The Alkenes 1 7 3 a state the





- Slides: 19

The Alkenes (1. 7. 3) a. state the general formula of alkenes and understand that they are unsaturated hydrocarbons with a carbon-carbon double bond which consists of a and a bond b. explain E-Z isomerism (geometric/cis-trans isomerism) in terms of restricted rotation around a C=C double bond and the nature of the substituents on the carbon atoms c. demonstrate an understanding the E-Z naming system and why it is necessary to use this when the cis- and trans- naming system breaks down Independent Learning (HW) – pp 124 -128 and ALL questions therein Connector: Write the structural formulae and displayed formulae for ethene, propene and the three isomers of butene Crowe 2008

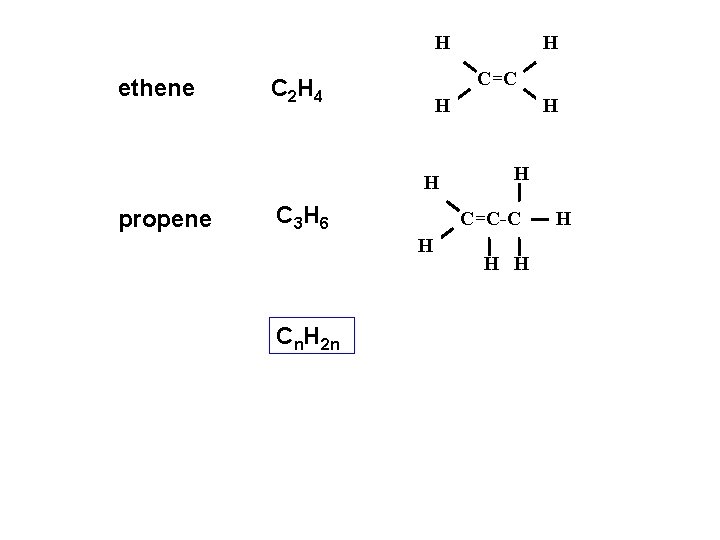
H ethene C=C C 2 H 4 H H propene C 3 H 6 H H C=C-C H Cn. H 2 n H H

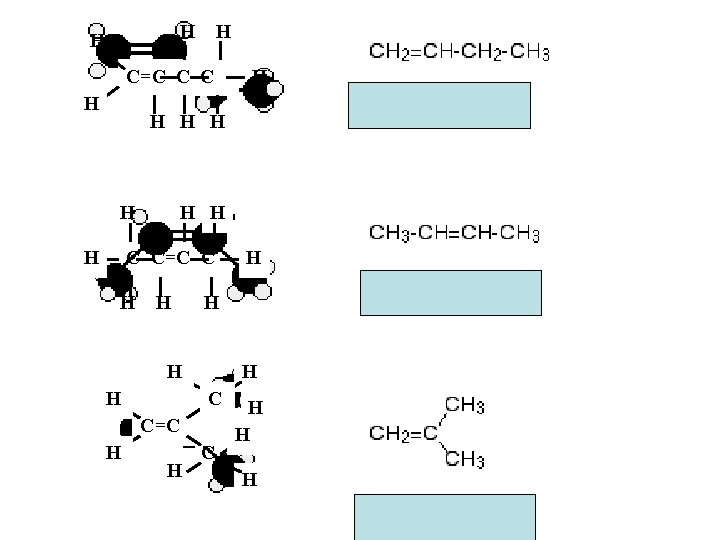
H H H C=C C C H H H H H C C=C C H H H C C=C H H H

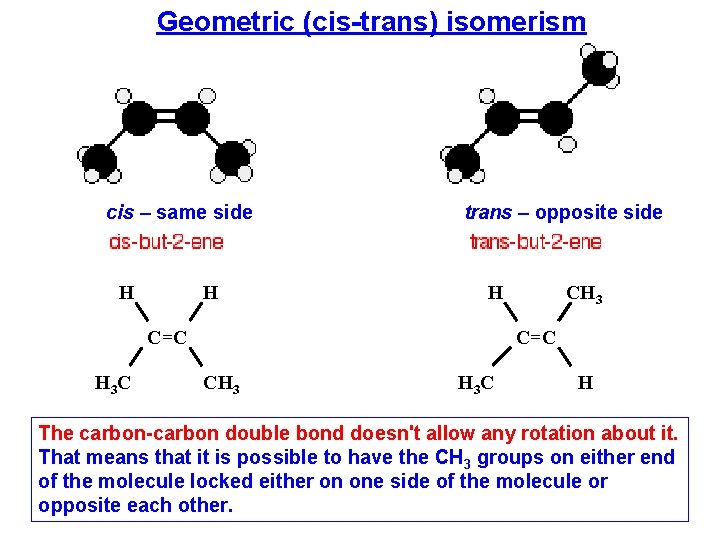
Geometric (cis-trans) isomerism cis – same side H H trans – opposite side H C=C H 3 C CH 3 C=C CH 3 C H The carbon-carbon double bond doesn't allow any rotation about it. That means that it is possible to have the CH 3 groups on either end of the molecule locked either on one side of the molecule or opposite each other.

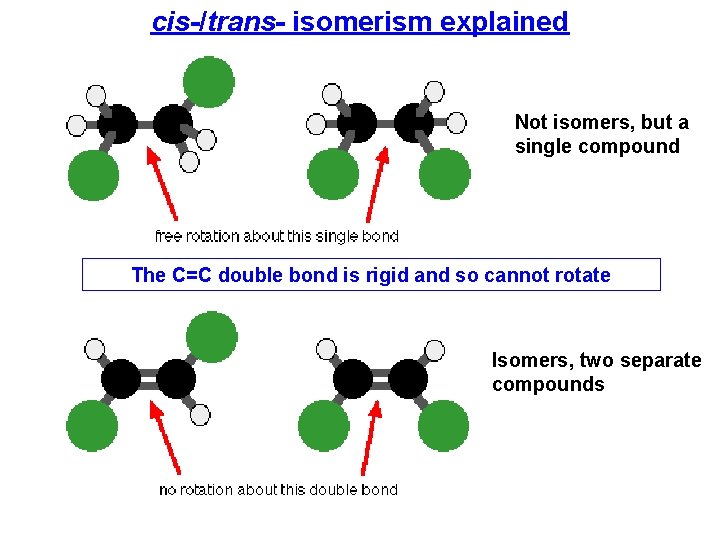
cis-/trans- isomerism explained Not isomers, but a single compound The C=C double bond is rigid and so cannot rotate Isomers, two separate compounds

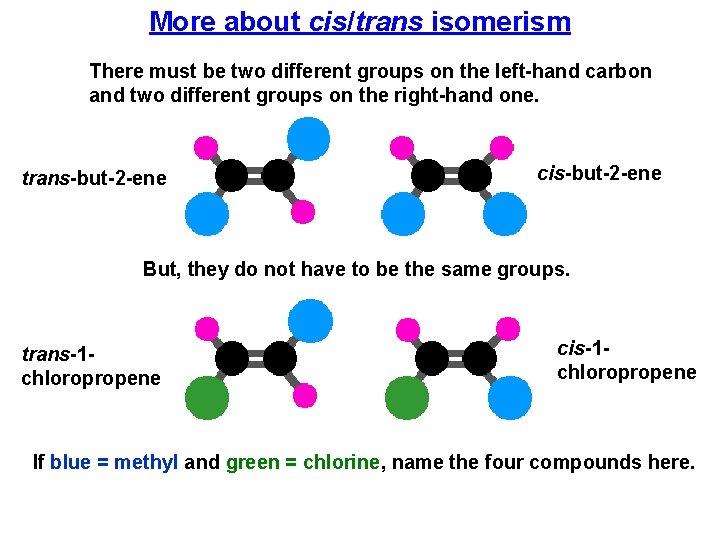
More about cis/trans isomerism There must be two different groups on the left-hand carbon and two different groups on the right-hand one. trans-but-2 -ene cis-but-2 -ene But, they do not have to be the same groups. trans-1 chloropropene cis-1 chloropropene If blue = methyl and green = chlorine, name the four compounds here.

Does 2 -chloropropene exhibit cis-/trans- isomerism? No because there needs to be two different groups on each carbon!

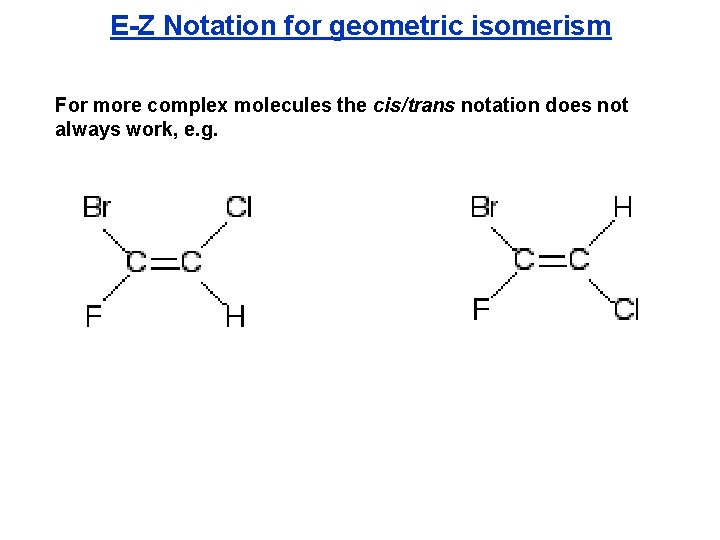
E-Z Notation for geometric isomerism For more complex molecules the cis/trans notation does not always work, e. g.

How the E-Z system works • The carbon atoms at each end of the double bond are considered separately • The atom which has the higher atomic number is given the higher priority • If the two groups with the higher priorities are on the same side of the double bond, that is described as the (Z)- isomer. (The Z comes from a German word zusammen which means together. ) (Zame Zide) • If the two groups with the higher priorities are on opposite sides of the double bond, then this is the (E)- isomer. (The E comes from the German entgegen which means opposite. )

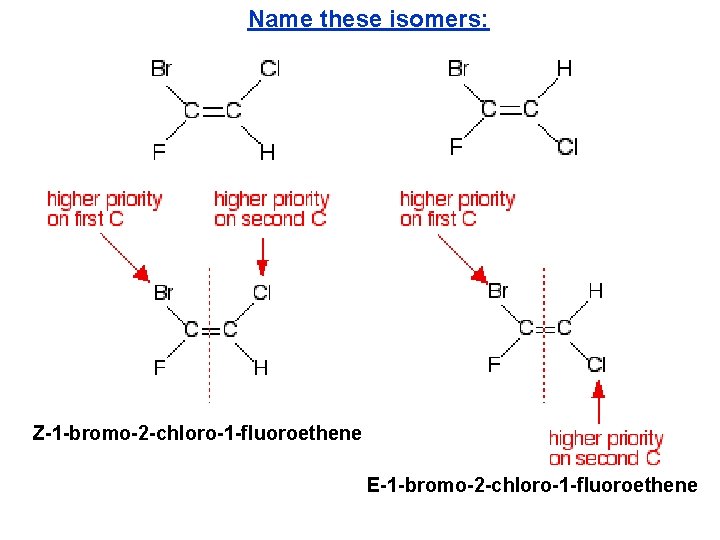
Name these isomers: Z-1 -bromo-2 -chloro-1 -fluoroethene E-1 -bromo-2 -chloro-1 -fluoroethene

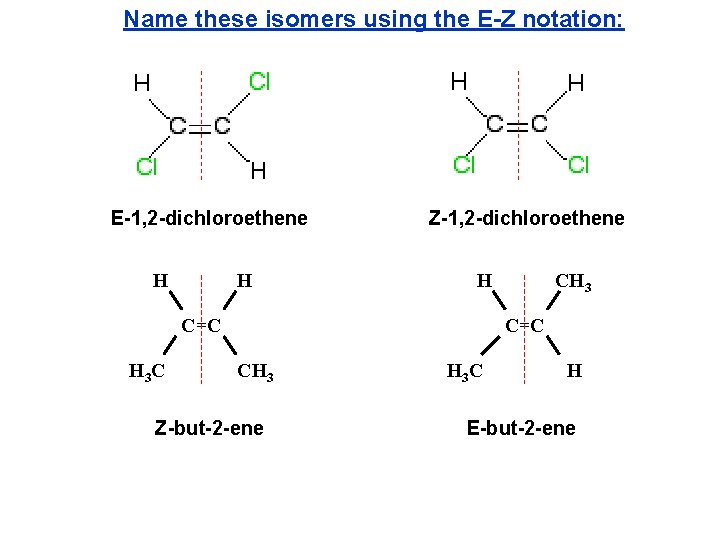
Name these isomers using the E-Z notation: E-1, 2 -dichloroethene H H Z-1, 2 -dichloroethene H C=C H 3 C CH 3 C=C CH 3 Z-but-2 -ene H 3 C H E-but-2 -ene

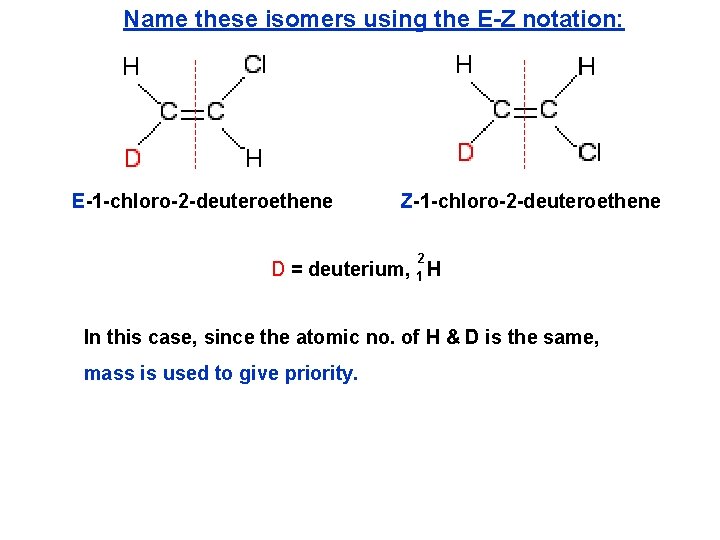
Name these isomers using the E-Z notation: E-1 -chloro-2 -deuteroethene Z-1 -chloro-2 -deuteroethene D = deuterium, 2 1 H In this case, since the atomic no. of H & D is the same, mass is used to give priority.

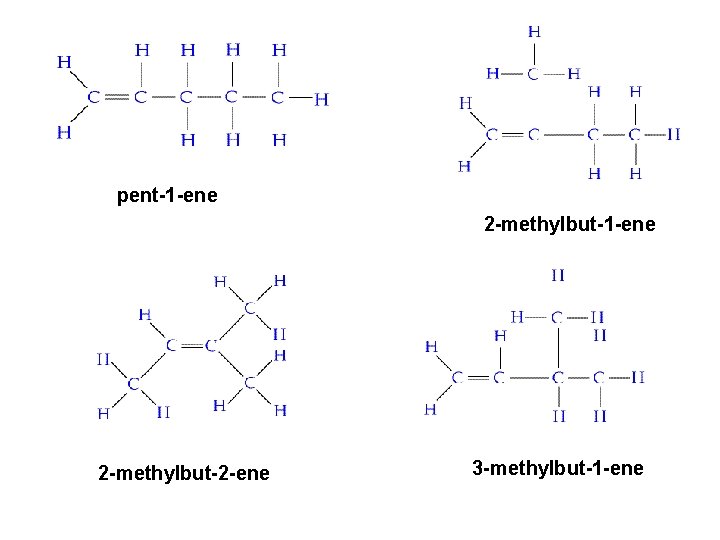
Homework Draw the isomers of pentene (6) and name them. Use the E-Z notation for any geometric isomers.

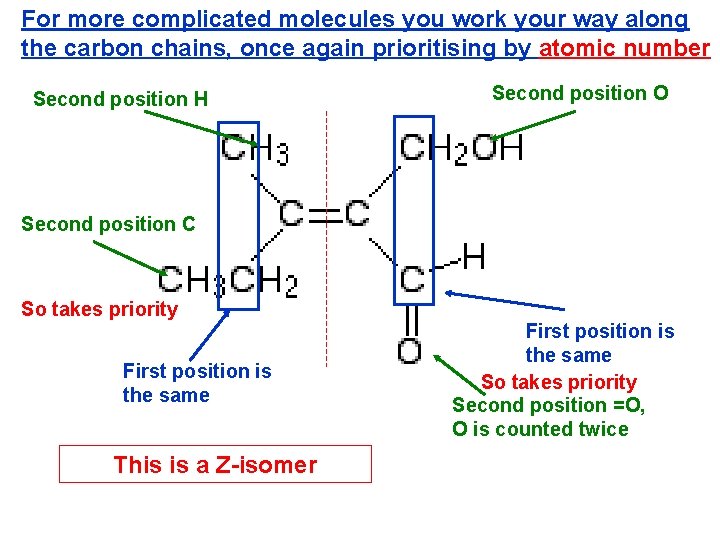
For more complicated molecules you work your way along the carbon chains, once again prioritising by atomic number Second position H Second position O Second position C So takes priority First position is the same This is a Z-isomer First position is the same So takes priority Second position =O, O is counted twice

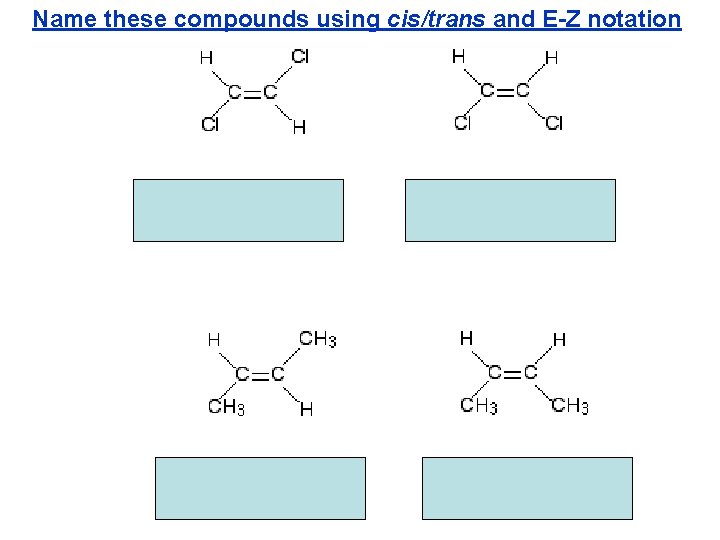
Name these compounds using cis/trans and E-Z notation

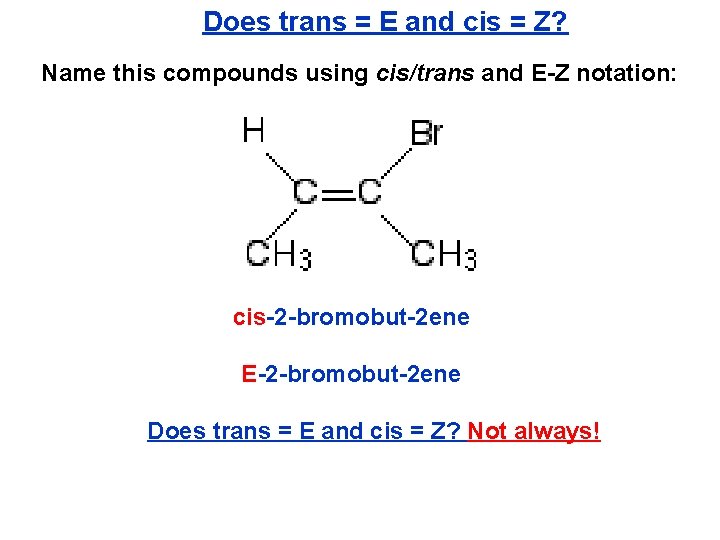
Does trans = E and cis = Z? Name this compounds using cis/trans and E-Z notation: cis-2 -bromobut-2 ene E-2 -bromobut-2 ene Does trans = E and cis = Z? Not always!


pent-1 -ene 2 -methylbut-2 -ene 3 -methylbut-1 -ene

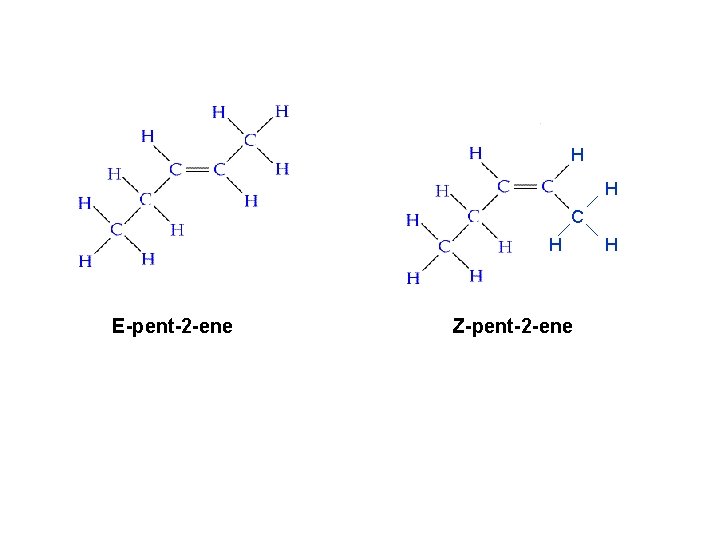
H H C H E-pent-2 -ene Z-pent-2 -ene H