Unsaturated hydrocarbons Unsaturated hydrocarbons ALKENES Alkenes shape Ethene

Unsaturated hydrocarbons
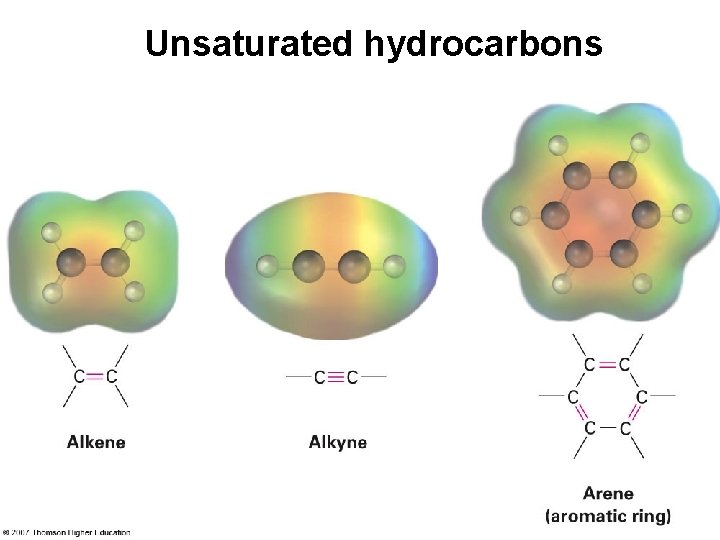
Unsaturated hydrocarbons

ALKENES
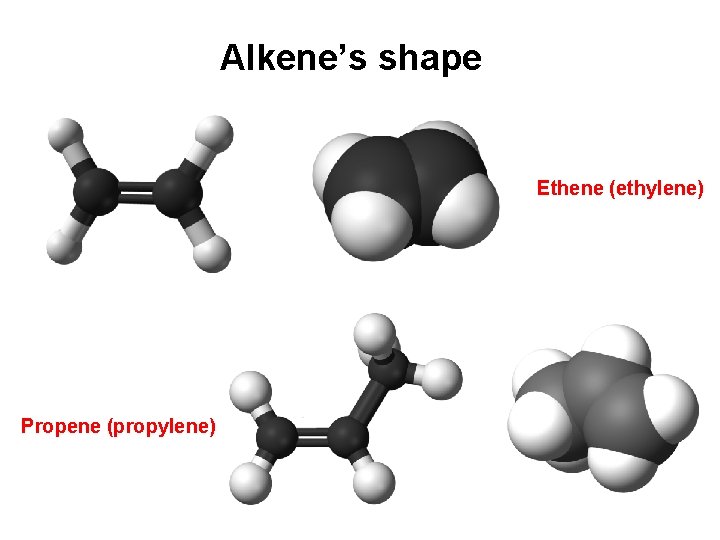
Alkene’s shape Ethene (ethylene) Propene (propylene)
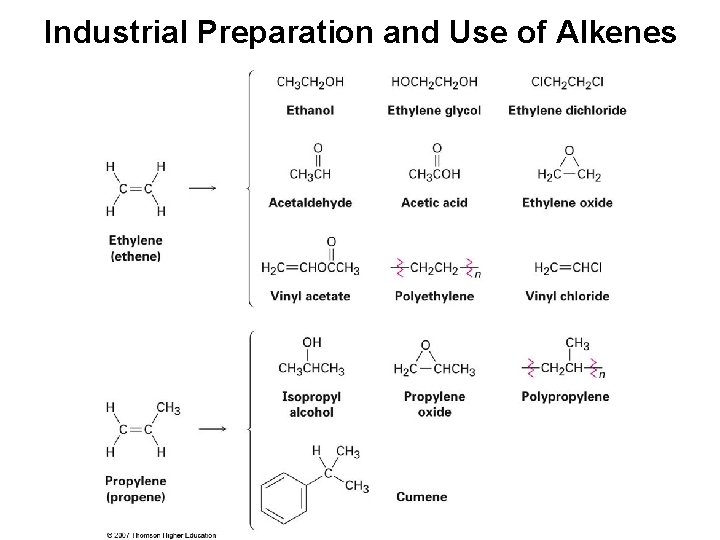
Industrial Preparation and Use of Alkenes
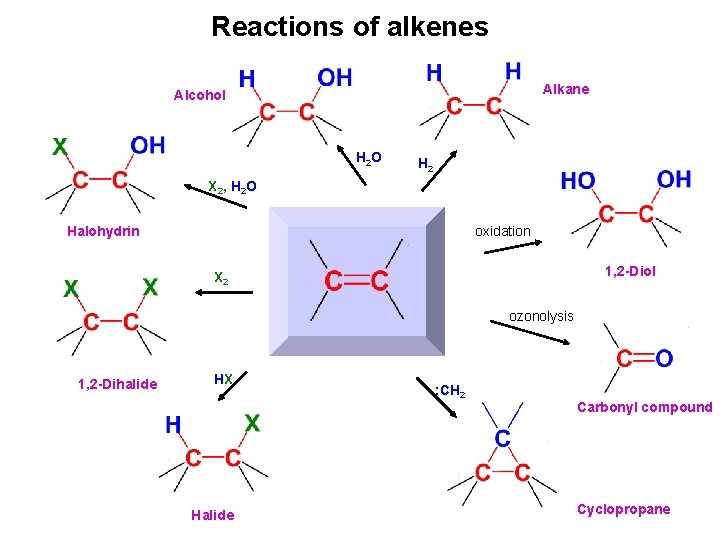
Reactions of alkenes Alkane Alcohol H 2 O H 2 X 2, H 2 O Halohydrin oxidation 1, 2 -Diol X 2 ozonolysis 1, 2 -Dihalide HX Halide : CH 2 Carbonyl compound Cyclopropane
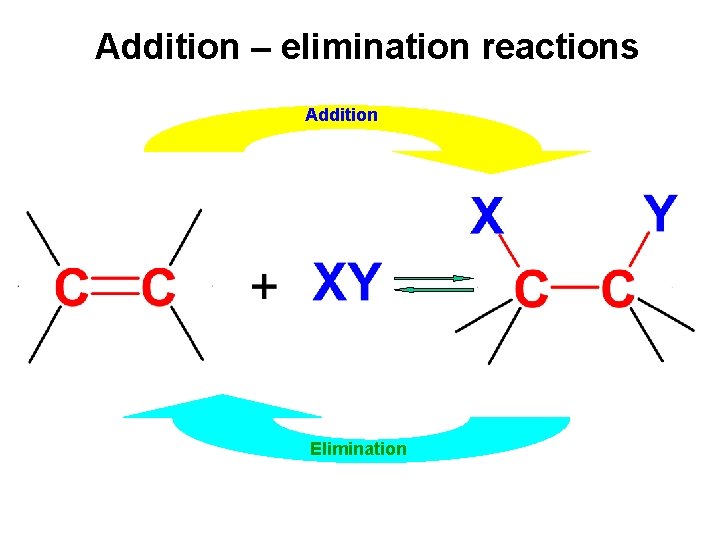
Addition – elimination reactions Addition Elimination
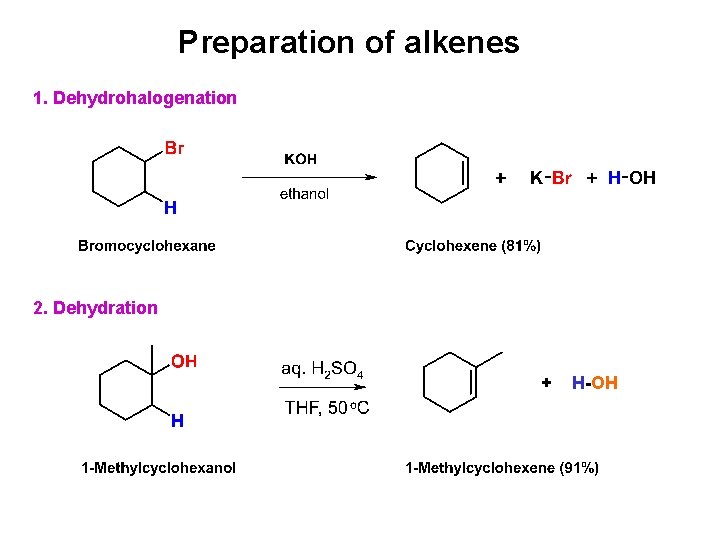
Preparation of alkenes 1. Dehydrohalogenation 2. Dehydration + H-OH
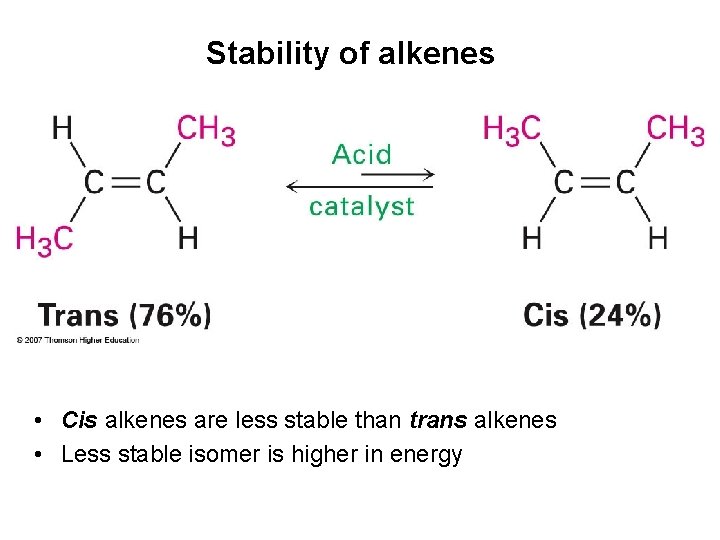
Stability of alkenes • Cis alkenes are less stable than trans alkenes • Less stable isomer is higher in energy
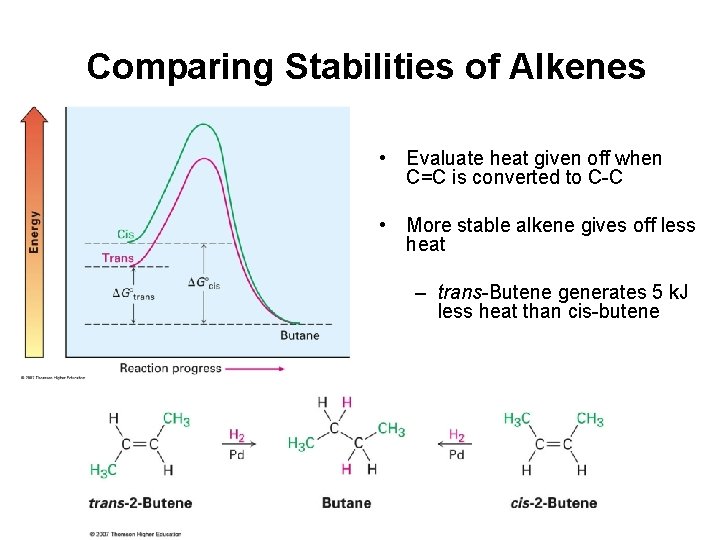
Comparing Stabilities of Alkenes • Evaluate heat given off when C=C is converted to C-C • More stable alkene gives off less heat – trans-Butene generates 5 k. J less heat than cis-butene
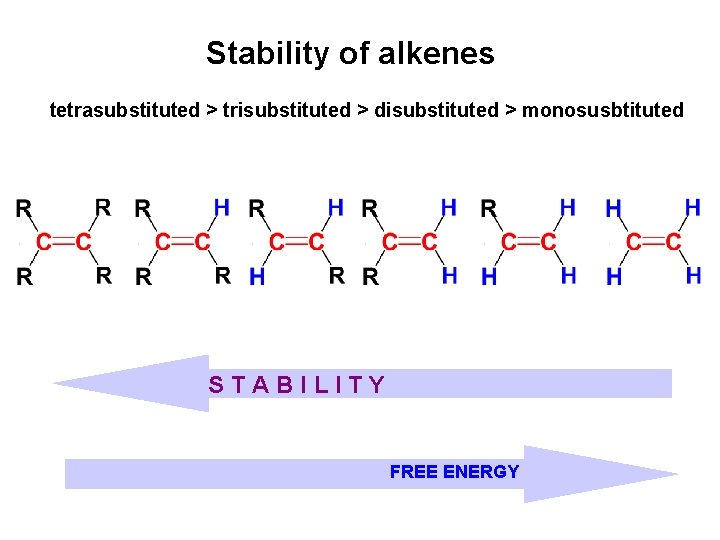
Stability of alkenes tetrasubstituted > trisubstituted > disubstituted > monosusbtituted STABILITY FREE ENERGY
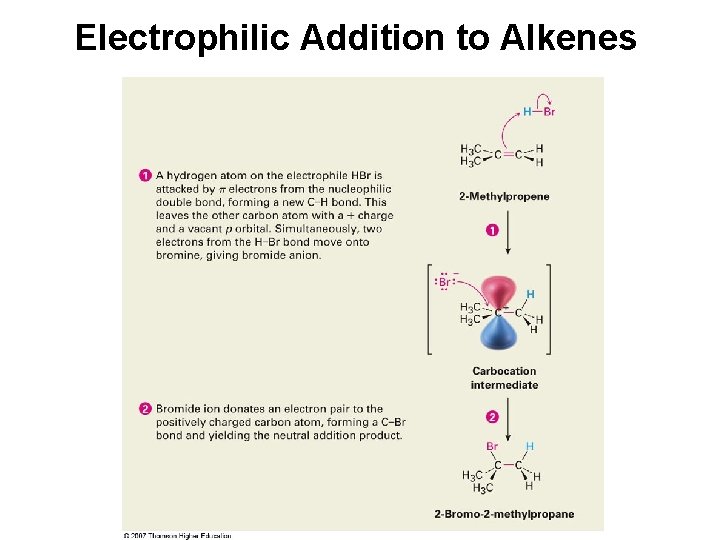
Electrophilic Addition to Alkenes
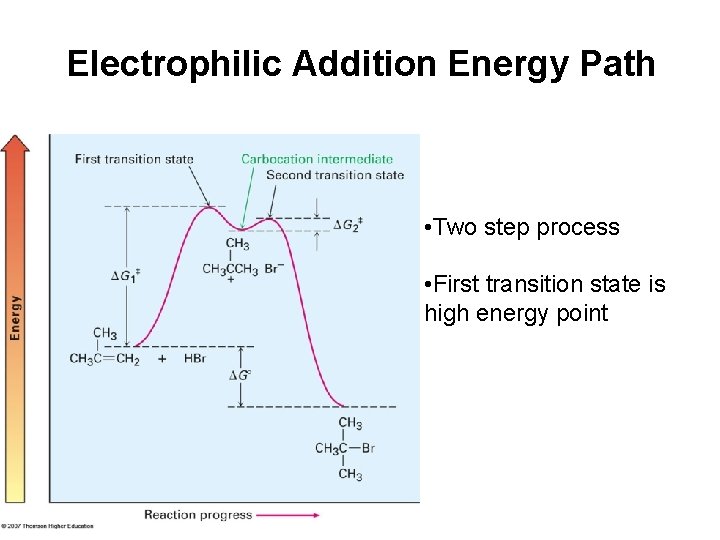
Electrophilic Addition Energy Path • Two step process • First transition state is high energy point
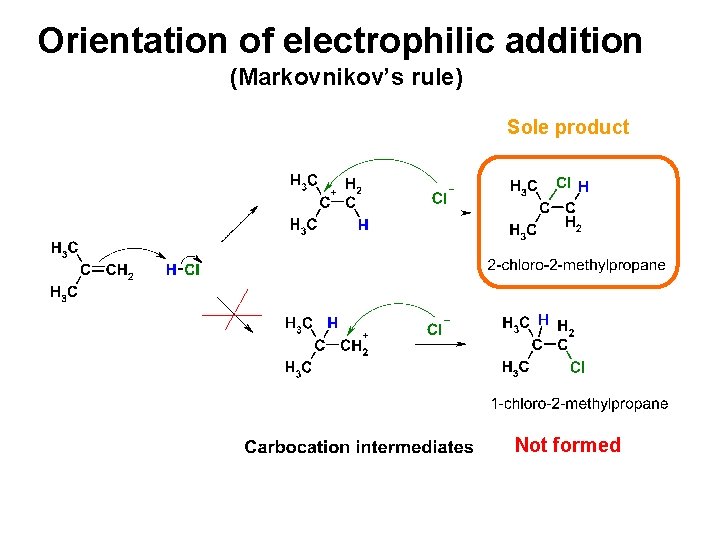
Orientation of electrophilic addition (Markovnikov’s rule) Sole product Not formed
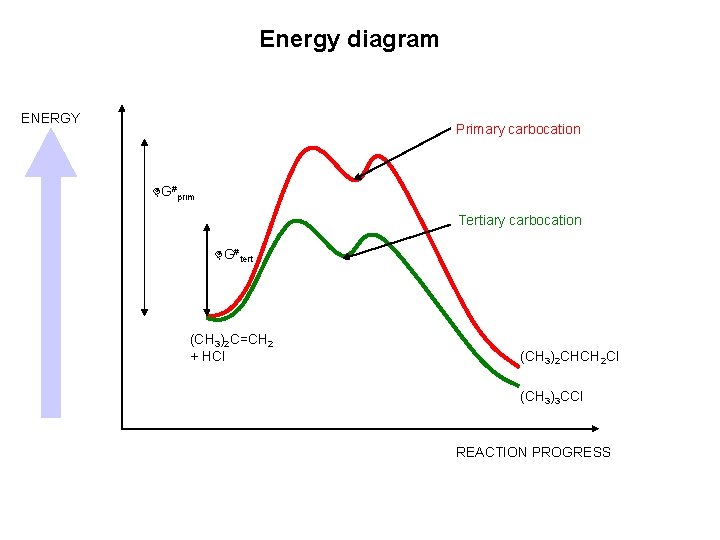
Energy diagram ENERGY Primary carbocation G#prim Tertiary carbocation G#tert (CH 3)2 C=CH 2 + HCl (CH 3)2 CHCH 2 Cl (CH 3)3 CCl REACTION PROGRESS
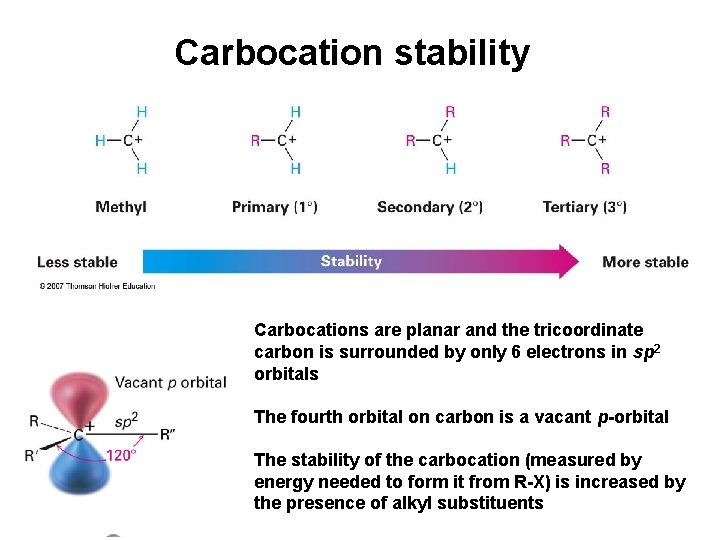
Carbocation stability Carbocations are planar and the tricoordinate carbon is surrounded by only 6 electrons in sp 2 orbitals The fourth orbital on carbon is a vacant p-orbital The stability of the carbocation (measured by energy needed to form it from R-X) is increased by the presence of alkyl substituents
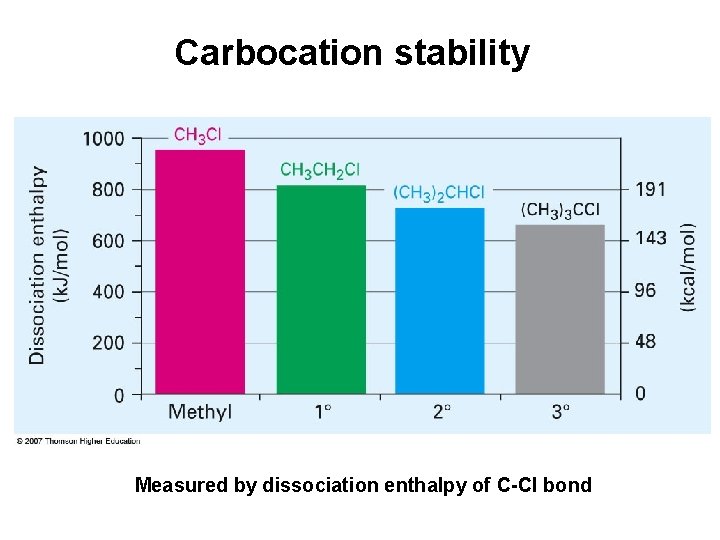
Carbocation stability Measured by dissociation enthalpy of C-Cl bond
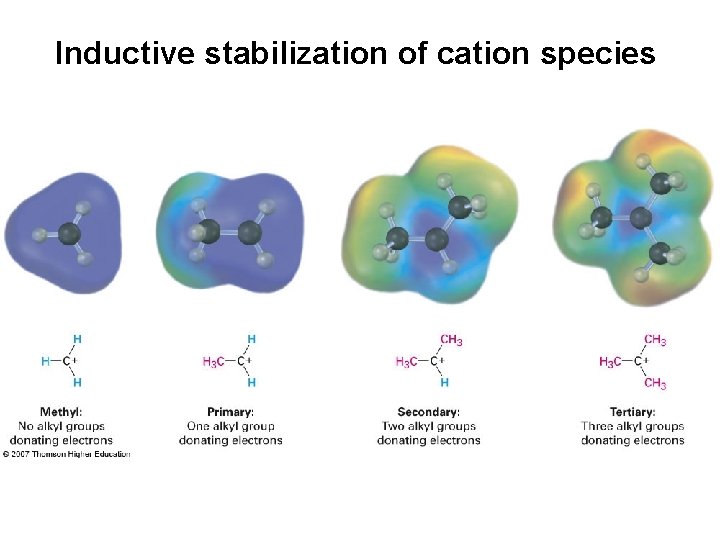
Inductive stabilization of cation species
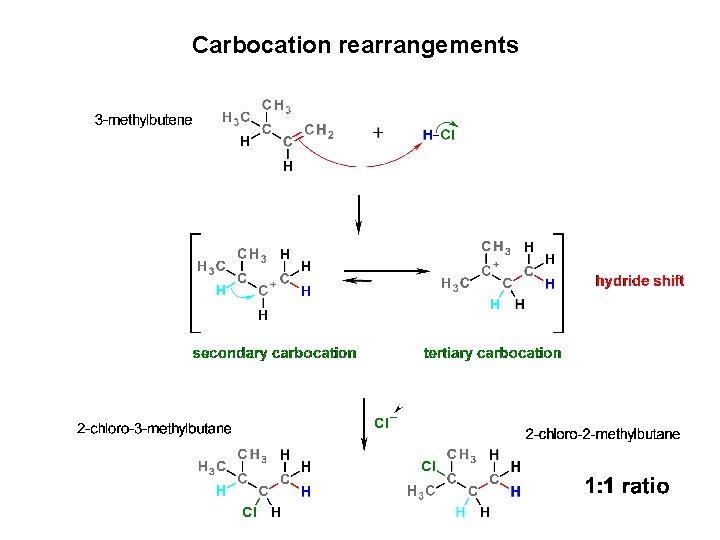
Carbocation rearrangements
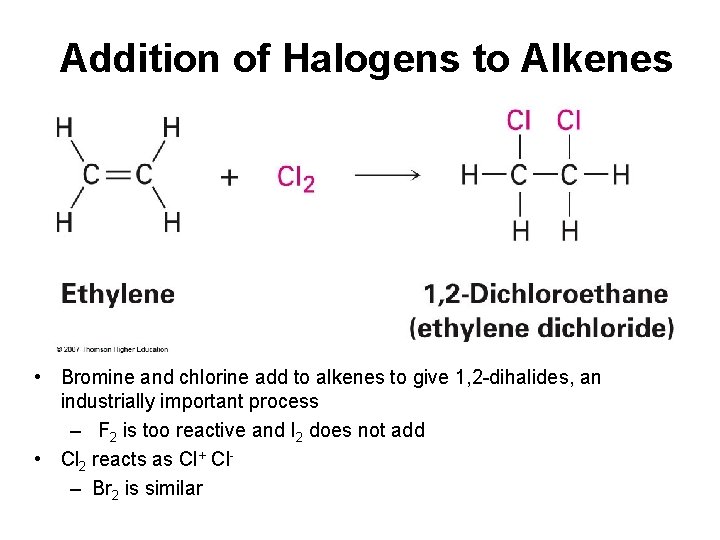
Addition of Halogens to Alkenes • Bromine and chlorine add to alkenes to give 1, 2 -dihalides, an industrially important process – F 2 is too reactive and I 2 does not add • Cl 2 reacts as Cl+ Cl– Br 2 is similar
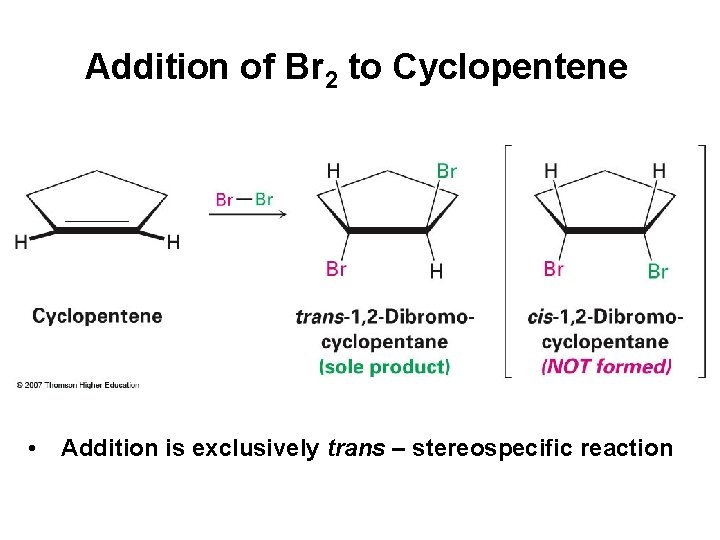
Addition of Br 2 to Cyclopentene • Addition is exclusively trans – stereospecific reaction
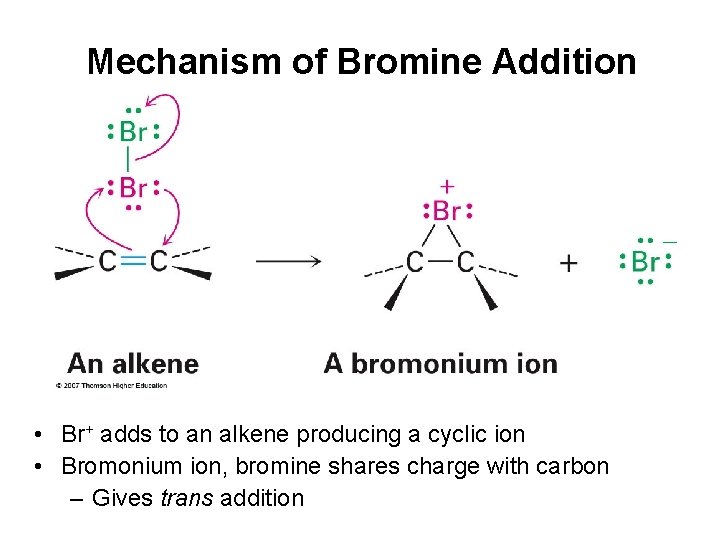
Mechanism of Bromine Addition • Br+ adds to an alkene producing a cyclic ion • Bromonium ion, bromine shares charge with carbon – Gives trans addition
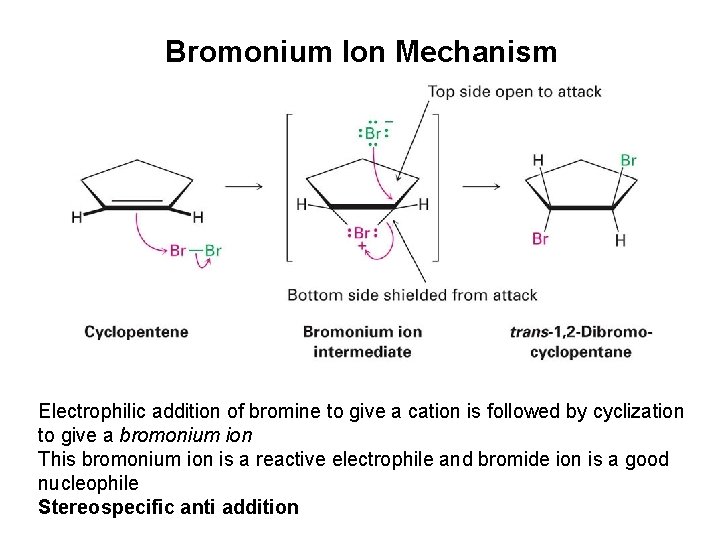
Bromonium Ion Mechanism Electrophilic addition of bromine to give a cation is followed by cyclization to give a bromonium ion This bromonium ion is a reactive electrophile and bromide ion is a good nucleophile Stereospecific anti addition
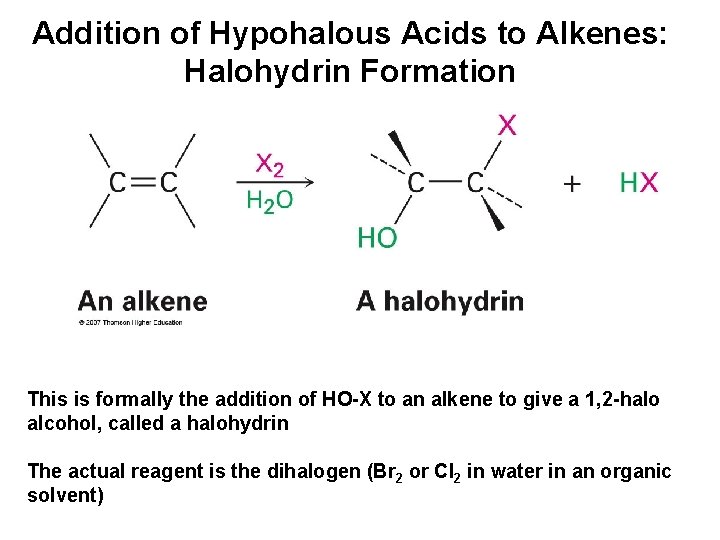
Addition of Hypohalous Acids to Alkenes: Halohydrin Formation This is formally the addition of HO-X to an alkene to give a 1, 2 -halo alcohol, called a halohydrin The actual reagent is the dihalogen (Br 2 or Cl 2 in water in an organic solvent)
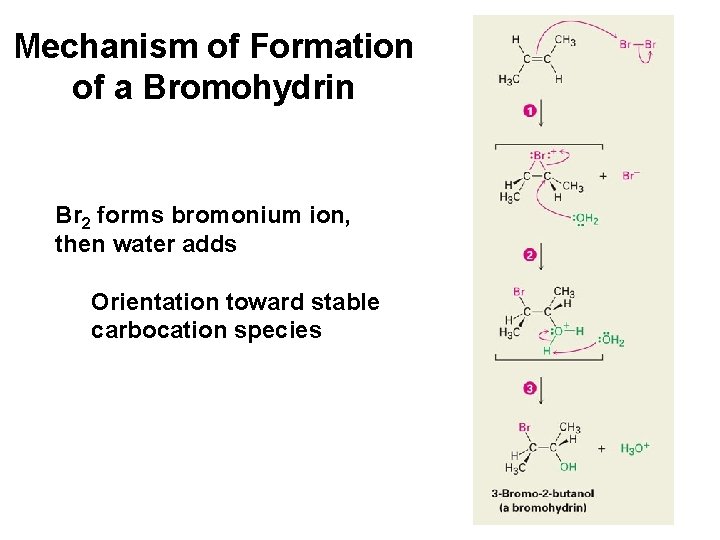
Mechanism of Formation of a Bromohydrin Br 2 forms bromonium ion, then water adds Orientation toward stable carbocation species
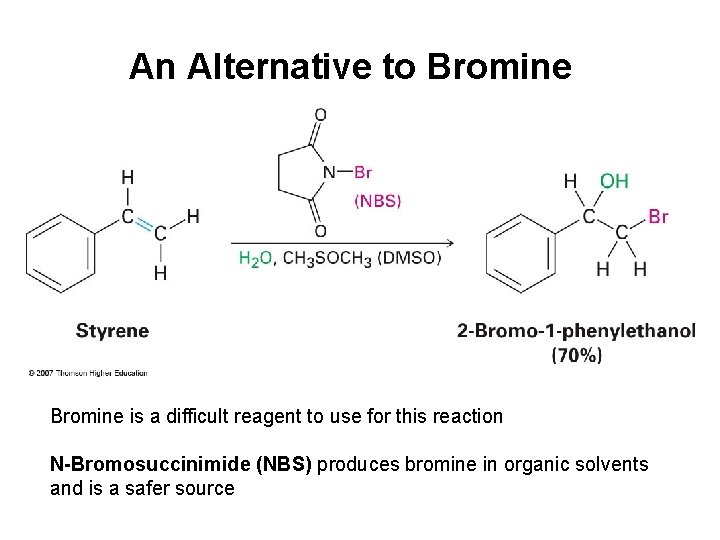
An Alternative to Bromine is a difficult reagent to use for this reaction N-Bromosuccinimide (NBS) produces bromine in organic solvents and is a safer source
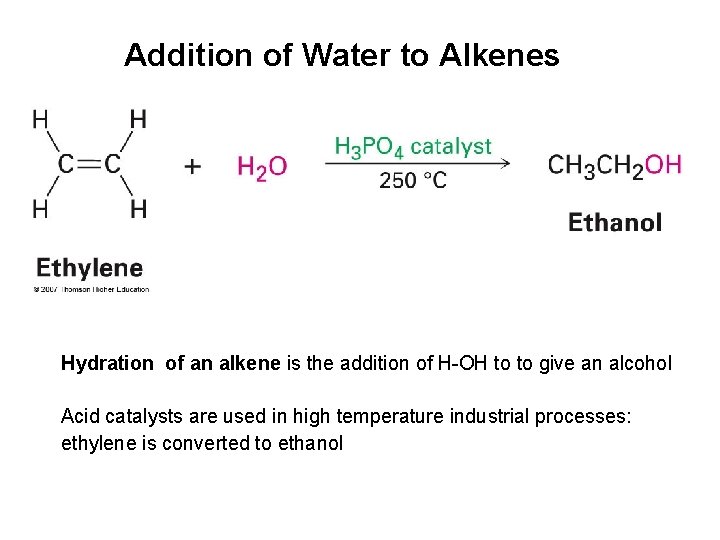
Addition of Water to Alkenes Hydration of an alkene is the addition of H-OH to to give an alcohol Acid catalysts are used in high temperature industrial processes: ethylene is converted to ethanol
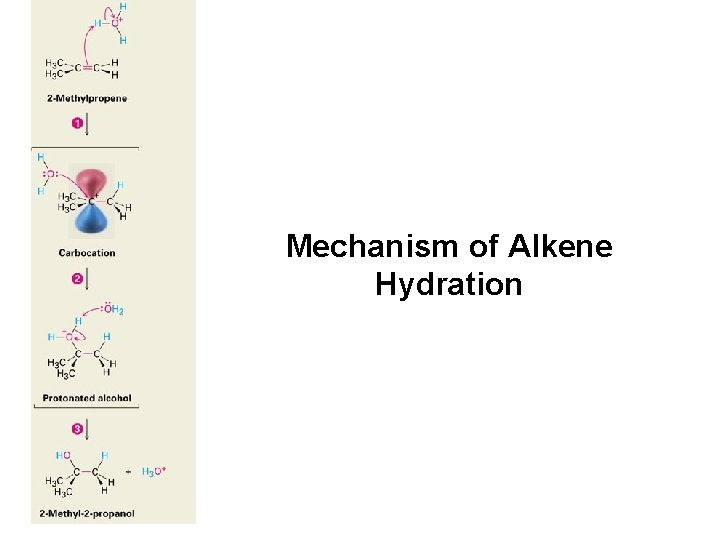
Mechanism of Alkene Hydration
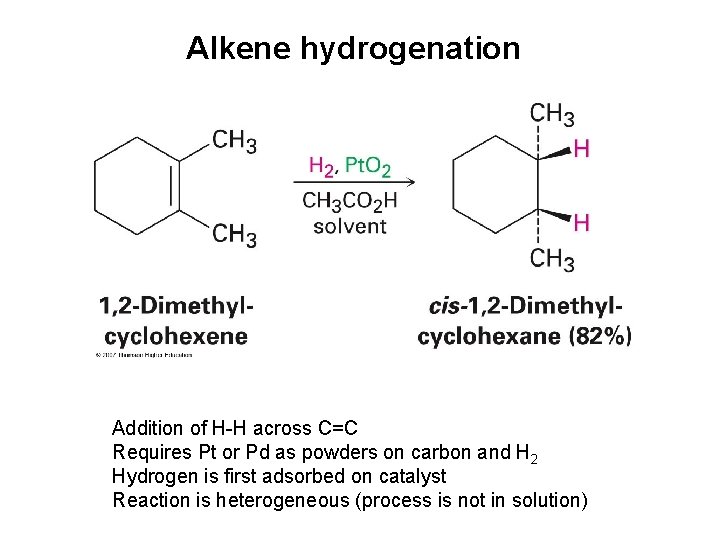
Alkene hydrogenation Addition of H-H across C=C Requires Pt or Pd as powders on carbon and H 2 Hydrogen is first adsorbed on catalyst Reaction is heterogeneous (process is not in solution)
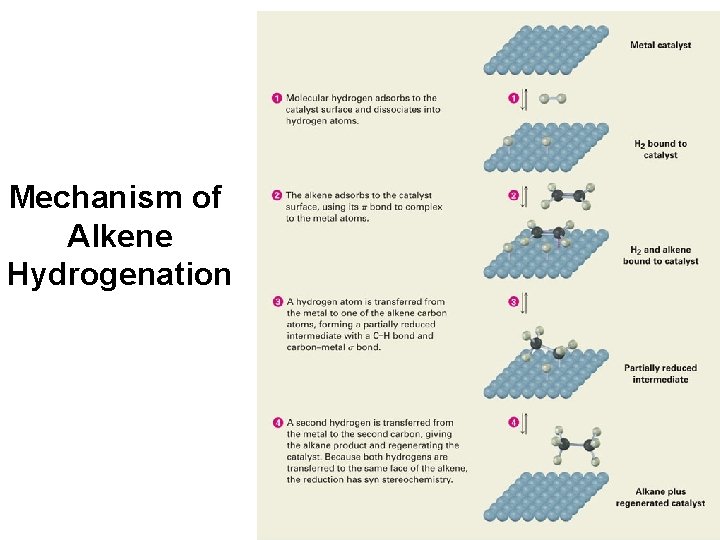
Mechanism of Alkene Hydrogenation
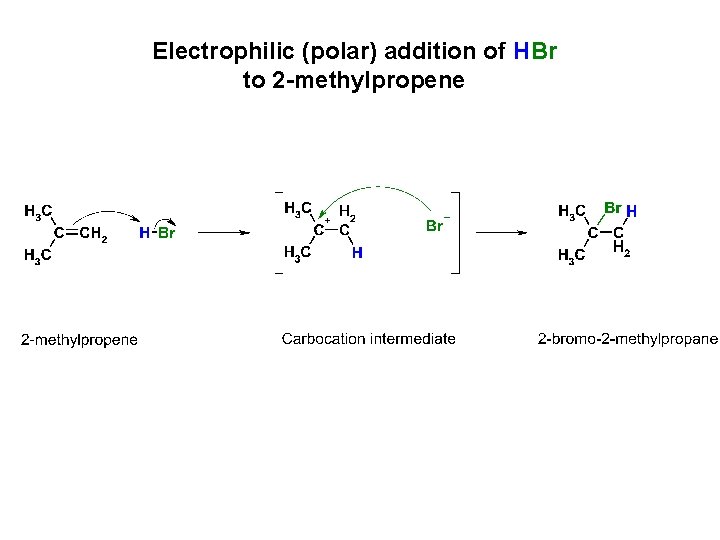
Electrophilic (polar) addition of HBr to 2 -methylpropene
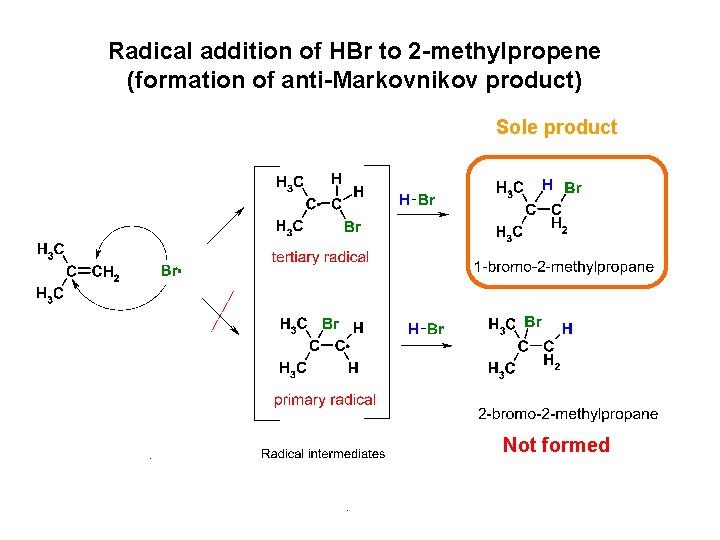
Radical addition of HBr to 2 -methylpropene (formation of anti-Markovnikov product) Sole product Not formed
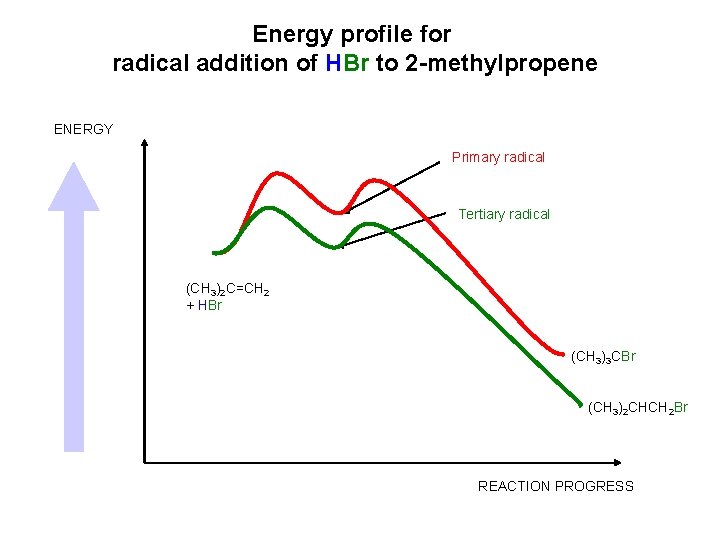
Energy profile for radical addition of HBr to 2 -methylpropene ENERGY Primary radical Tertiary radical (CH 3)2 C=CH 2 + HBr (CH 3)3 CBr (CH 3)2 CHCH 2 Br REACTION PROGRESS
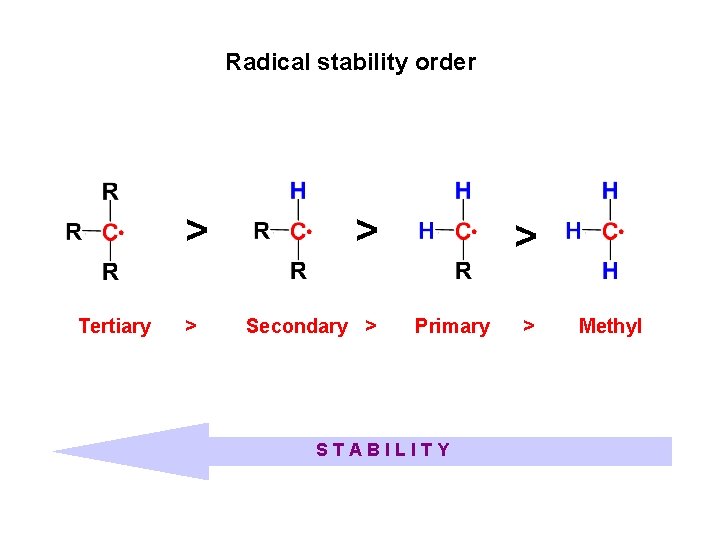
Radical stability order > Tertiary > > Secondary > > Primary STABILITY > Methyl

Alkene oxidation - epoxidation Epoxidation results in a cyclic ether with an oxygen atom Stereochemistry of addition is syn
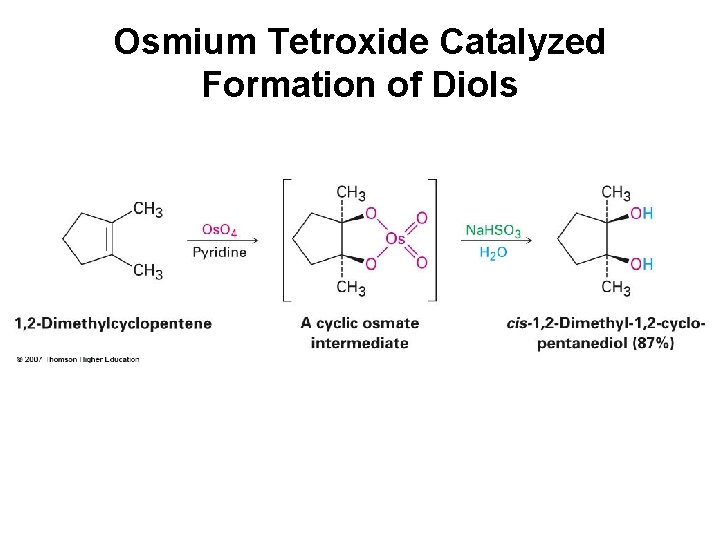
Osmium Tetroxide Catalyzed Formation of Diols
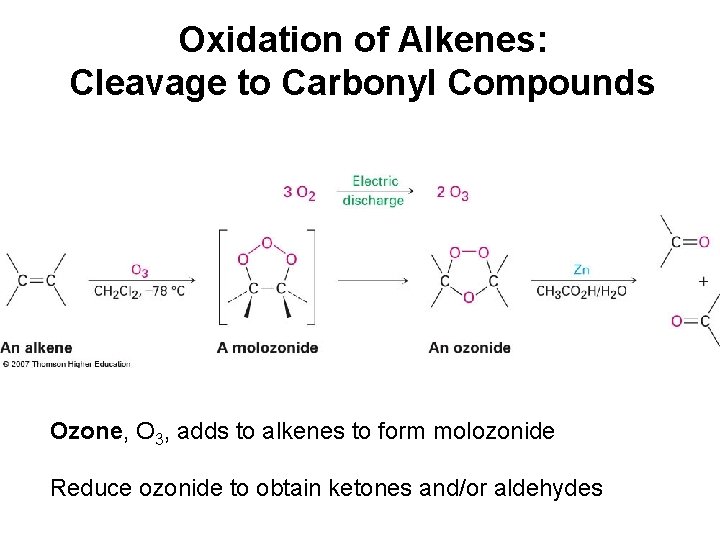
Oxidation of Alkenes: Cleavage to Carbonyl Compounds Ozone, O 3, adds to alkenes to form molozonide Reduce ozonide to obtain ketones and/or aldehydes
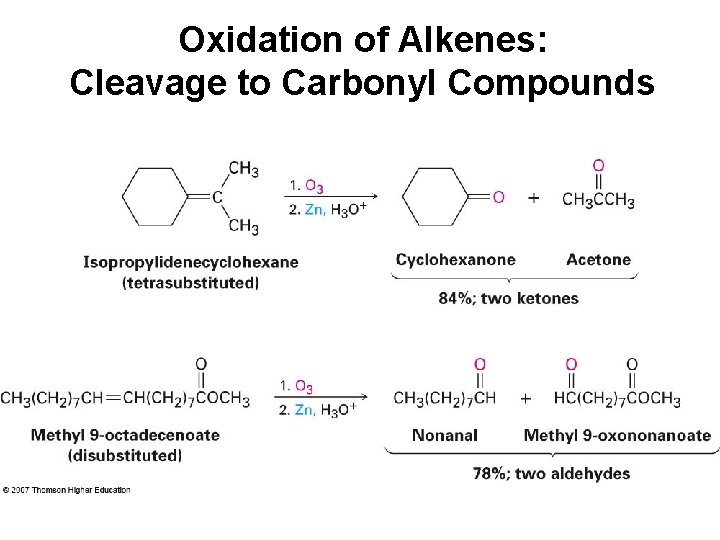
Oxidation of Alkenes: Cleavage to Carbonyl Compounds
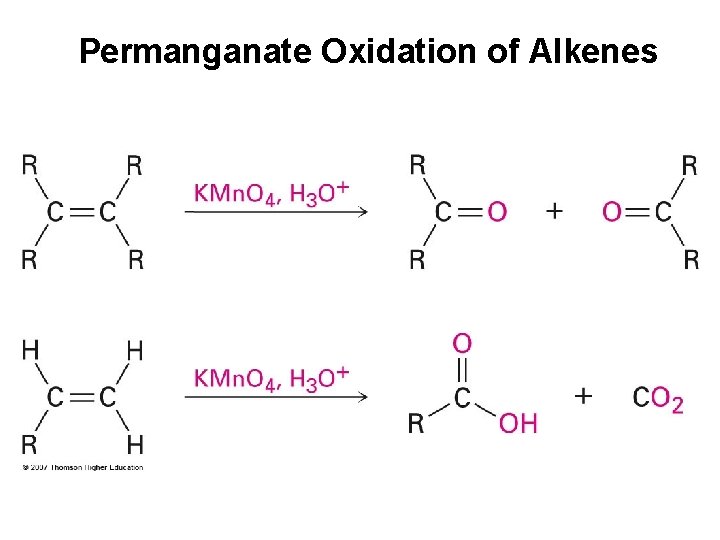
Permanganate Oxidation of Alkenes

ALKYNES
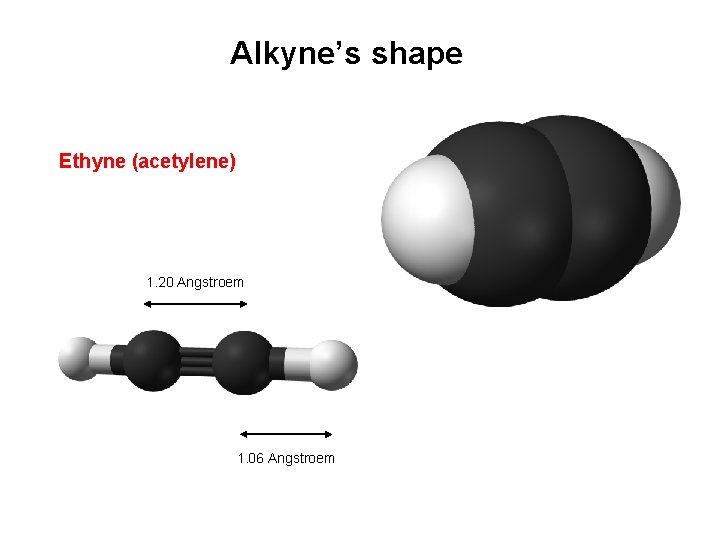
Alkyne’s shape Ethyne (acetylene) 1. 20 Angstroem 1. 06 Angstroem
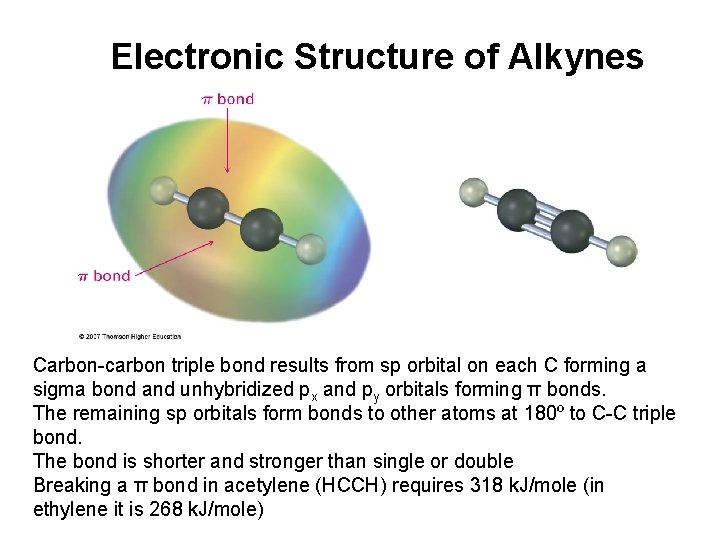
Electronic Structure of Alkynes Carbon-carbon triple bond results from sp orbital on each C forming a sigma bond and unhybridized px and py orbitals forming π bonds. The remaining sp orbitals form bonds to other atoms at 180º to C-C triple bond. The bond is shorter and stronger than single or double Breaking a π bond in acetylene (HCCH) requires 318 k. J/mole (in ethylene it is 268 k. J/mole)
1 -Butyne
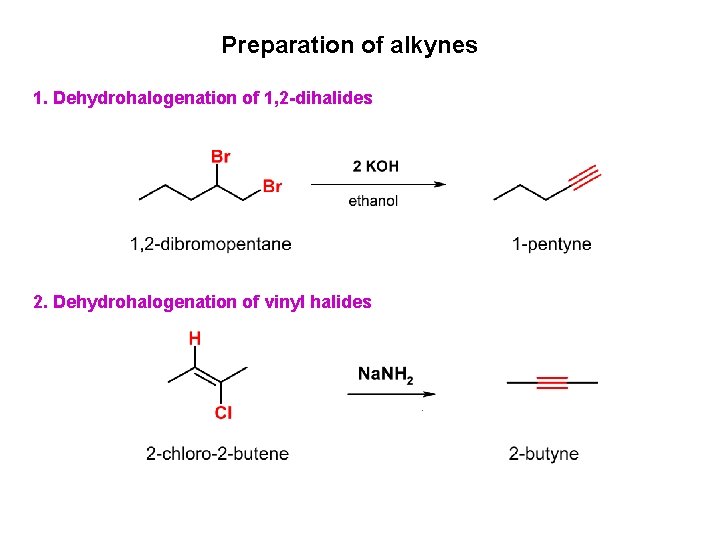
Preparation of alkynes 1. Dehydrohalogenation of 1, 2 -dihalides 2. Dehydrohalogenation of vinyl halides
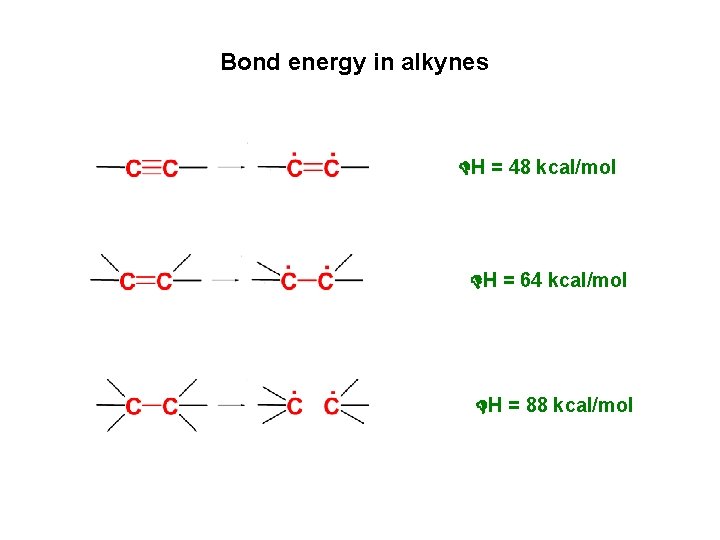
Bond energy in alkynes H = 48 kcal/mol H = 64 kcal/mol H = 88 kcal/mol
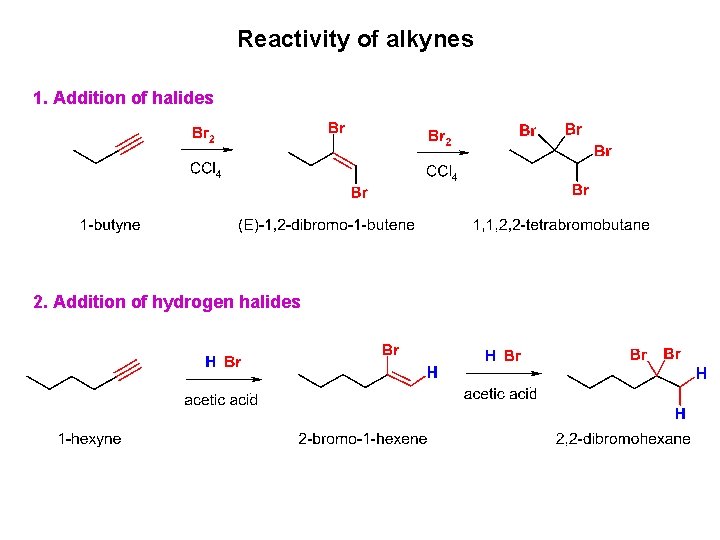
Reactivity of alkynes 1. Addition of halides 2. Addition of hydrogen halides
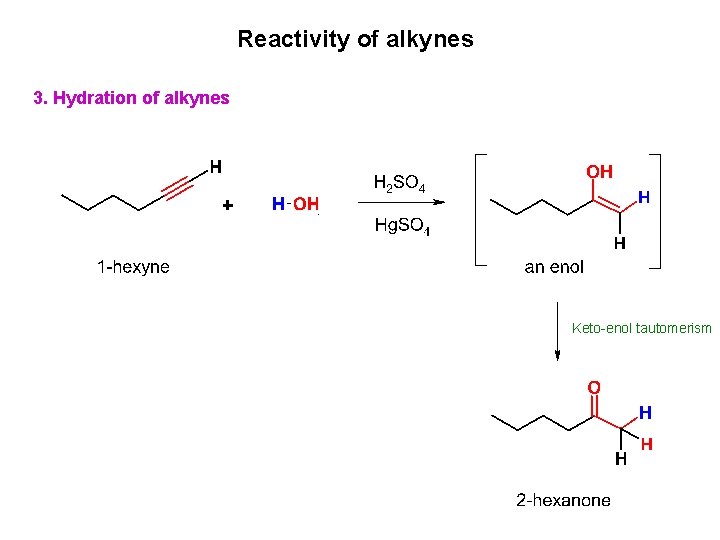
Reactivity of alkynes 3. Hydration of alkynes Keto-enol tautomerism

Reactivity of alkynes 4. Reduction of alkynes
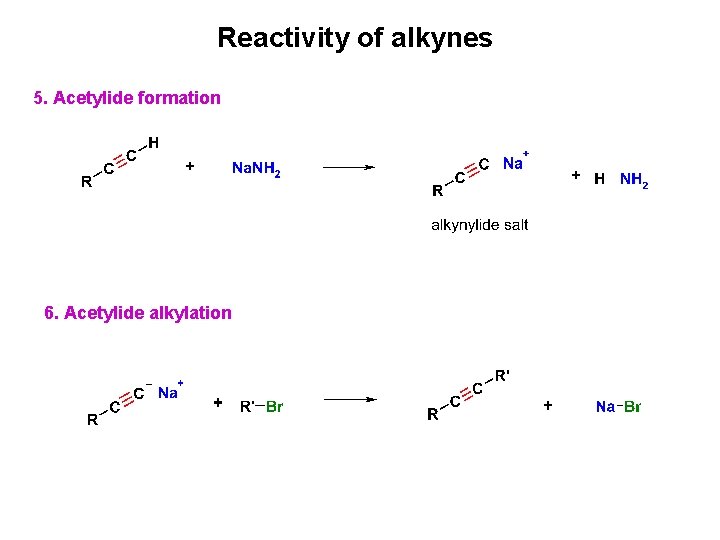
Reactivity of alkynes 5. Acetylide formation 6. Acetylide alkylation

Alkyne Acidity: Formation of Acetylide Anions • Terminal alkynes are weak Brønsted acids (alkenes and alkanes are much less acidic • Reaction of strong anhydrous bases with a terminal acetylene produces an acetylide ion • The sp-hybridization at carbon holds negative charge relatively close to the positive nucleus
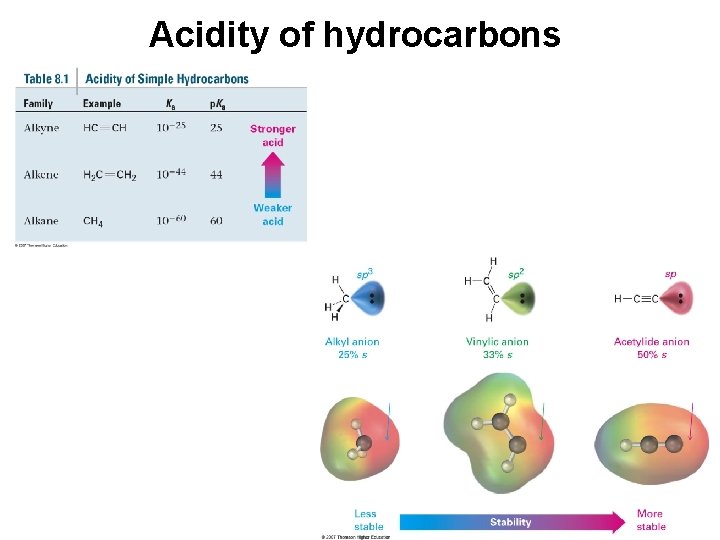
Acidity of hydrocarbons
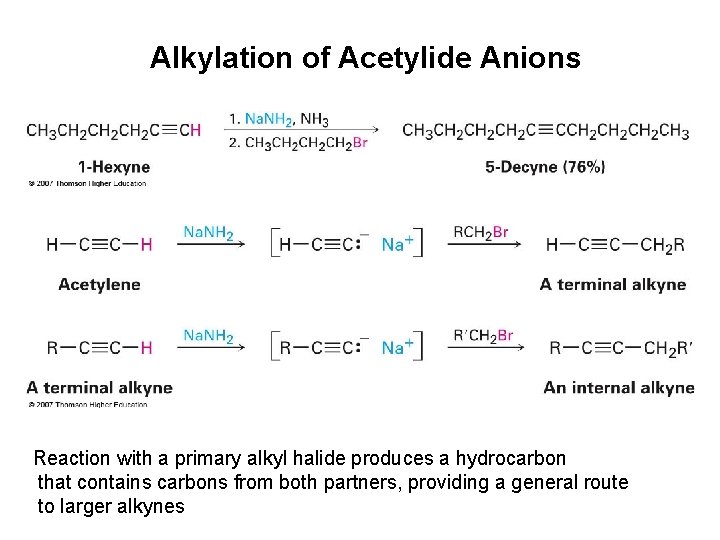
Alkylation of Acetylide Anions Reaction with a primary alkyl halide produces a hydrocarbon that contains carbons from both partners, providing a general route to larger alkynes
- Slides: 52