AS Chemistry Alkenes Alkenes Lesson objective To know

AS Chemistry Alkenes

Alkenes Lesson objective: To know about alkenes Success criteria: • understand the term ‘stereoisomerism’, as illustrated by E/Z isomerism (including cis-trans isomerism where two of the substituent groups are the same) • know the general formula for alkenes • know that alkenes and cycloalkenes are unsaturated hydrocarbons • understand the bonding in alkenes in terms of σand π- bonds

What are alkenes? The alkenes are a homologous series of hydrocarbons with the general formula Cn. H 2 n and names ending –ene. Alkenes contain a carbon – carbon double bond and so are unsaturated. 3 of 34 No. of carbon atoms Molecular formula 2 C 2 H 4 ethene 3 C 3 H 6 propene 4 C 4 H 8 butene 5 C 5 H 10 pentene 6 C 6 H 12 hexene Name © Boardworks Ltd 2009

Alkenes and the Double Bond C=C • Anything with a double bond in it is classed as unsaturated. • This includes alkenes and cycloalkenes. • Some very important molecules are unsaturated:
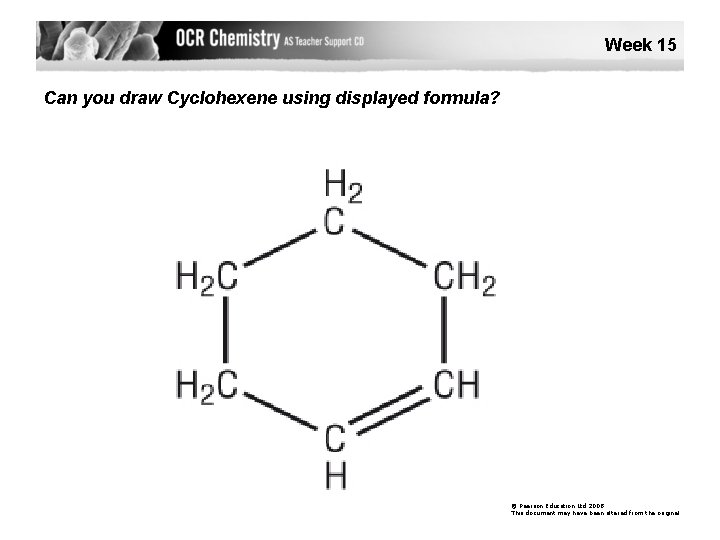
Week 15 Can you draw Cyclohexene using displayed formula? © Pearson Education Ltd 2008 This document may have been altered from the original
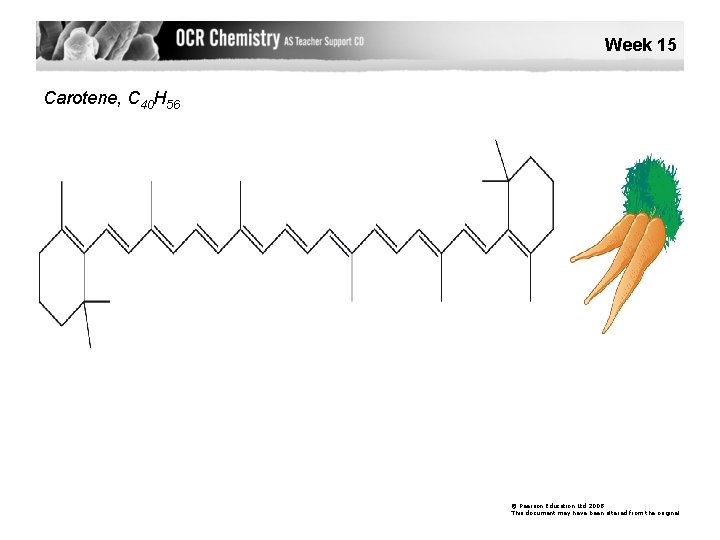
Week 15 Carotene, C 40 H 56 © Pearson Education Ltd 2008 This document may have been altered from the original
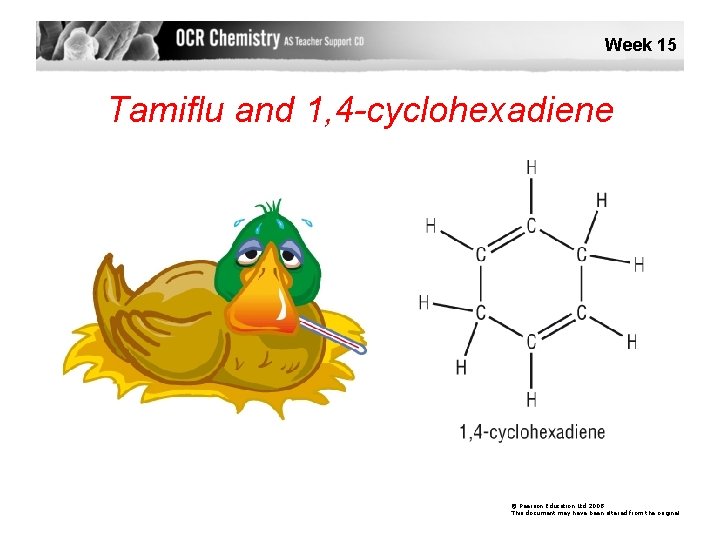
Week 15 Tamiflu and 1, 4 -cyclohexadiene © Pearson Education Ltd 2008 This document may have been altered from the original

Alkane: Bromine water remains orange Alkene: Bromine water turns from orange to colourless, forming a dibromo compound

Alkenes Questions Two hydrocarbons (A and B) have the molecular formula C 5 H 10. Hydrocarbon A decolourises bromine water but hydrocarbon B does not. Explain this observation, and suggest a structure and name for hydrocarbon B. Hydrocarbon A must be unsaturated but hydrocarbon B must be saturated. It could be cyclopentane.

Alkenes Questions Octene is an alkene whose molecules each contain 8 carbon atoms. Write the molecular and empirical formulae for octene.

Alkenes Lesson objective: To know about alkenes Success criteria: • understand the term ‘stereoisomerism’, as illustrated by E/Z isomerism (including cis-trans isomerism where two of the substituent groups are the same) • know the general formula for alkenes • know that alkenes and cycloalkenes are unsaturated hydrocarbons • understand the bonding in alkenes in terms of σand π- bonds

Alkenes and the Double Bond • For a bond to happen, electron orbitals must overlap. • Write out the electronic structure of carbon: 1 s 2 2 p 2 • In full? 1 s 2 2 px 1 2 py 1 Now draw all the orbitals in the second shell
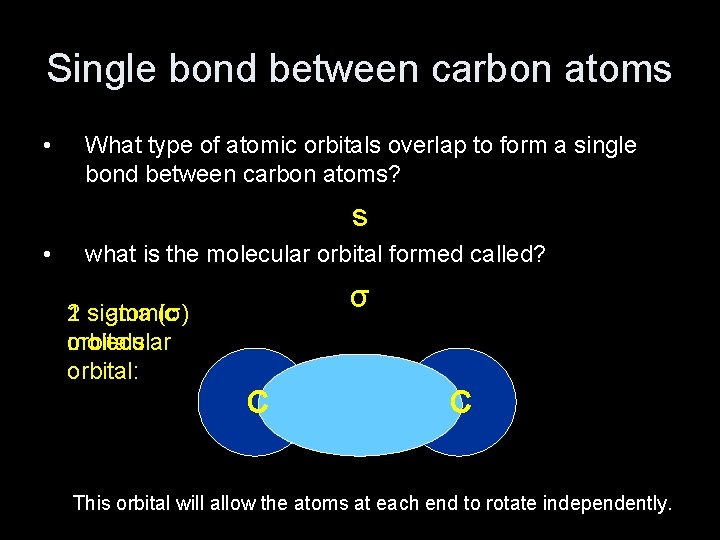
Single bond between carbon atoms • What type of atomic orbitals overlap to form a single bond between carbon atoms? s • what is the molecular orbital formed called? σ 2 sigma s atomic 1 (σ) orbitals: molecular orbital: C C This orbital will allow the atoms at each end to rotate independently.
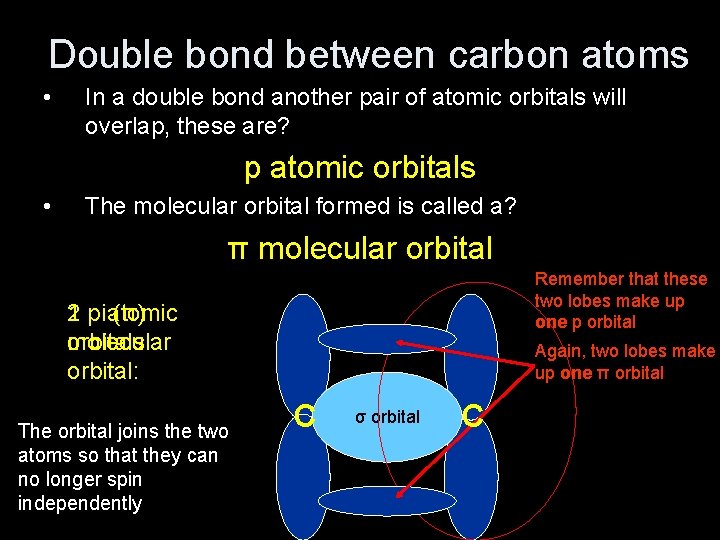
Double bond between carbon atoms • In a double bond another pair of atomic orbitals will overlap, these are? p atomic orbitals • The molecular orbital formed is called a? π molecular orbital Remember that these two lobes make up one p orbital 1 pi 2 p atomic (π) orbitals: molecular orbital: The orbital joins the two atoms so that they can no longer spin independently Again, two lobes make up one π orbital C σ orbital C
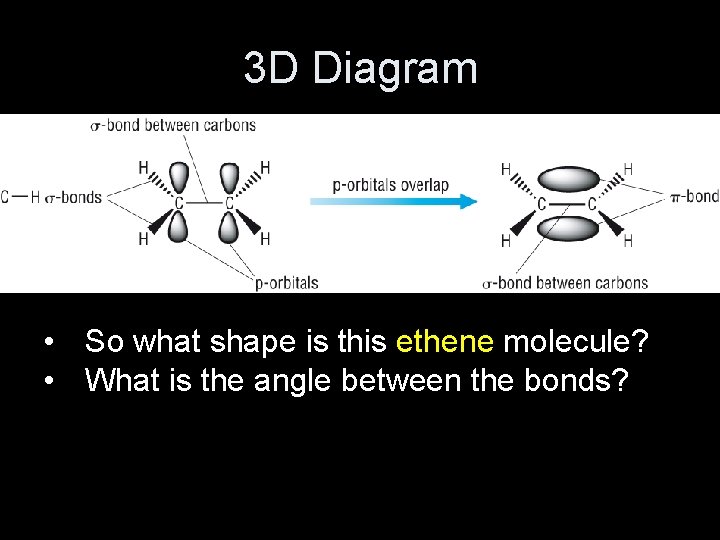
3 D Diagram • So what shape is this ethene molecule? • What is the angle between the bonds?
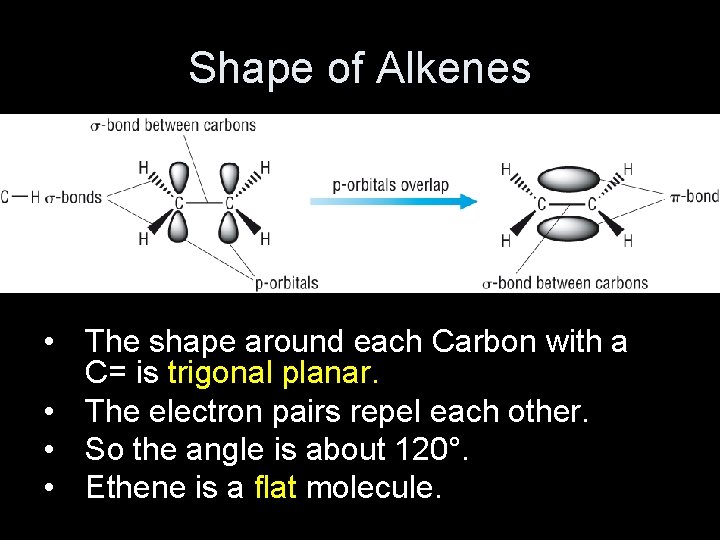
Shape of Alkenes • The shape around each Carbon with a C= is trigonal planar. • The electron pairs repel each other. • So the angle is about 120°. • Ethene is a flat molecule.

Alkenes Lesson objective: To know about alkenes Success criteria: • understand the term ‘stereoisomerism’, as illustrated by E/Z isomerism (including cis-trans isomerism where two of the substituent groups are the same) • know the general formula for alkenes • know that alkenes and cycloalkenes are unsaturated hydrocarbons • understand the bonding in alkenes in terms of σand π- bonds
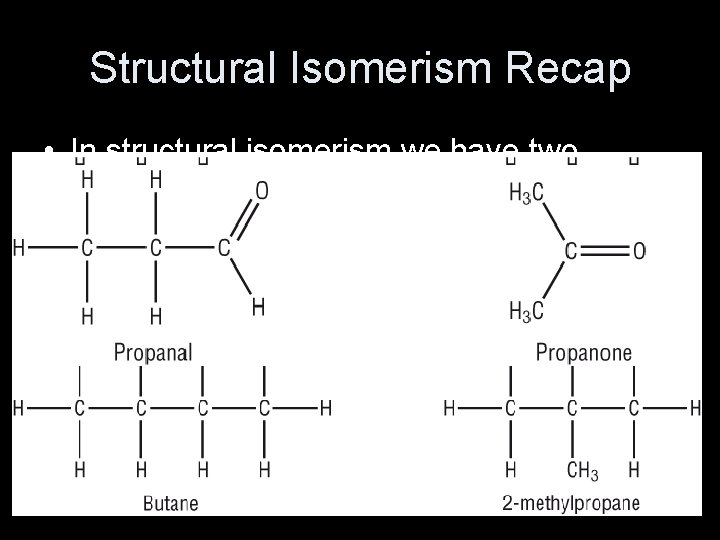
Structural Isomerism Recap • In structural isomerism we have two molecules with the same atoms joined up in different orders. • This can be by: – Branching – Moving a functional group – Having a different functional group – Or by a combination of the above

Alkenes • • The most important feature of alkenes is the double bond. Use the molecular models to make: 1. Ethane 2. Ethene • What do you notice about the bonds between the two carbons in each molecule?
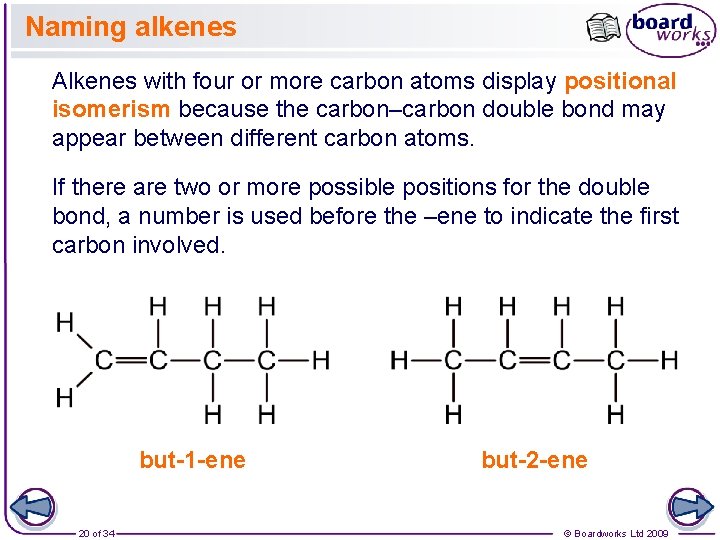
Naming alkenes Alkenes with four or more carbon atoms display positional isomerism because the carbon–carbon double bond may appear between different carbon atoms. If there are two or more possible positions for the double bond, a number is used before the –ene to indicate the first carbon involved. but-1 -ene 20 of 34 but-2 -ene © Boardworks Ltd 2009

Alkenes Questions Buta – 1, 3 – diene is used in the manufacture of synthetic rubber. Suggest its displayed and skeletal formulae.
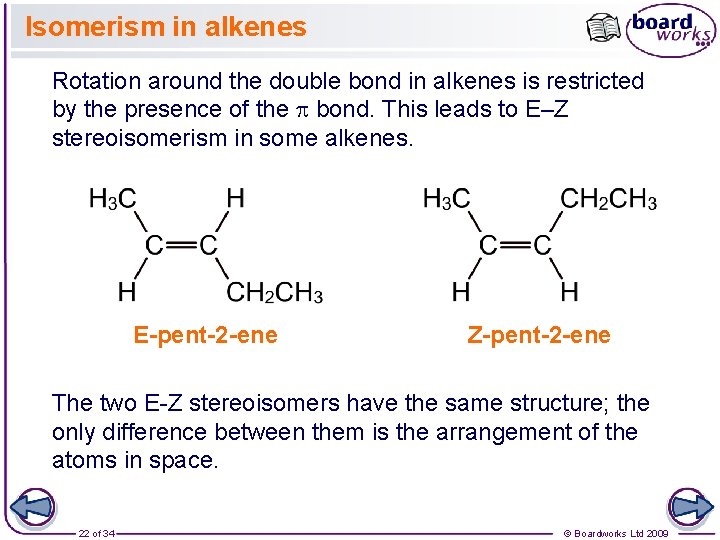
Isomerism in alkenes Rotation around the double bond in alkenes is restricted by the presence of the bond. This leads to E–Z stereoisomerism in some alkenes. E-pent-2 -ene Z-pent-2 -ene The two E-Z stereoisomers have the same structure; the only difference between them is the arrangement of the atoms in space. 22 of 34 © Boardworks Ltd 2009

Alkenes and the Double Bond C=C • Just like in the molecules you made, a single C-C bond will rotate, a C=C bond will not rotate. • Why might this be?

Cis and Trans isomers • Take your ethene model. • Swap one of the hydrogens on each carbon atom for a different atoms. • Does everyone have the same thing? • Because there is no rotation about the double bond there are two possibilities.
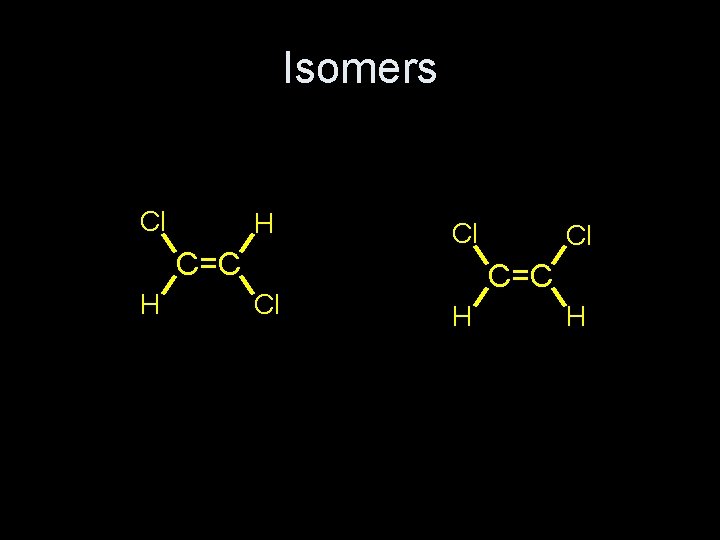
Isomers Cl H C=C H Cl Cl Cl C=C H H

Cis and Trans isomers • These two configurations are stereoisomers. • This is called E/Z stereoisomerism. • In the special case where each Carbon has one hydrogen and one “nonhydrogen” the isomers are called cis and trans.
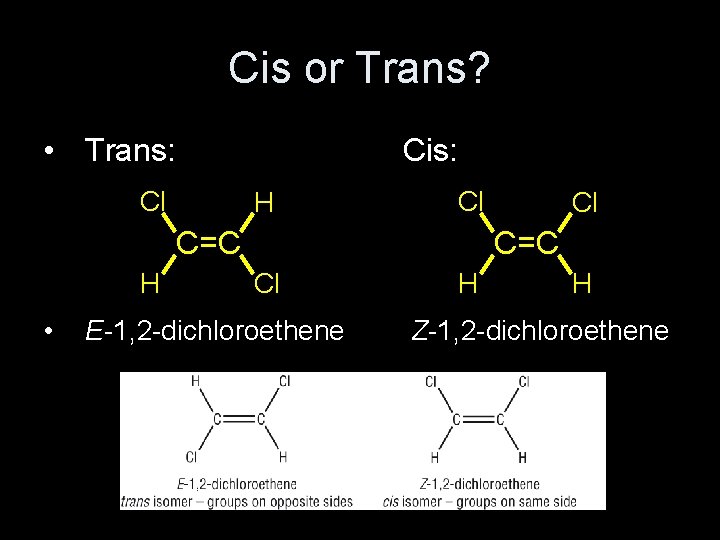
Cis or Trans? • Trans: Cis: Cl H Cl C=C H • Cl C=C Cl E-1, 2 -dichloroethene H H Z-1, 2 -dichloroethene
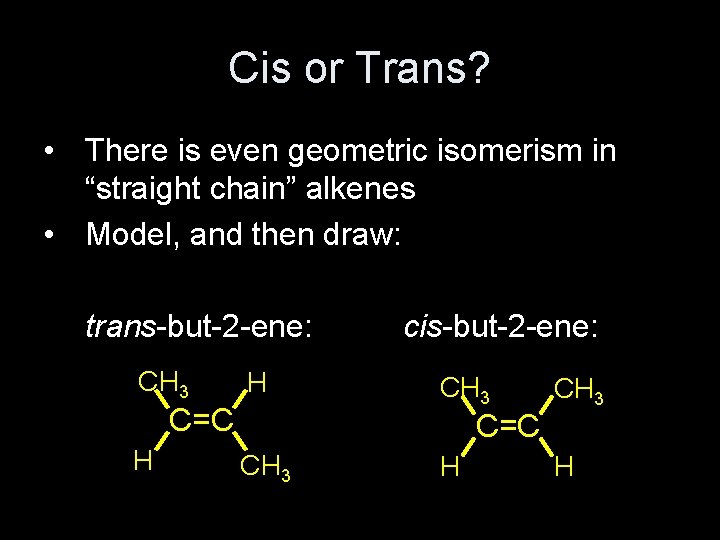
Cis or Trans? • There is even geometric isomerism in “straight chain” alkenes • Model, and then draw: trans-but-2 -ene: CH 3 H C=C H cis-but-2 -ene: CH 3 C=C CH 3 H
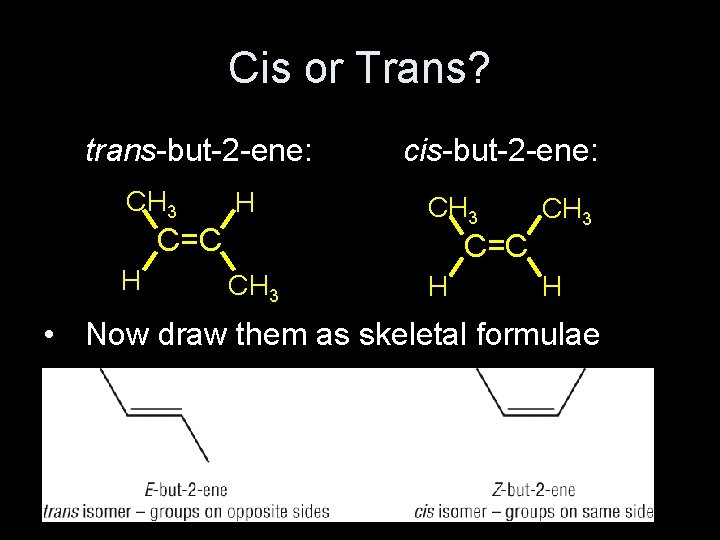
Cis or Trans? trans-but-2 -ene: CH 3 H C=C H cis-but-2 -ene: CH 3 C=C CH 3 H • Now draw them as skeletal formulae

E or Z? • The E/Z naming is designed to work whatever the groups are attached to the C=C carbons. • On each carbon on the C=C the group with the highest atomic number is given high priority (1), the other is given low (2). • If high priority groups are on the same side it is together (Z), if they are opposite it is apart (E)

For German speakers • Zusamemen = together • Entgegen = opposite
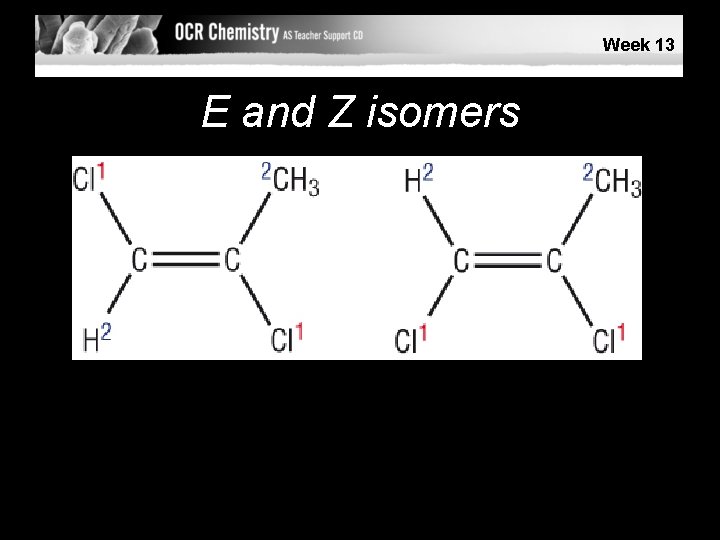
Week 13 E and Z isomers © Pearson Education Ltd 2008 This document may have been altered from the original

Alkenes Lesson objective: To know about alkenes Success criteria: • understand the term ‘stereoisomerism’, as illustrated by E/Z isomerism (including cis-trans isomerism where two of the substituent groups are the same) • know the general formula for alkenes • know that alkenes and cycloalkenes are unsaturated hydrocarbons • understand the bonding in alkenes in terms of σand π- bonds

Isomers of alkenes questions Name this alkene:

Isomers of alkenes questions Pent – 2 – ene has two stereoisomers. Name ach one and draw its displayed formula. .

Alkenes Lesson objective: To know about alkenes Success criteria: • understand the term ‘stereoisomerism’, as illustrated by E/Z isomerism (including cis-trans isomerism where two of the substituent groups are the same) • know the general formula for alkenes • know that alkenes and cycloalkenes are unsaturated hydrocarbons • understand the bonding in alkenes in terms of σand π- bonds
- Slides: 36