Alkenes Alkenes Low density Insoluble in water Non

Alkenes

Alkenes • • • Low density Insoluble in water Non polar Torsion due to double bond Reactive double bond

Alkenes • Aliphatic hydrocarbon molecules with at least one double bond • Unsaturated bonds: double bonds (can turn into single bonds by adding hydrogen)
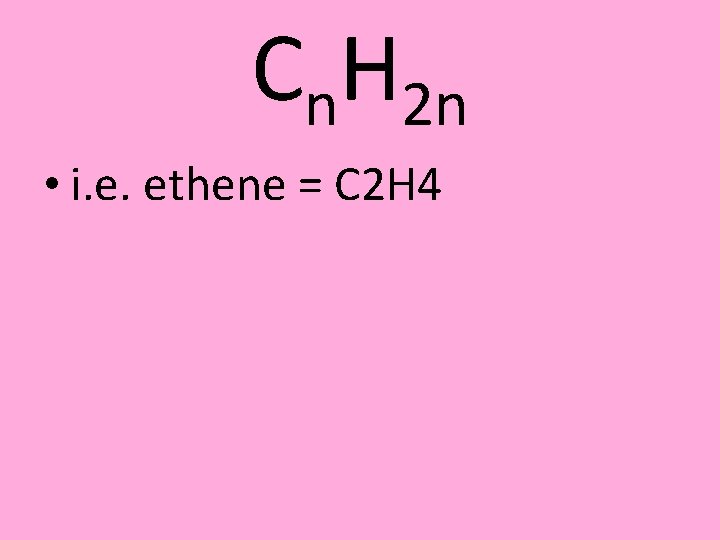
Cn. H 2 n • i. e. ethene = C 2 H 4

IUPAC Naming • Find the longest parent chain that includes the double bond • Change the ending of the parent to “ene”

• number the chain so that the double bond has the lowest number • if there is more than one double bond, use the Greek language prefixes to indicate quantity
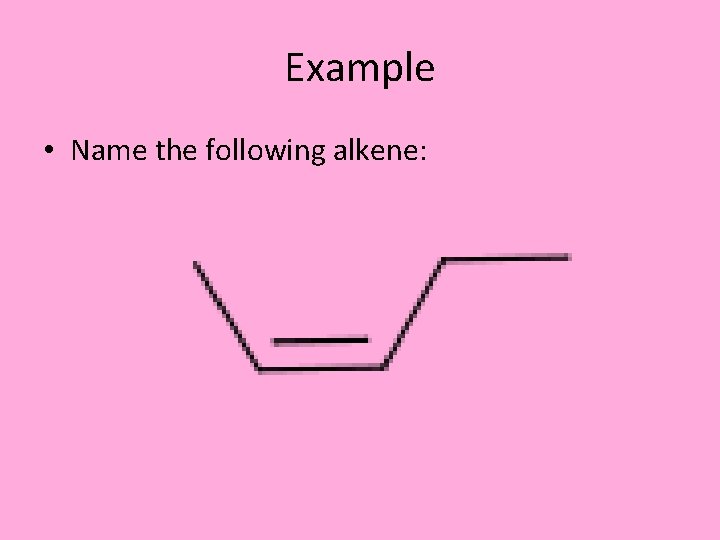
Example • Name the following alkene:

Example 2 • 2, 3 -dimethylnon-4 -ene

Cycloalkenes • Unsaturated aliphatic hydrocarbons (alicyclic) • Cn. H 2 n-2

Example • 1, 9 -diethyl cyclodec-1, 3, 5, 7 -tetra-ene

Alkynes

Alkynes • Aliphatic hydrocarbon molecules with at least one triple bond • The triple bond causes the alkyne to act differently than with a double or single bond
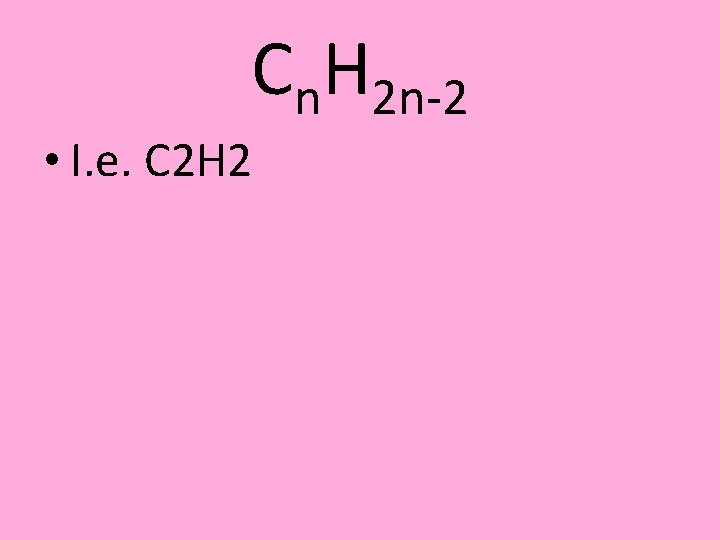
Cn. H 2 n-2 • I. e. C 2 H 2

IUPAC Naming • Find the longest parent chain that includes the triple bond • Change the ending of the parent to “yne”

• number the chain so that the triple bond has the lowest number • if there is more than one triple bond, use the Greek language prefixes to indicate quantity
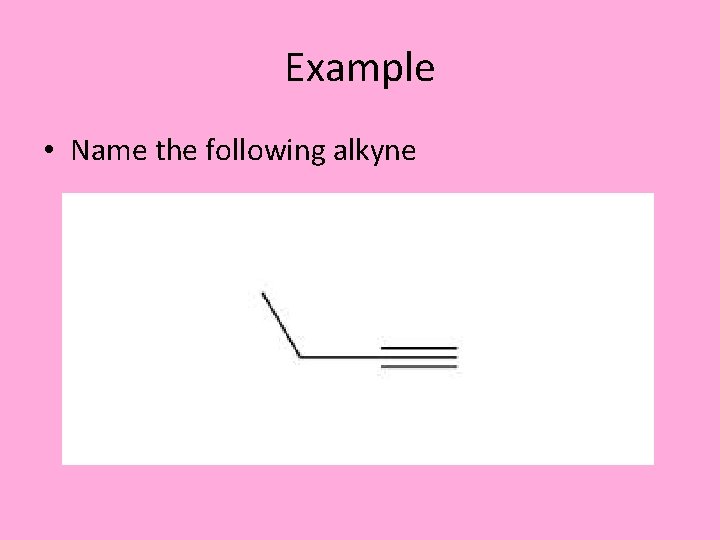
Example • Name the following alkyne
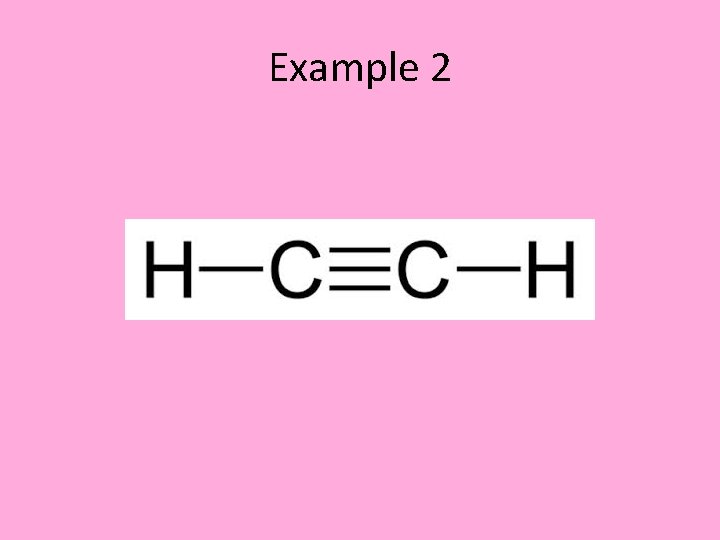
Example 2

Example 3 & 4 • 4 -methyl pent-2 -yne • 4 -pentyl hept-1, 6 -diyne - *** wrong naming** – 3 -pentyl hept-1, 6 -diyne
- Slides: 18