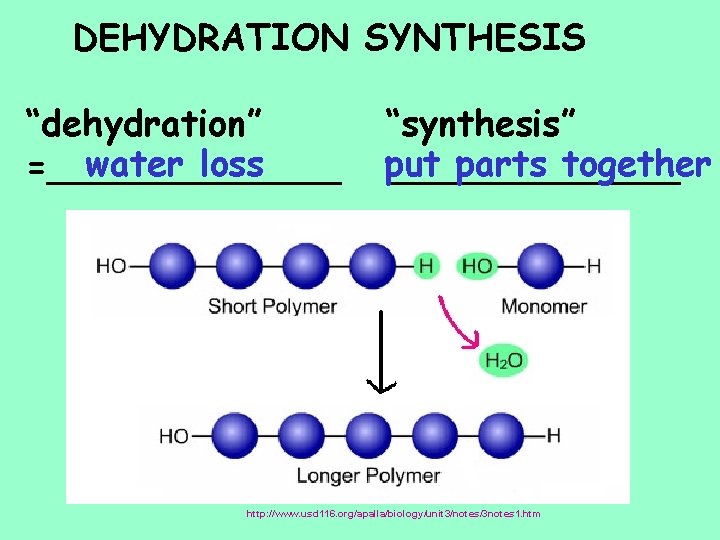
DEHYDRATION SYNTHESIS “dehydration” water loss =_______ “synthesis” put parts together _______ http: //io. uwinnipeg. ca/~simmons/macromol/sld 003. htm http: //www. usd 116. org/apalla/biology/unit 3/notes/3 notes 1. htm

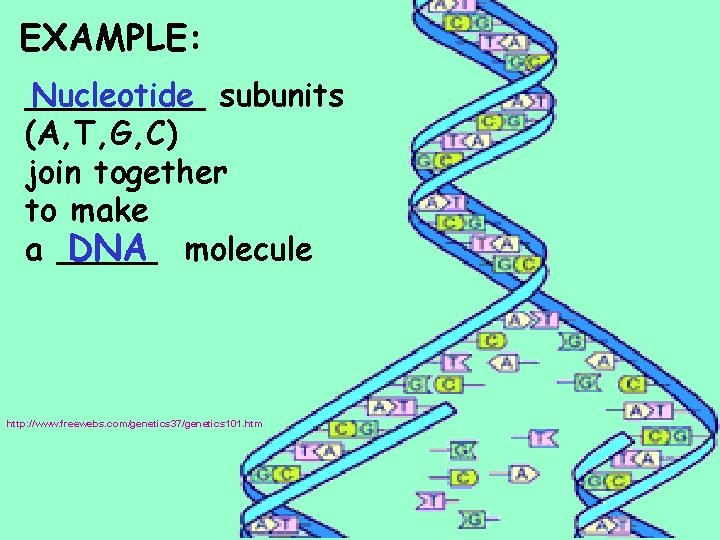
EXAMPLE: _____ Nucleotide subunits (A, T, G, C) join together to make a _____ DNA molecule http: //www. freewebs. com/genetics 37/genetics 101. htm

Chemical reactions can also break ____ molecules apart. HYDROLYSIS = kind of _______ chemical reaction in which a molecule is broken apart by adding WATER a____ molecule. hydro = water “_____” See an animation lysis = break apart “_____” http: //science. nhmccd. edu/BIOL/dehydrat. html

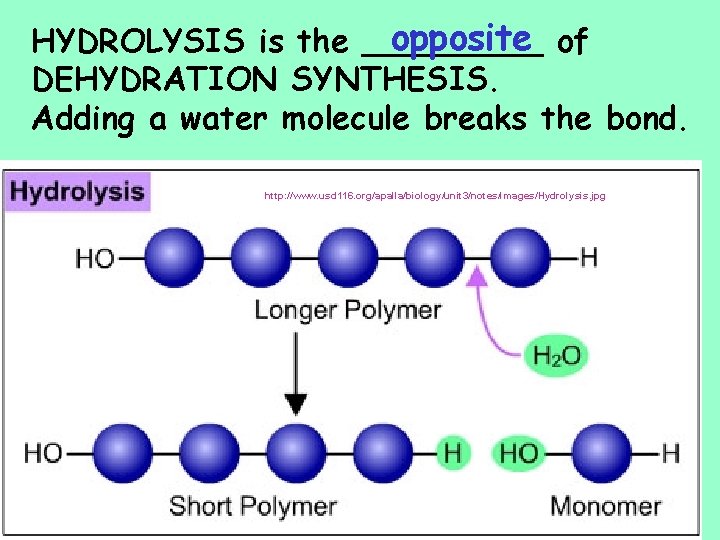
opposite of HYDROLYSIS is the _____ DEHYDRATION SYNTHESIS. Adding a water molecule breaks the bond. http: //www. usd 116. org/apalla/biology/unit 3/notes/images/Hydrolysis. jpg

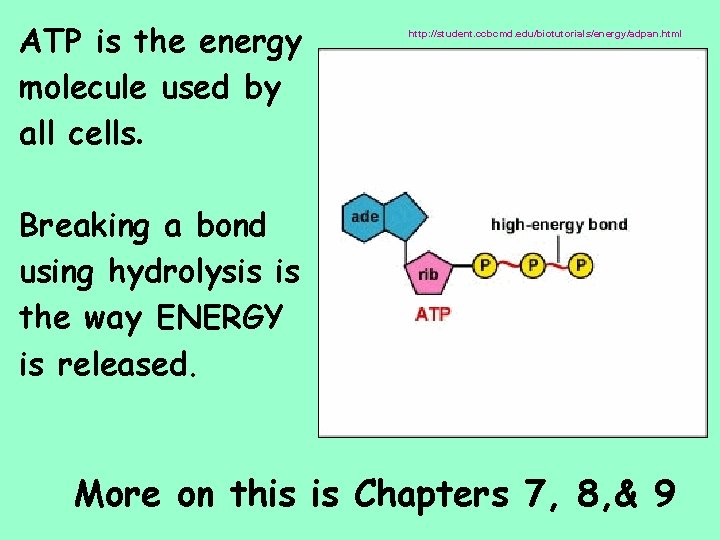
ATP is the energy molecule used by all cells. http: //student. ccbcmd. edu/biotutorials/energy/adpan. html Breaking a bond using hydrolysis is the way ENERGY is released. More on this is Chapters 7, 8, & 9

use BOTH of these Living things _______ reactions (and MORE) kinds of _______ to get the _______they need. materials http: //www. animationlibrary. com http: //www. cibike. org/Cartoon. Eating. gif

SOUTH DAKOTA CORE SCIENCE STANDARDS LIFE SCIENCE: Indicator 1: Understand the fundamental structures, functions, classifications, and mechanisms found in living things 9 -12. L. 1. 1. Students are able to relate cellular functions and processes to specialized structures within cells.

SOUTH DAKOTA ADVANCED STANDARDS LIFE SCIENCE Indicator 1: Understand the fundamental structures, functions, classifications, and mechanisms found in living things. 9 -12. L. 1. 1 A. Students are able to explain the physical and chemical processes of photosynthesis and cell respiration and their importance to plant and animal life. (INTRO TO BE ABLE TO DO THIS LATER) 9 -12. L. 1. 2 A. Describe how living systems use biofeedback mechanisms to maintain homeostasis. (SYNTHESIS) 9 -12. L. 1. 4 A. Identify factors that change the rates of enzyme catalyzed reactions. (APPLICATION)

Core High School Life Science Performance Descriptors High school students performing at the ADVANCED level: INTRODUCTION TO BE ABLE TO DO THE FOLLOWING LATER: explain the steps of photophosphorylation and the Calvin Cycle; analyze chemical reaction and chemical processes involved in the Calvin Cycle and Krebs Cycle; predict the function of a given structure; explain how protein production is regulated; High school students performing at the PROFICIENT level: describe and give examples of chemical reactions required to sustain life (hydrolysis, dehydration synthesis, photosynthesis, cellular respiration, ADP/ATP, role of enzymes); INTRODUCTION TO BE ABLE TO DO THE FOLLOWING LATER describe the relationship between structure and function (cells, tissues, organ systems, and organisms); tell how DNA determines protein formation; predict how life systems respond to changes in the environment; High school students performing at the BASIC level name chemical reactions required to sustain life (hydrolysis, dehydration synthesis, photosynthesis, cellular respiration, ADP/ATP, role of enzymes); INTRODUCTION TO BE ABLE TO DO THE FOLLOWING LATER recognize that different structures perform different functions; identify DNA as the structure that carries the genetic code;