7 Alkenes Reactions and Synthesis Based on Mc

7. Alkenes: Reactions and Synthesis Based on Mc. Murry’s Organic Chemistry, 6 th edition © 2003 Ronald Kluger Department of Chemistry University of Toronto Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003

Diverse Reactions of Alkenes n Alkenes react with many electrophiles to give useful products by addition (often through special reagents) n alcohols (add H-OH) n alkanes (add H-H) n halohydrins (add HO-X) n dihalides (add X-X) n halides (add H-X) n diols (add HO-OH) n cyclopropane (add : CH 2) Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 2

7. 1 Preparation of Alkenes: A Preview of Elimination Reactions n Alkenes are commonly made by n n elimination of HX from alkyl halide (dehydrohalogenation) n Uses heat and KOH elimination of H-OH from an alcohol (dehydration) n require strong acids (sulfuric acid, 50 ºC) Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 3
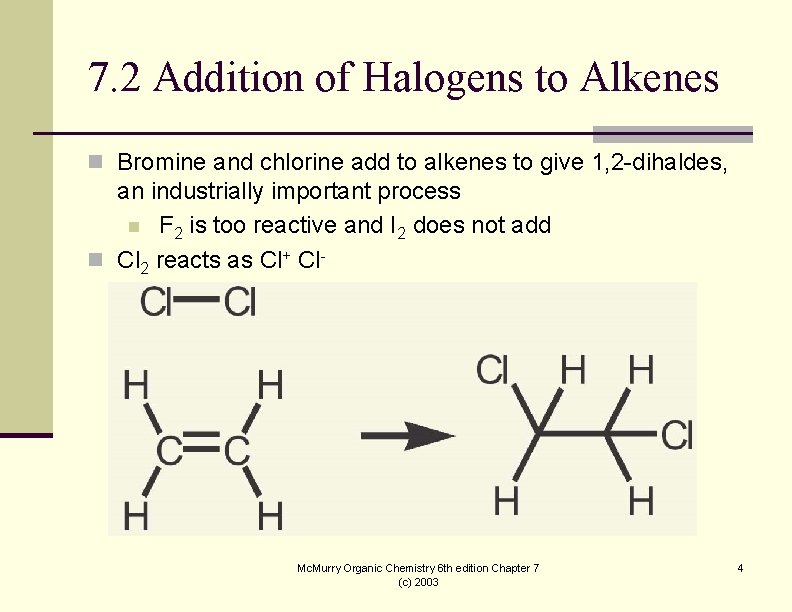
7. 2 Addition of Halogens to Alkenes n Bromine and chlorine add to alkenes to give 1, 2 -dihaldes, an industrially important process n F 2 is too reactive and I 2 does not add n Cl 2 reacts as Cl+ Cln Br 2 is similar Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 4

Addition of Br 2 to Cyclopentene n Addition is exclusively trans + Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 5
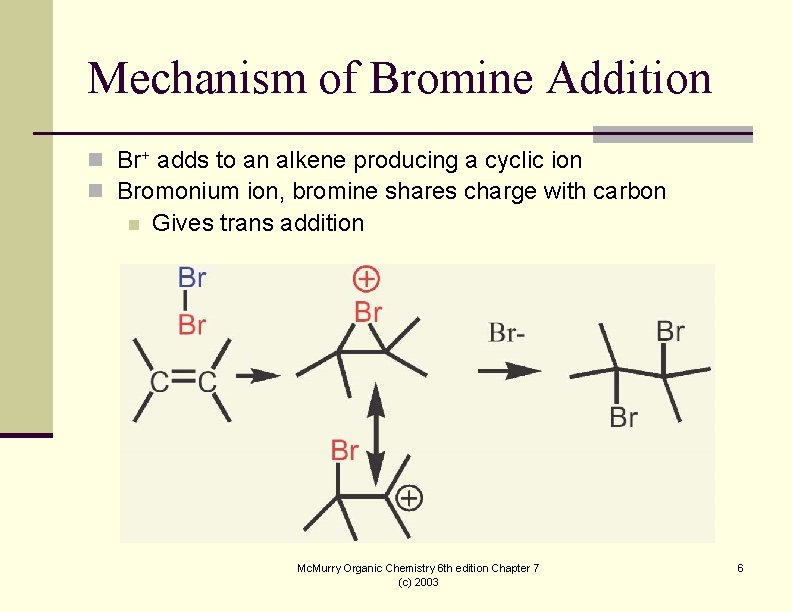
Mechanism of Bromine Addition n Br+ adds to an alkene producing a cyclic ion n Bromonium ion, bromine shares charge with carbon n Gives trans addition Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 6

Bromonium Ion Mechanism n Electrophilic addition of bromine to give a cation is followed by cyclization to give a bromonium ion n This bromoniun ion is a reactive electrophile and bromide ion is a good nucleophile Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 7
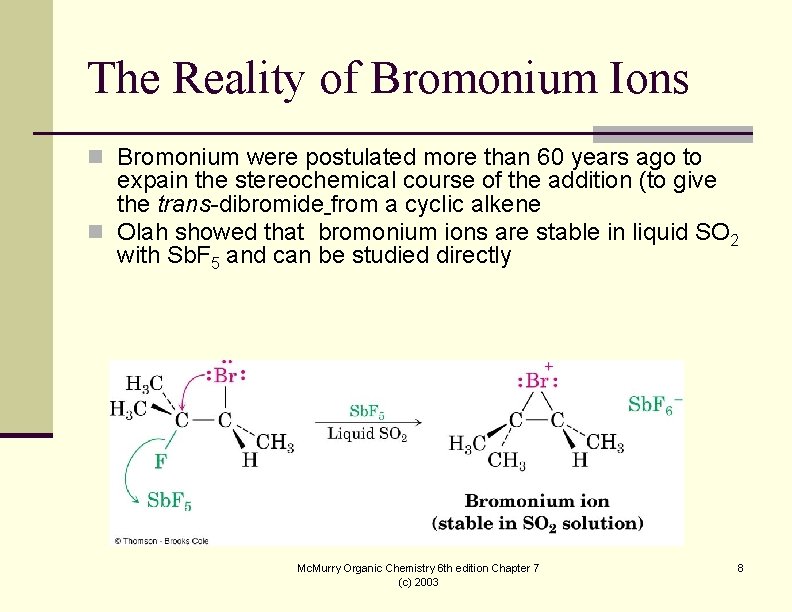
The Reality of Bromonium Ions n Bromonium were postulated more than 60 years ago to expain the stereochemical course of the addition (to give the trans-dibromide from a cyclic alkene n Olah showed that bromonium ions are stable in liquid SO 2 with Sb. F 5 and can be studied directly Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 8
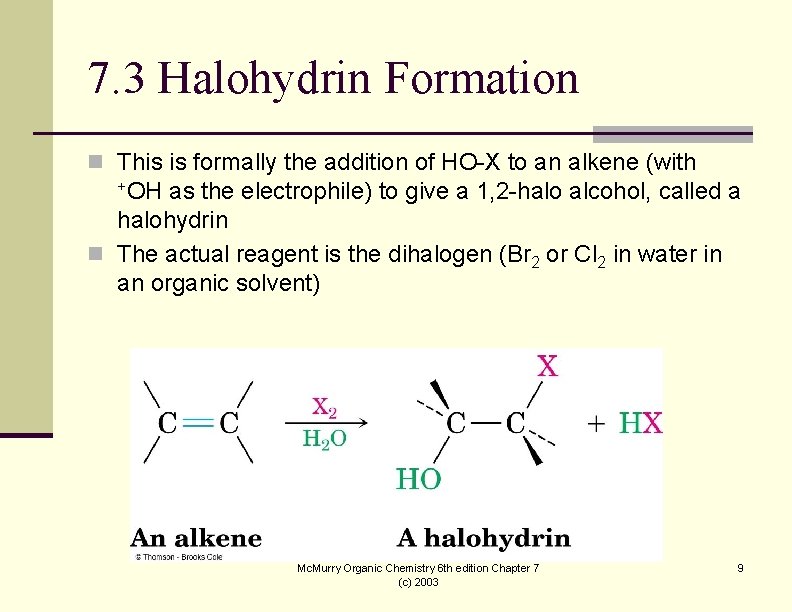
7. 3 Halohydrin Formation n This is formally the addition of HO-X to an alkene (with +OH as the electrophile) to give a 1, 2 -halo alcohol, called a halohydrin n The actual reagent is the dihalogen (Br 2 or Cl 2 in water in an organic solvent) Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 9
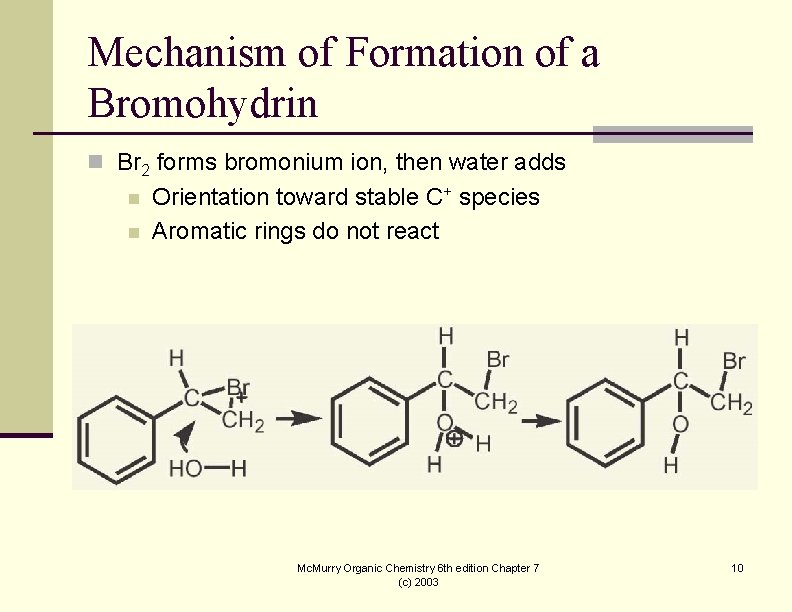
Mechanism of Formation of a Bromohydrin n Br 2 forms bromonium ion, then water adds n n Orientation toward stable C+ species Aromatic rings do not react Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 10
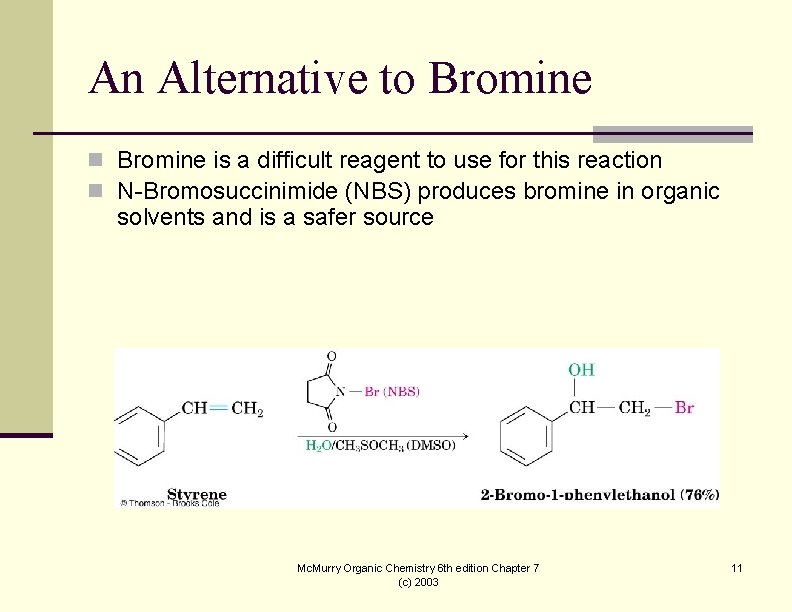
An Alternative to Bromine n Bromine is a difficult reagent to use for this reaction n N-Bromosuccinimide (NBS) produces bromine in organic solvents and is a safer source Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 11

7. 4 Addition of Water to Alkenes: Oxymercuration n Hydration of an alkene is the addition of H-OH to to give an alcohol n Acid catalysts are used in high temperature industrial processes: ethylene is converted to ethanol Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 12
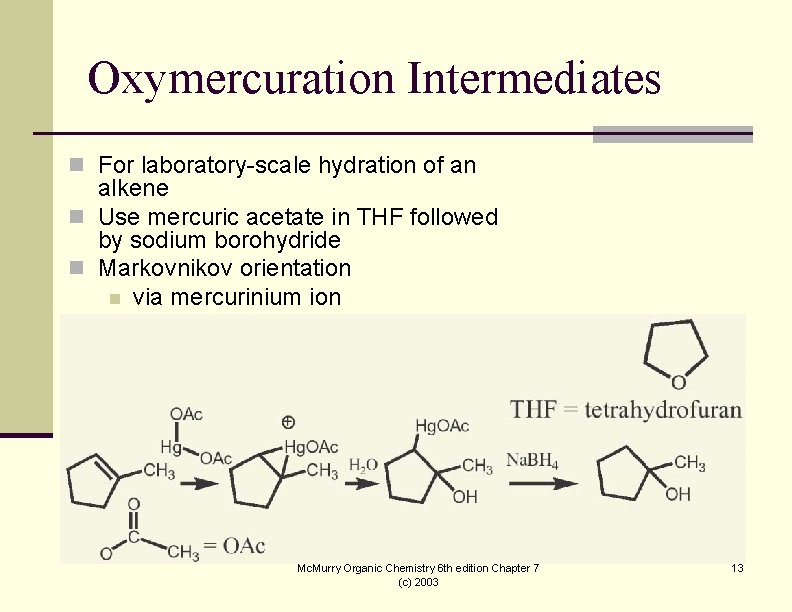
Oxymercuration Intermediates n For laboratory-scale hydration of an alkene n Use mercuric acetate in THF followed by sodium borohydride n Markovnikov orientation n via mercurinium ion Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 13
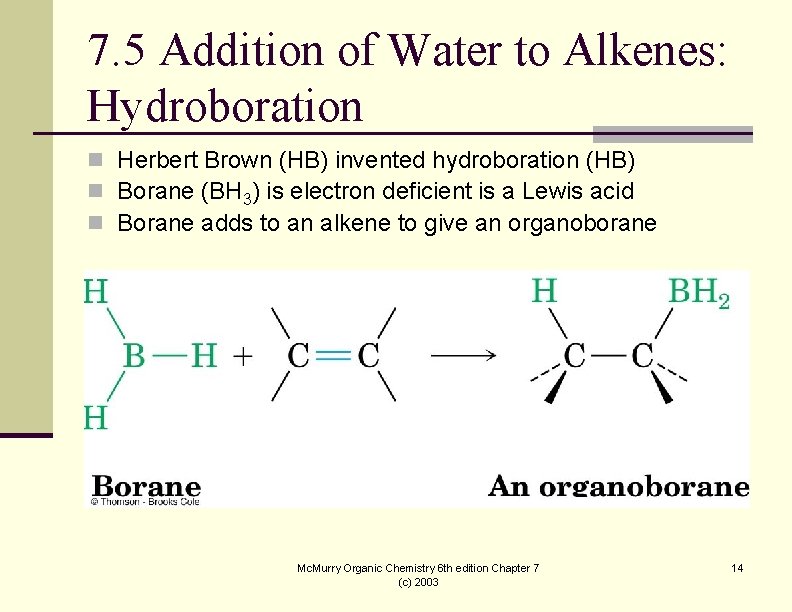
7. 5 Addition of Water to Alkenes: Hydroboration n Herbert Brown (HB) invented hydroboration (HB) n Borane (BH 3) is electron deficient is a Lewis acid n Borane adds to an alkene to give an organoborane Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 14
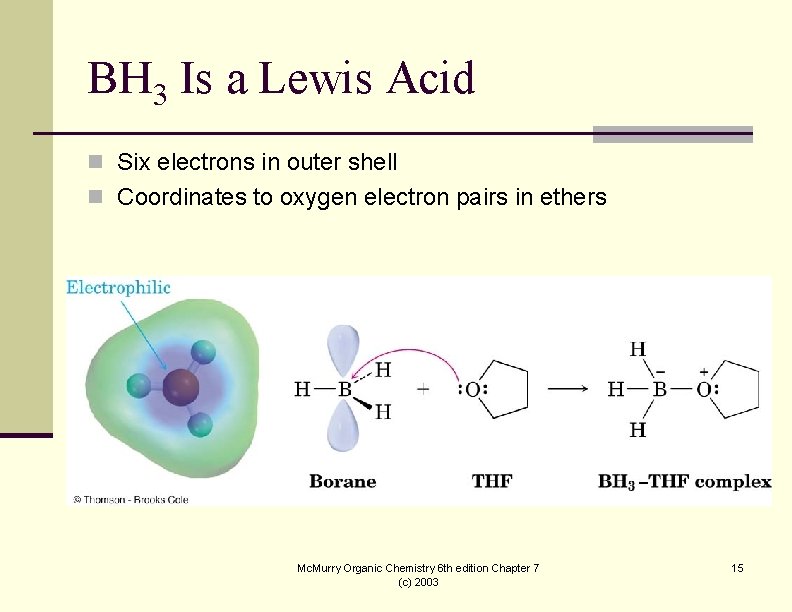
BH 3 Is a Lewis Acid n Six electrons in outer shell n Coordinates to oxygen electron pairs in ethers Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 15
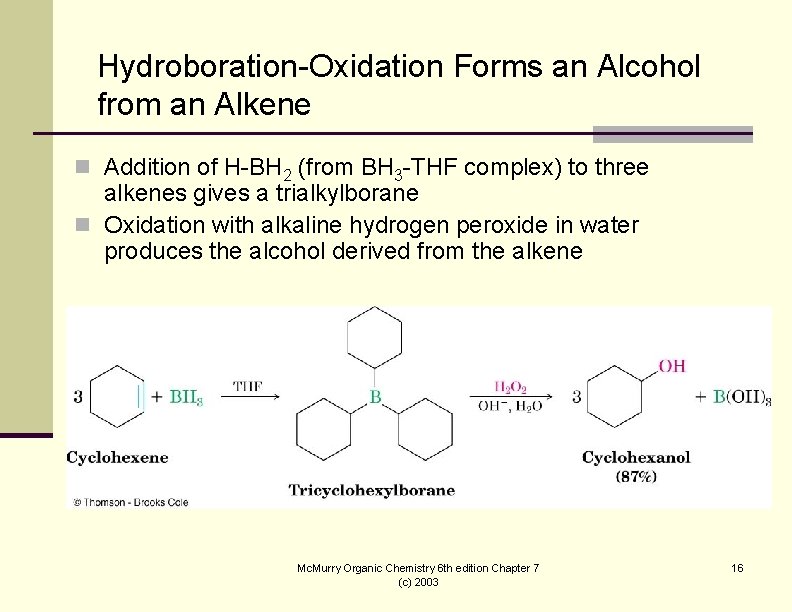
Hydroboration-Oxidation Forms an Alcohol from an Alkene n Addition of H-BH 2 (from BH 3 -THF complex) to three alkenes gives a trialkylborane n Oxidation with alkaline hydrogen peroxide in water produces the alcohol derived from the alkene Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 16
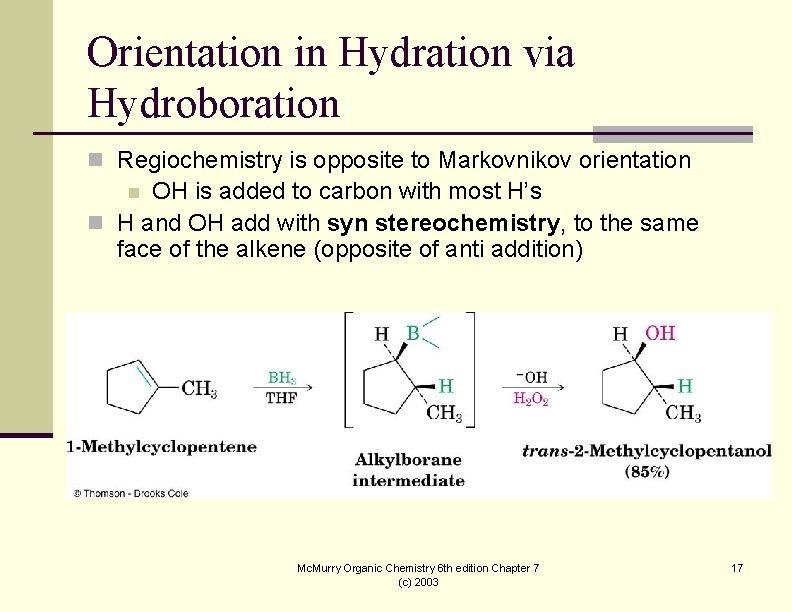
Orientation in Hydration via Hydroboration n Regiochemistry is opposite to Markovnikov orientation OH is added to carbon with most H’s n H and OH add with syn stereochemistry, to the same face of the alkene (opposite of anti addition) n Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 17
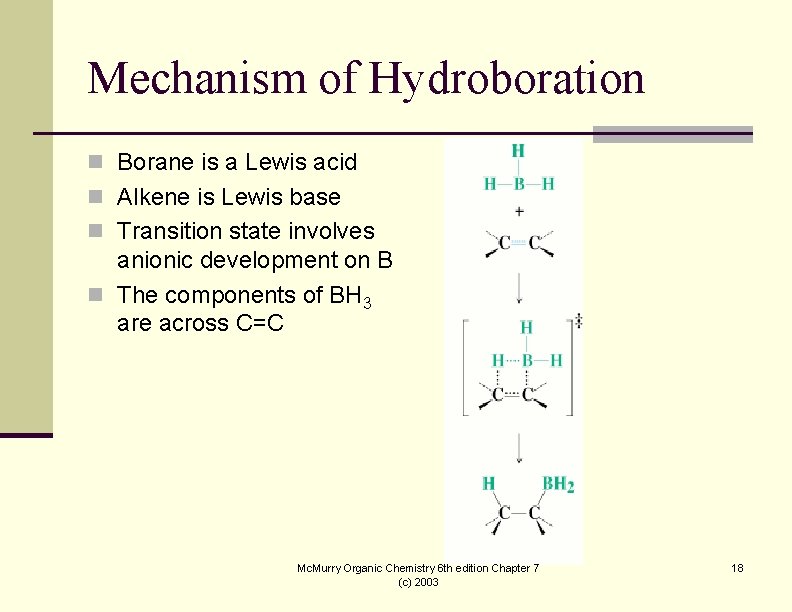
Mechanism of Hydroboration n Borane is a Lewis acid n Alkene is Lewis base n Transition state involves anionic development on B n The components of BH 3 are across C=C Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 18
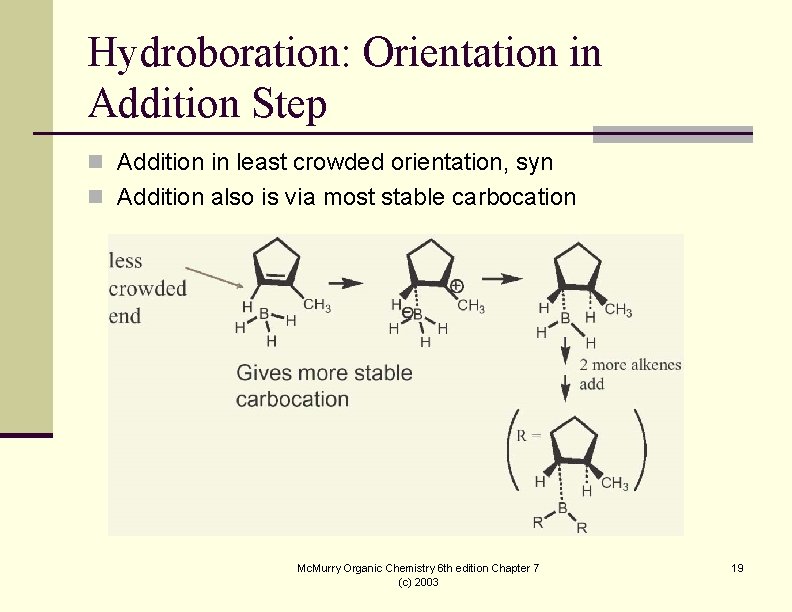
Hydroboration: Orientation in Addition Step n Addition in least crowded orientation, syn n Addition also is via most stable carbocation Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 19
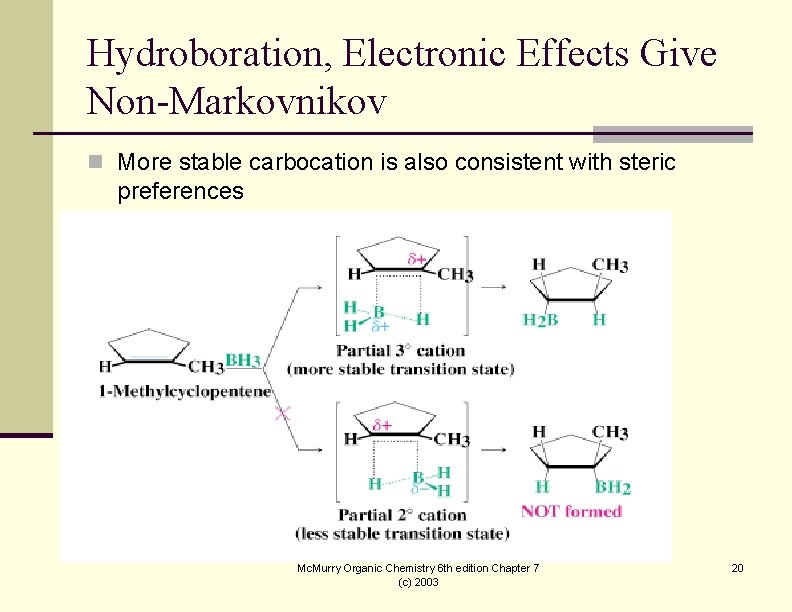
Hydroboration, Electronic Effects Give Non-Markovnikov n More stable carbocation is also consistent with steric preferences Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 20
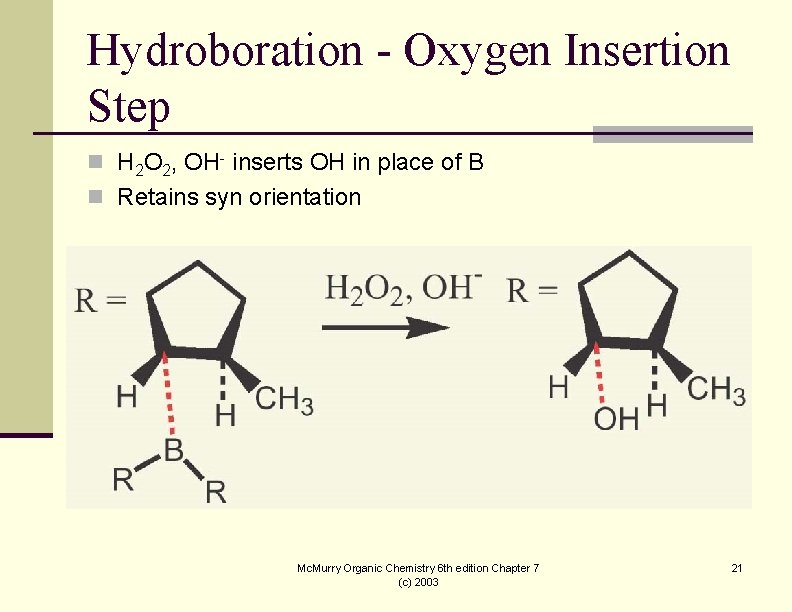
Hydroboration - Oxygen Insertion Step n H 2 O 2, OH- inserts OH in place of B n Retains syn orientation Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 21
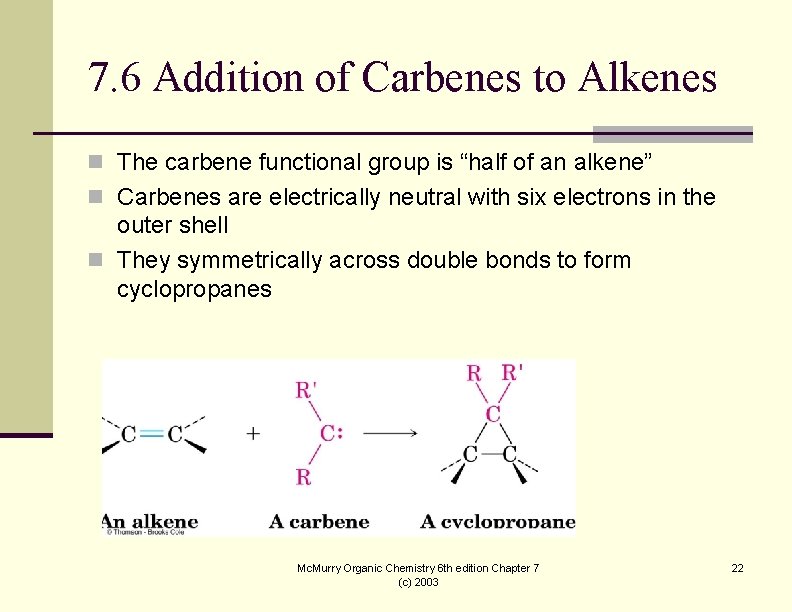
7. 6 Addition of Carbenes to Alkenes n The carbene functional group is “half of an alkene” n Carbenes are electrically neutral with six electrons in the outer shell n They symmetrically across double bonds to form cyclopropanes Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 22

Formation of Dichlorocarbene n Base removes proton from chloroform n Stabilized carbanion remains n Unimolecular Elimination of Cl- gives electron deficient species, dichlorocarbene Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 23

Simmons-Smith Reaction n Equivalent of addition of CH 2: n Reaction of diiodomethane with zinc-copper alloy produces a carbenoid species n Forms cyclopropanes by cycloaddition Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 24
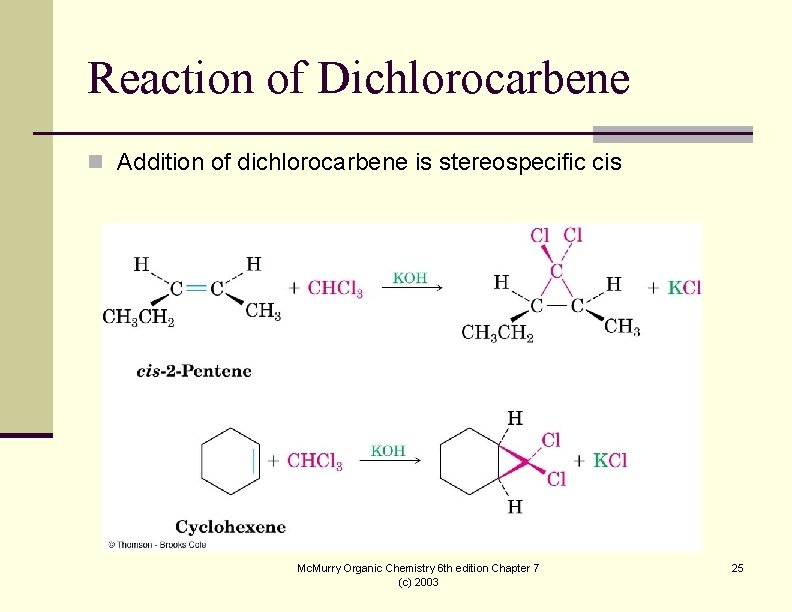
Reaction of Dichlorocarbene n Addition of dichlorocarbene is stereospecific cis Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 25

7. 7 Reduction of Alkenes: Hydrogenation n n Addition of H-H across C=C Reduction in general is addition of H 2 or its equivalent Requires Pt or Pd as powders on carbon and H 2 Hydrogen is first adsorbed on catalyst Reaction is heterogeneous (process is not in solution) Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 26
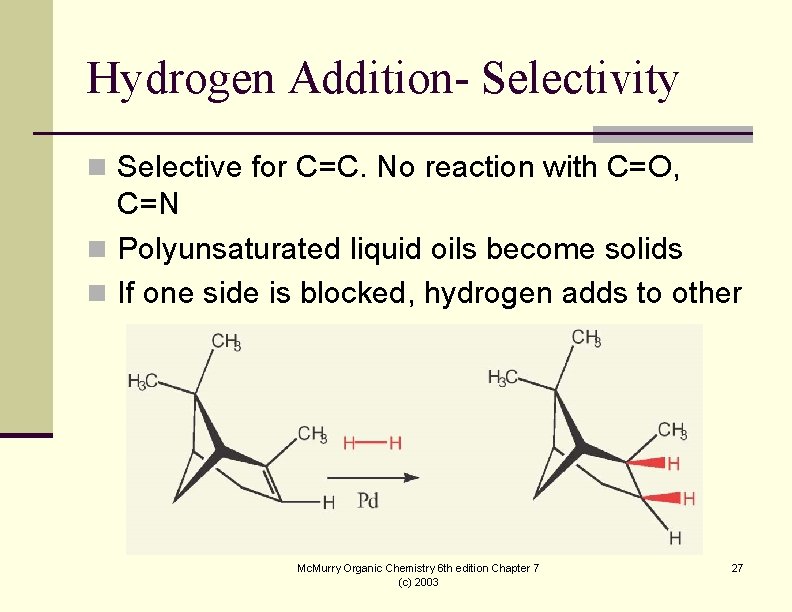
Hydrogen Addition- Selectivity n Selective for C=C. No reaction with C=O, C=N n Polyunsaturated liquid oils become solids n If one side is blocked, hydrogen adds to other Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 27
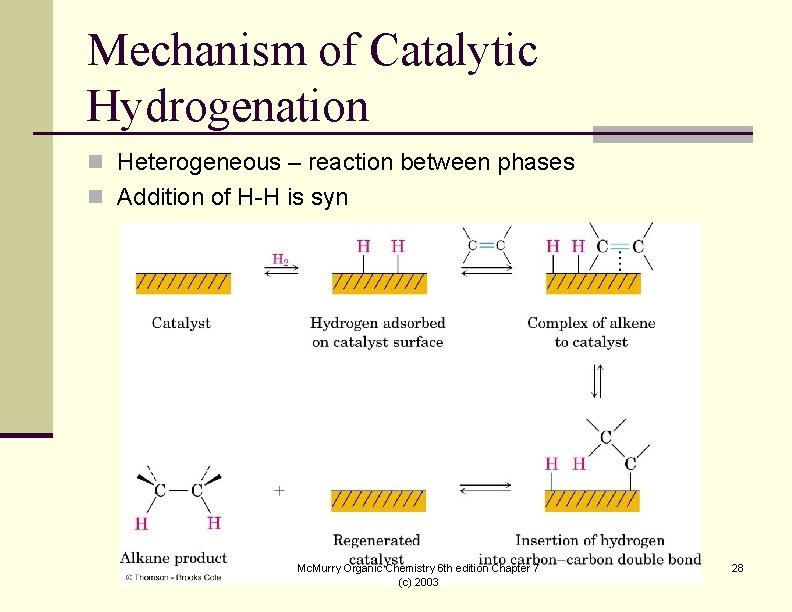
Mechanism of Catalytic Hydrogenation n Heterogeneous – reaction between phases n Addition of H-H is syn Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 28
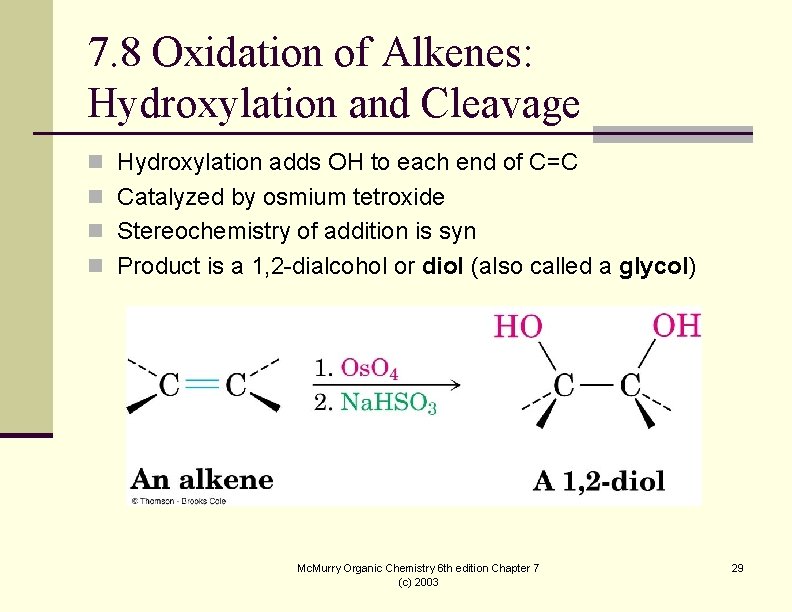
7. 8 Oxidation of Alkenes: Hydroxylation and Cleavage n Hydroxylation adds OH to each end of C=C n Catalyzed by osmium tetroxide n Stereochemistry of addition is syn n Product is a 1, 2 -dialcohol or diol (also called a glycol) Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 29
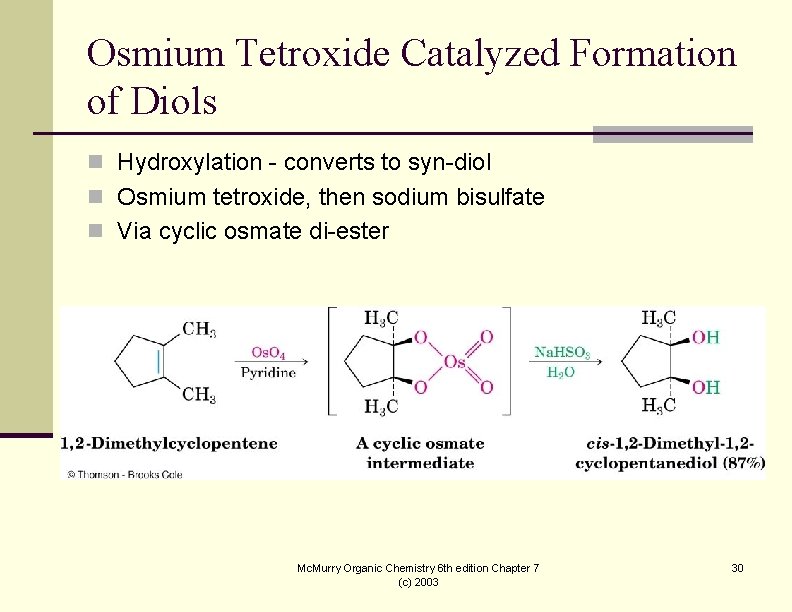
Osmium Tetroxide Catalyzed Formation of Diols n Hydroxylation - converts to syn-diol n Osmium tetroxide, then sodium bisulfate n Via cyclic osmate di-ester Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 30
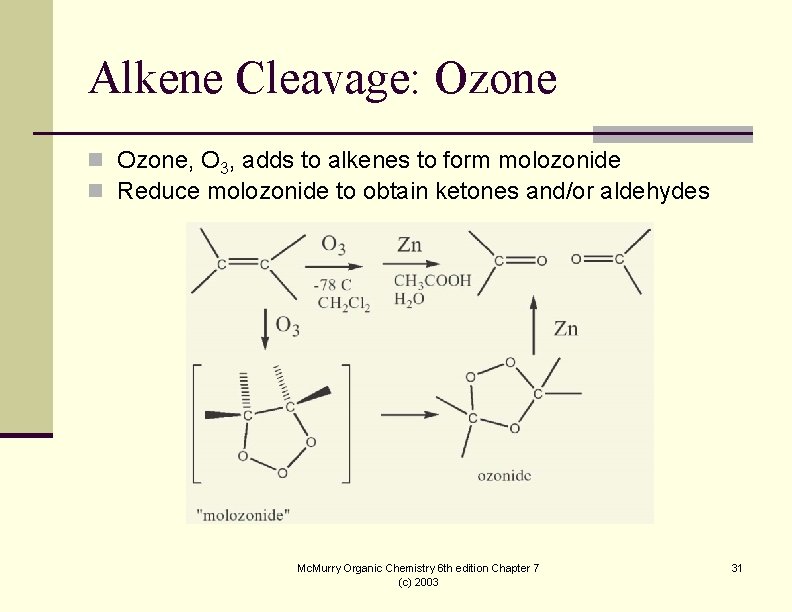
Alkene Cleavage: Ozone n Ozone, O 3, adds to alkenes to form molozonide n Reduce molozonide to obtain ketones and/or aldehydes Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 31
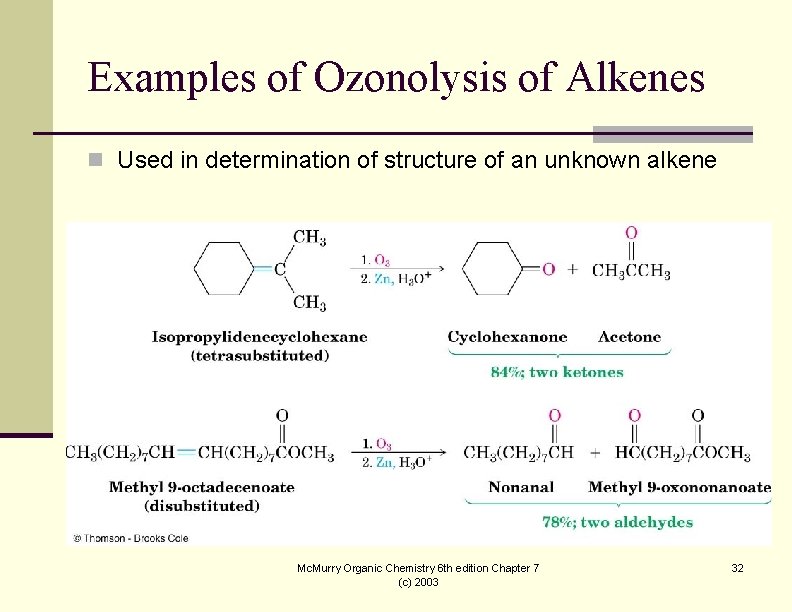
Examples of Ozonolysis of Alkenes n Used in determination of structure of an unknown alkene Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 32
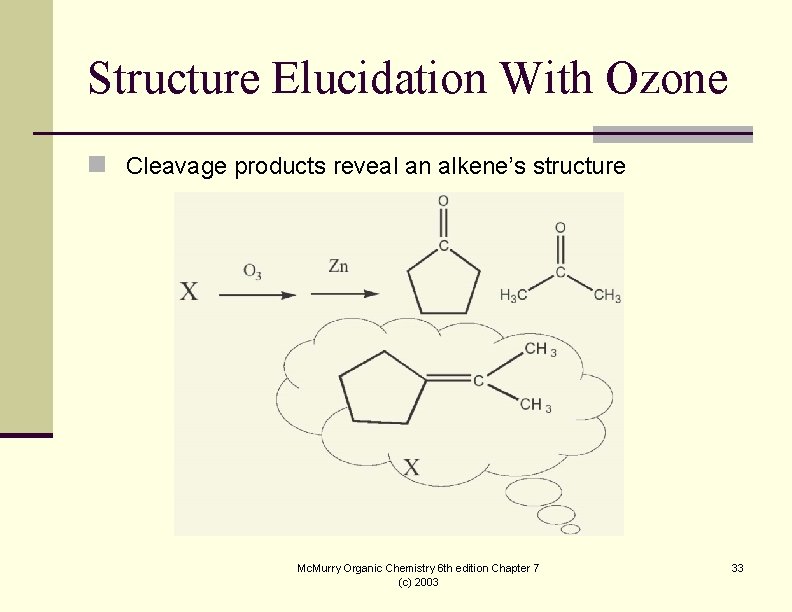
Structure Elucidation With Ozone n Cleavage products reveal an alkene’s structure Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 33

Permangante Oxidation of Alkenes n Oxidizing reagents other than ozone also cleave alkenes n Potassium permanganate (KMn. O 4) can produce carboxylic acids and carbon dioxide if H’s are present on C=C Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 34
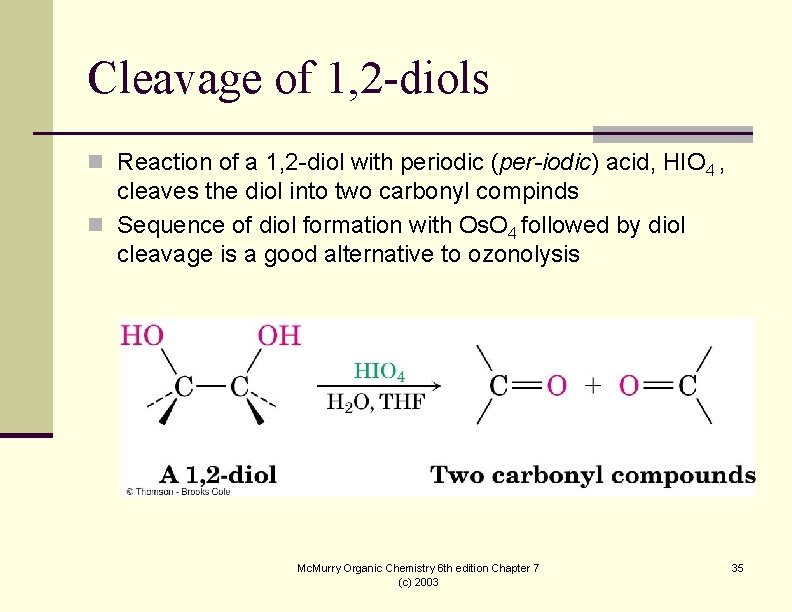
Cleavage of 1, 2 -diols n Reaction of a 1, 2 -diol with periodic (per-iodic) acid, HIO 4 , cleaves the diol into two carbonyl compinds n Sequence of diol formation with Os. O 4 followed by diol cleavage is a good alternative to ozonolysis Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 35
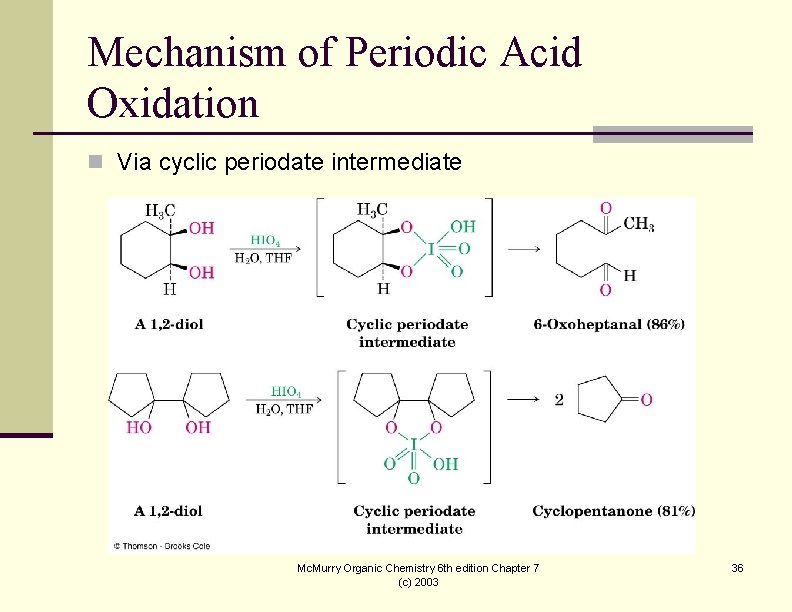
Mechanism of Periodic Acid Oxidation n Via cyclic periodate intermediate Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 36

7. 9 Biological Alkene Addition Reactions n Living organisms convert organic molecules using enzymes as catalysts n Many reactions are similar to organic chemistry conversions, except they occur in neutral water n Usually much specific for reactant and stereochemistry Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 37
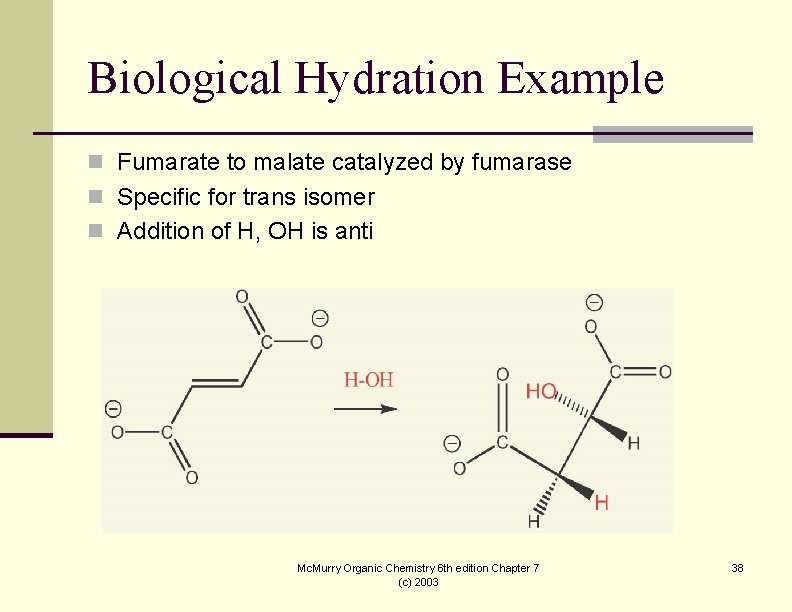
Biological Hydration Example n Fumarate to malate catalyzed by fumarase n Specific for trans isomer n Addition of H, OH is anti Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 38

7. 10 Addition of Radicals to Alkenes: Polymers n A polymer is a very large molecule consisting of repeating units of simpler molecules, formed by polymerization n Alkenes react with radical catalysts to undergo radical polymerization n Ethylene is polymerized to poyethylene, for example Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 39

Free Radical Polymerization of Alkenes n Alkenes combine many times to give polymer n Reactivity induced by formation of free radicals Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 40

Free Radical Polymerization: Initiation n Initiation - a few radicals are generated by the reaction of a molecule that readily forms radicals from a nonradical molecule n A bond is broken homolytically Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 41

Polymerization: Propagation n Radical from intiation adds to alkene to generate alkene derived radical n This radical adds to another alkene, and so on many times Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 42
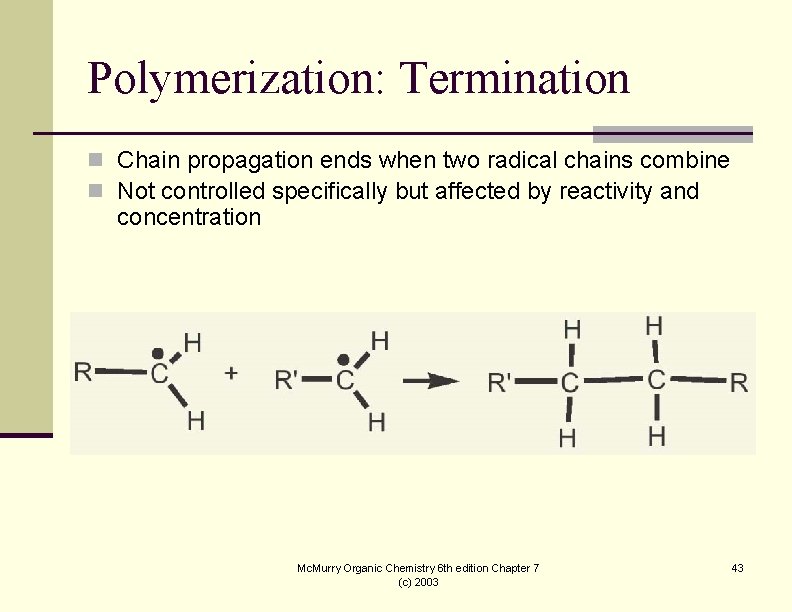
Polymerization: Termination n Chain propagation ends when two radical chains combine n Not controlled specifically but affected by reactivity and concentration Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 43
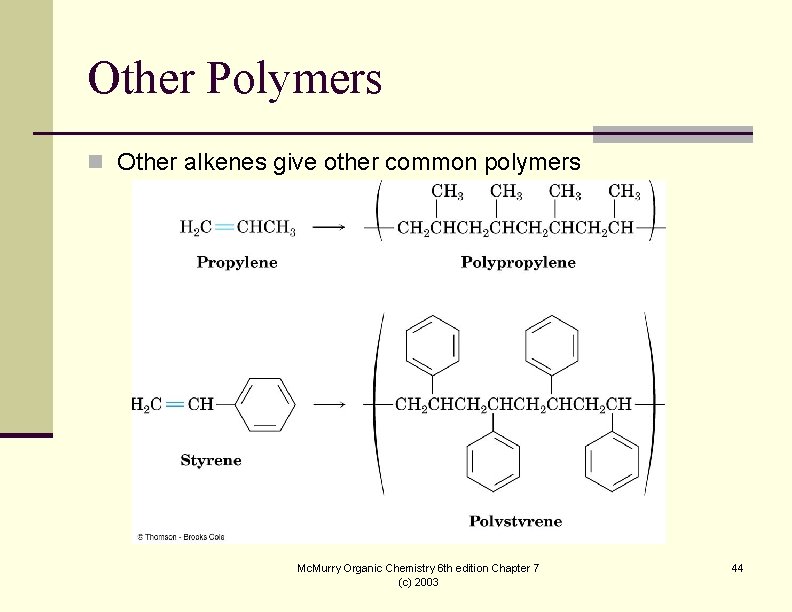
Other Polymers n Other alkenes give other common polymers Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 44

Unsymmetrical Monomers n If alkene is unsymmetrical, reaction is via more highly substituted radical Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 45
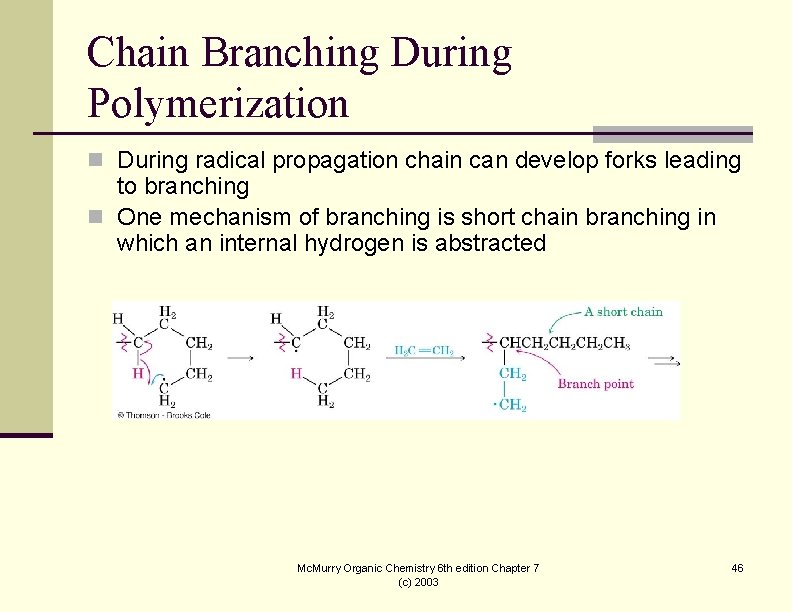
Chain Branching During Polymerization n During radical propagation chain can develop forks leading to branching n One mechanism of branching is short chain branching in which an internal hydrogen is abstracted Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 46
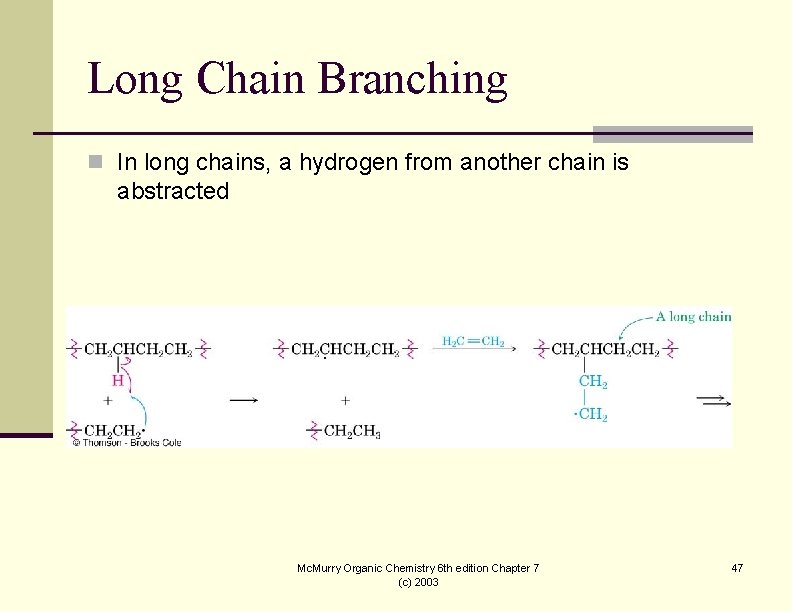
Long Chain Branching n In long chains, a hydrogen from another chain is abstracted Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 47

Cationic Polymerization n Vinyl monomers react with Brønsted or Lewis acid to produce a reactive carbocation that adds to alkenes and propagates via lengthening carbocations Mc. Murry Organic Chemistry 6 th edition Chapter 7 (c) 2003 48
- Slides: 48