ALKENES Alkenes Cn H 2 n unsaturated hydrocarbons

ALKENES
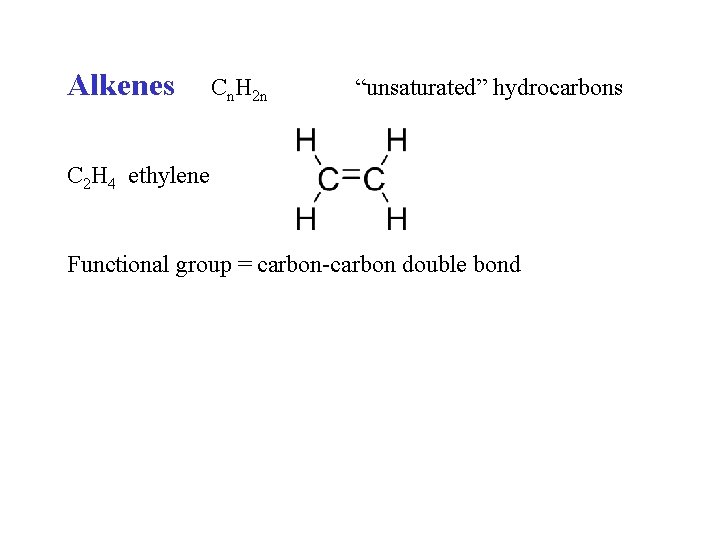
Alkenes Cn. H 2 n “unsaturated” hydrocarbons C 2 H 4 ethylene Functional group = carbon-carbon double bond
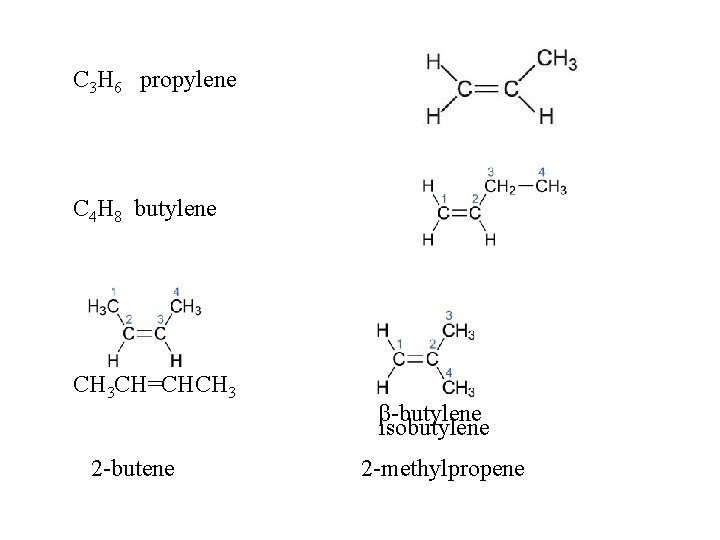
C 3 H 6 propylene C 4 H 8 butylene CH 3 CH=CHCH 3 2 -butene β-butylene isobutylene 2 -methylpropene
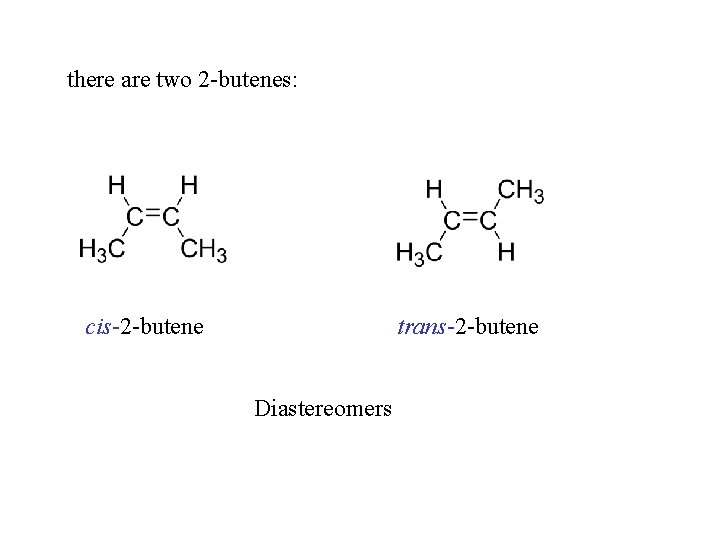
there are two 2 -butenes: cis-2 -butene trans-2 -butene Diastereomers

C=C are called “vinyl” carbons If either vinyl carbon is bonded to two equivalent groups, then no geometric isomerism exists. CH 3 CH=CHCH 3 yes (CH 3)2 C=CHCH 3 no CH 3 CH 2 CH=CH 2 no CH 3 CH=CCH 2 CH 3 yes
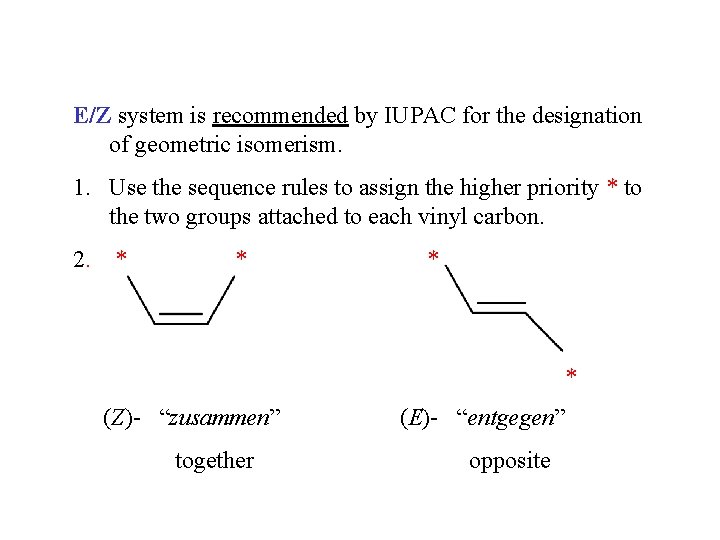
E/Z system is recommended by IUPAC for the designation of geometric isomerism. 1. Use the sequence rules to assign the higher priority * to the two groups attached to each vinyl carbon. 2. * * (Z)- “zusammen” together (E)- “entgegen” opposite
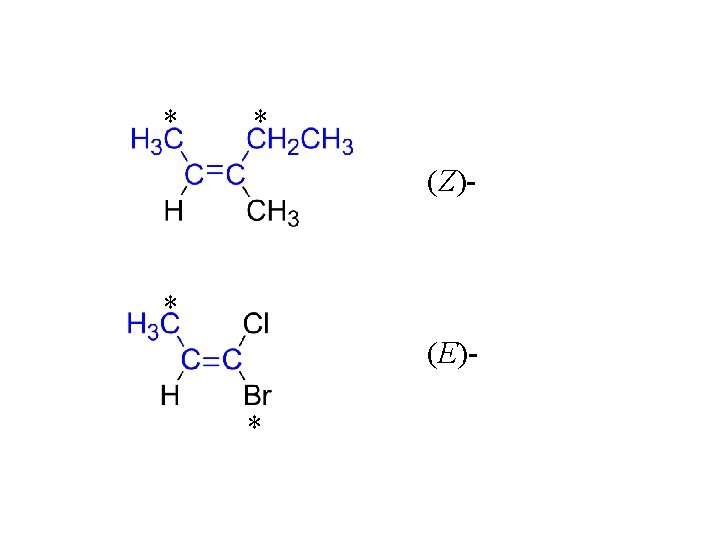

Nomenclature, alkenes: 1. Parent chain = longest continuous carbon chain that contains the C=C. alkane => change –ane to –ene prefix a locant for the carbon-carbon double bond using the principle of lower number. 2. Similar to alkanes 3. If a geometric isomer, use E/Z (or cis/trans) to indicate which isomer it is.
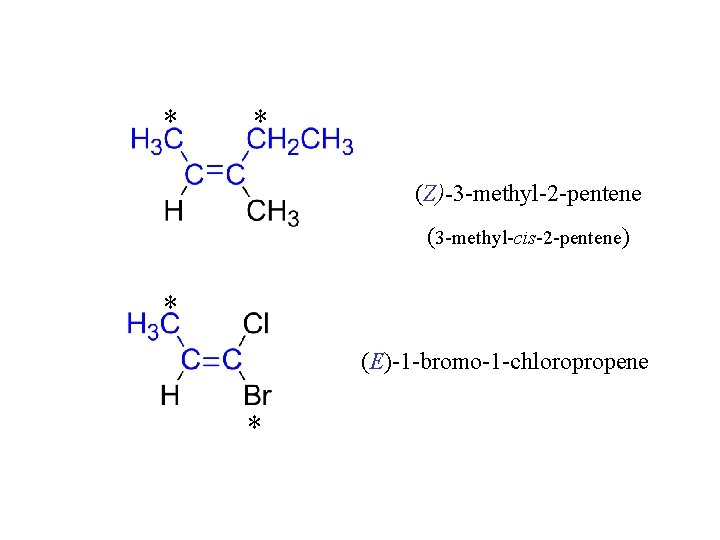
* * (Z)-3 -methyl-2 -pentene (3 -methyl-cis-2 -pentene) * (E)-1 -bromo-1 -chloropropene *
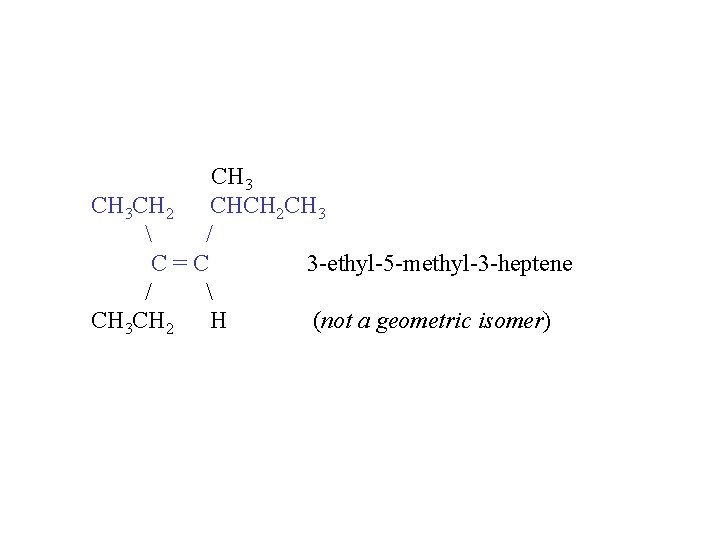
CH 3 CH 2 CHCH 2 CH 3 / C=C 3 -ethyl-5 -methyl-3 -heptene / CH 3 CH 2 H (not a geometric isomer)

-ol has the priority when naming CH 2=CHCH 2 -OH 2 -propen-1 -ol CH 3 CHCH=CH 2 OH 3 -buten-2 -ol

Physical properties: non-polar or weakly polar no hydrogen bonding low mp/bp similar to alkanes Insoluble in water Importance: common group in biological molecules

Preparation of alkenes: 1. dehydrohalogenation of alkyl halides 2. dehydration of alcohols 3. dehalogenation of vicinal dihalide
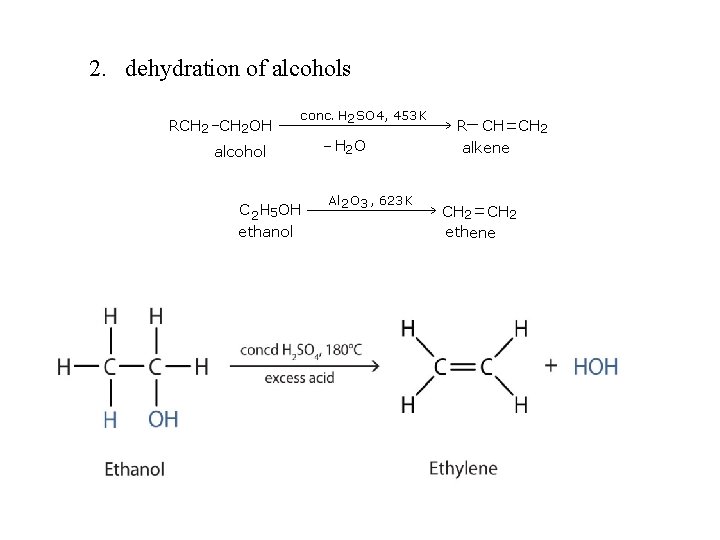
2. dehydration of alcohols
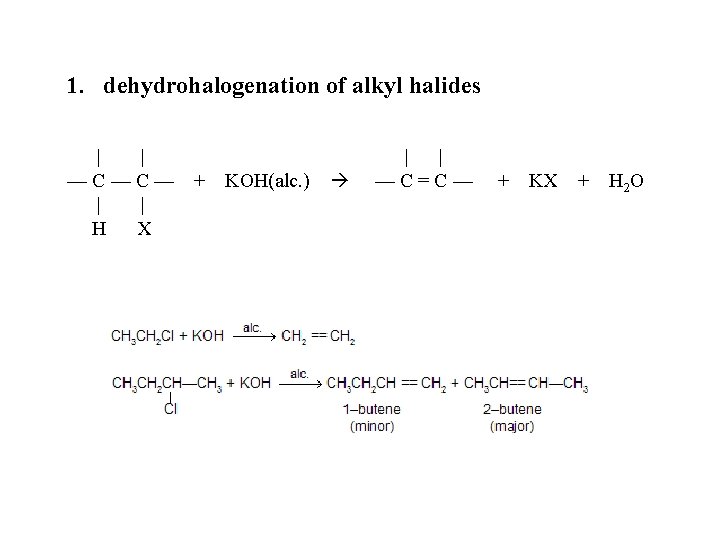
1. dehydrohalogenation of alkyl halides | | —C—C— | | H X + KOH(alc. ) | | —C=C— + KX + H 2 O
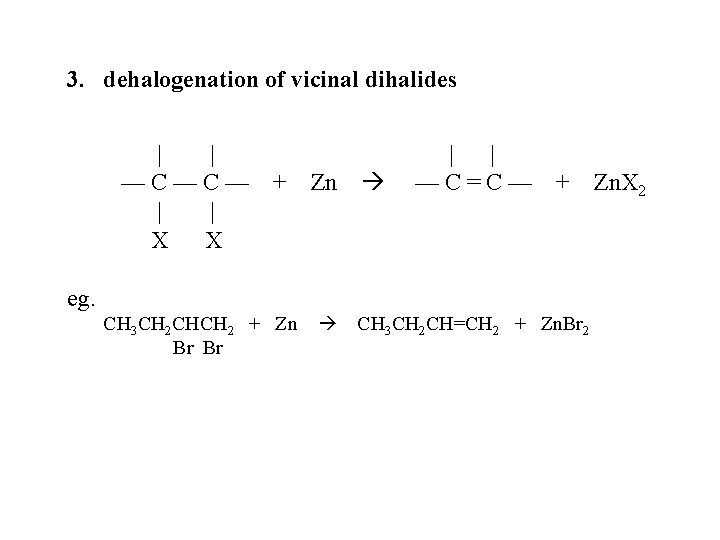
3. dehalogenation of vicinal dihalides | | —C—C— | | X X + Zn | | —C=C— + Zn. X 2 eg. CH 3 CH 2 CHCH 2 + Zn Br Br CH 3 CH 2 CH=CH 2 + Zn. Br 2
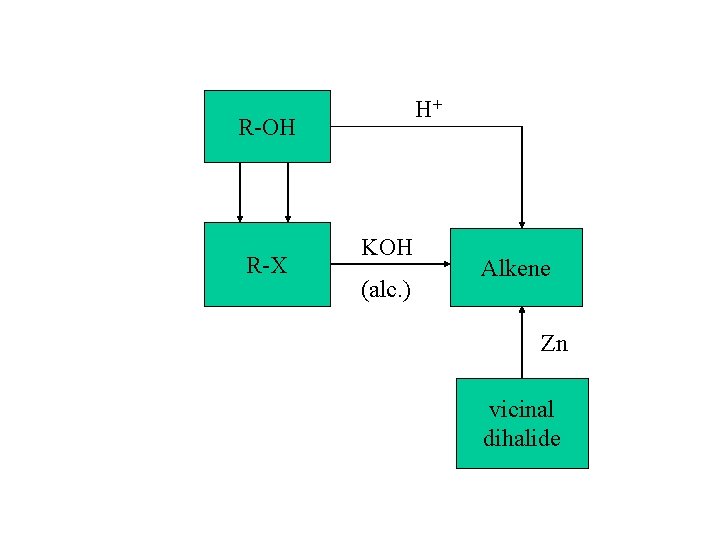
H+ R-OH R-X KOH (alc. ) Alkene Zn vicinal dihalide
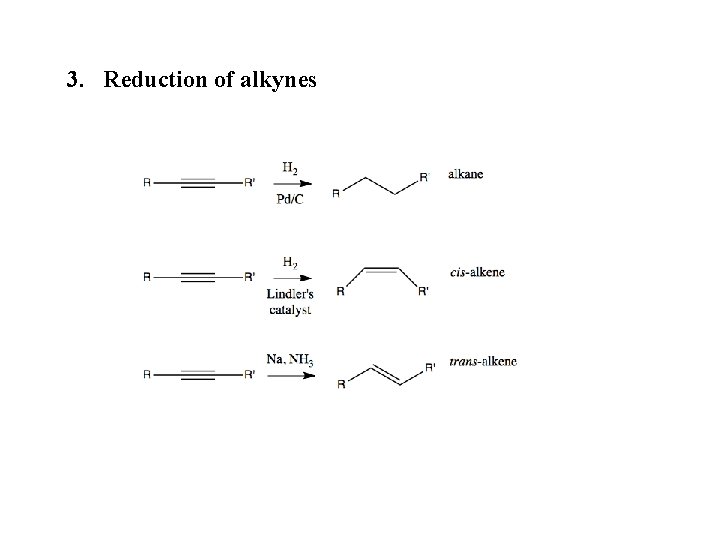
3. Reduction of alkynes
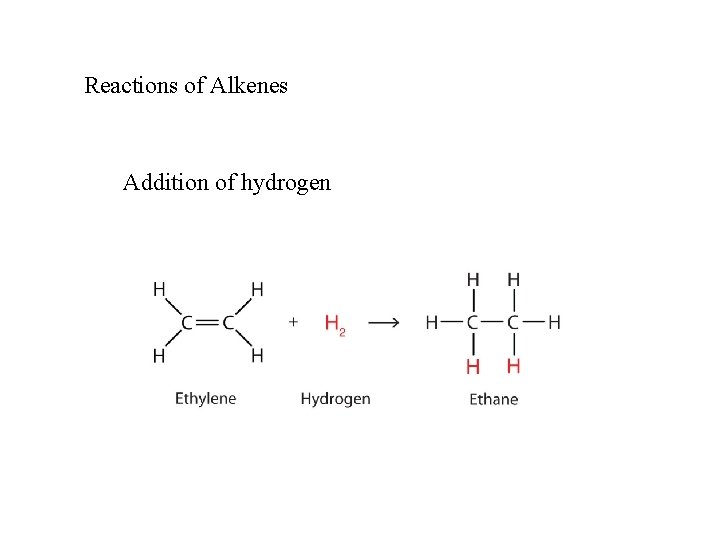
Reactions of Alkenes Addition of hydrogen
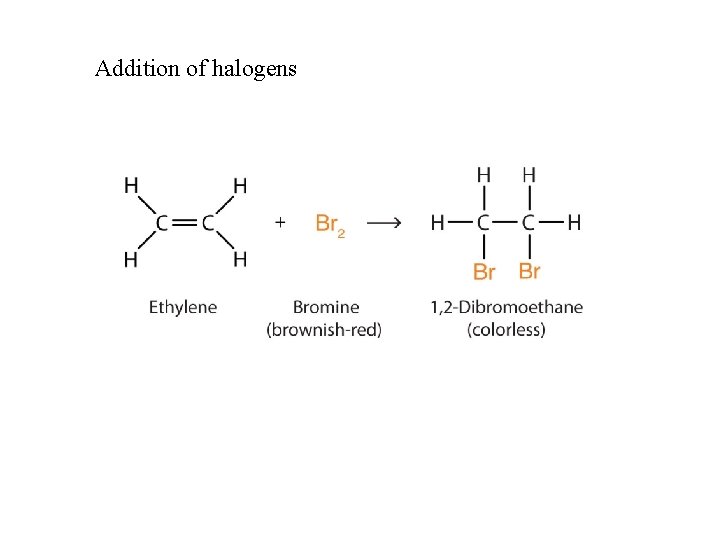
Addition of halogens
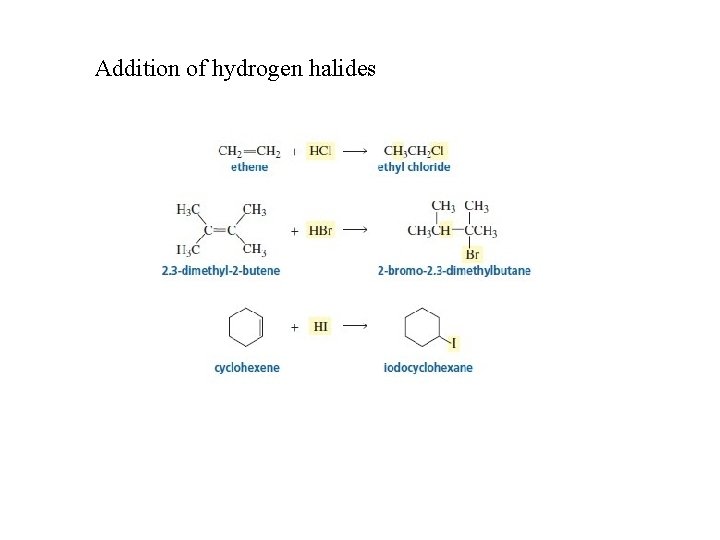
Addition of hydrogen halides
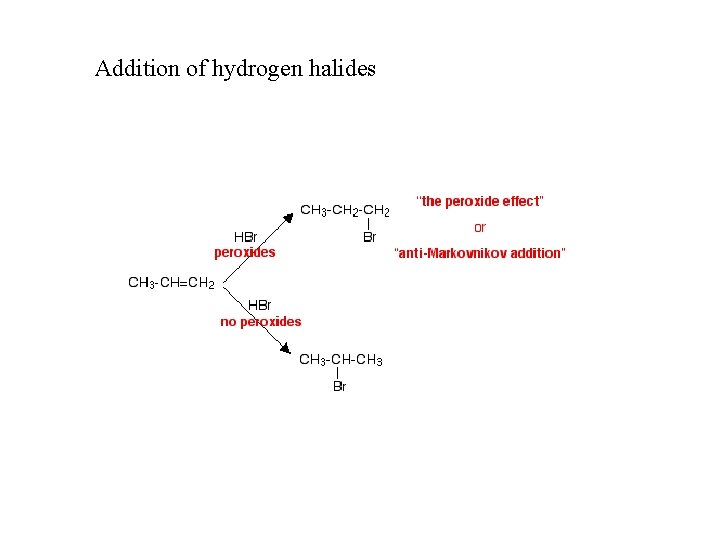
Addition of hydrogen halides
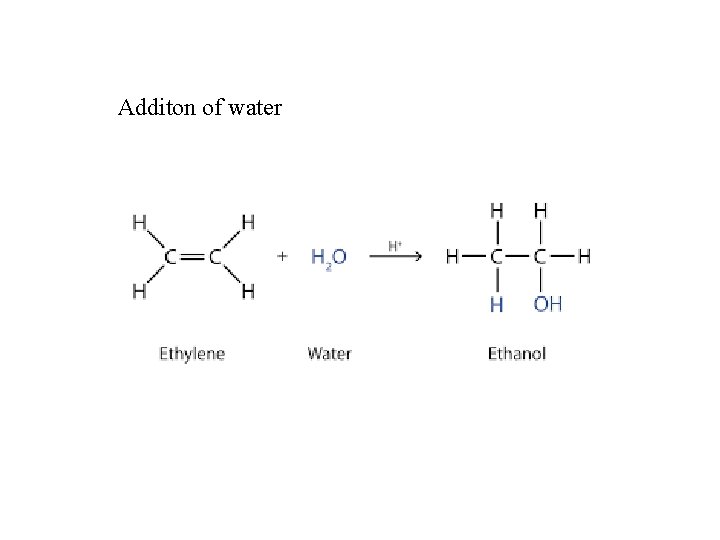
Additon of water
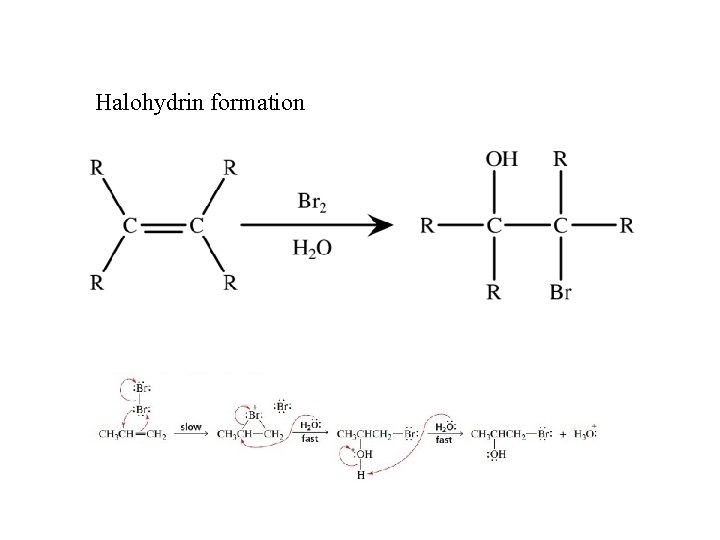
Halohydrin formation
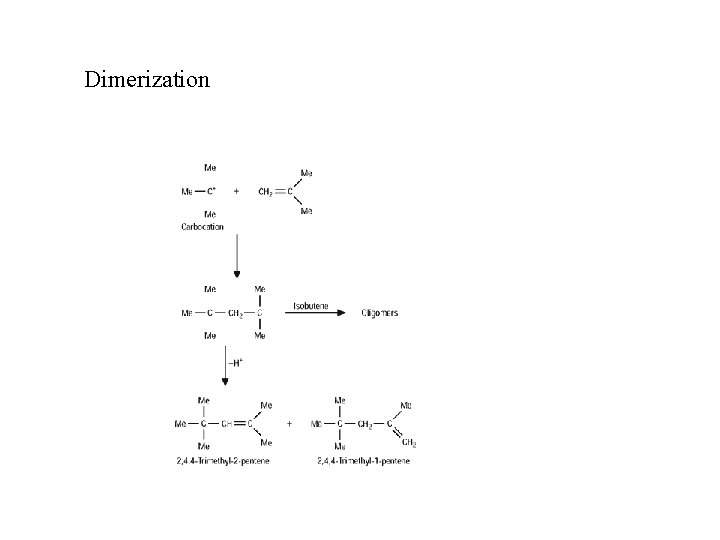
Dimerization
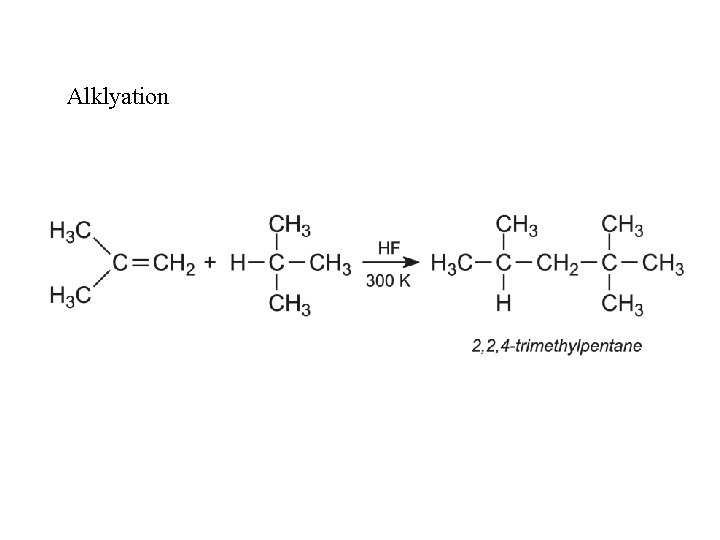
Alklyation

Oxymercuration-Demercuration
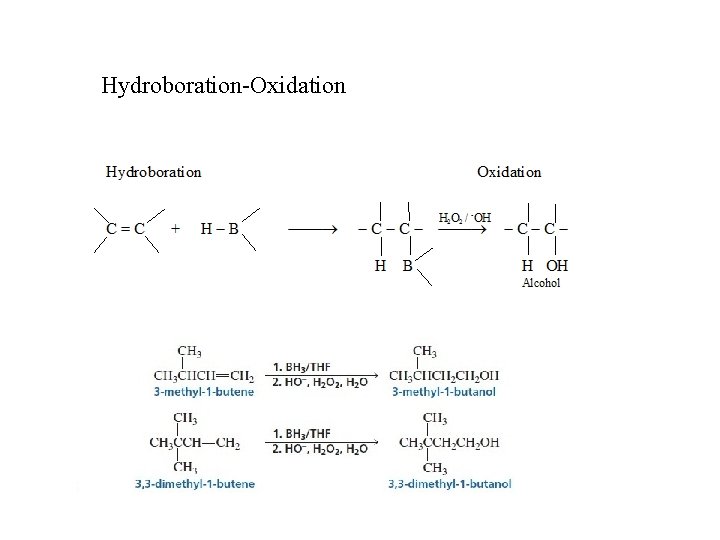
Hydroboration-Oxidation
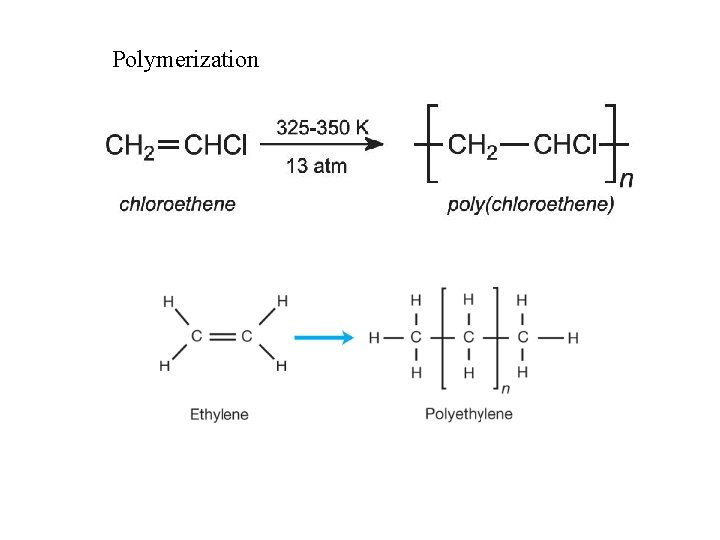
Polymerization
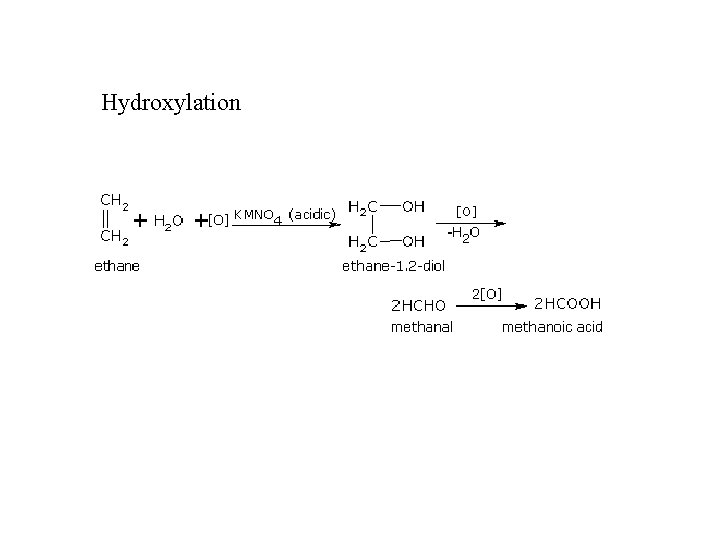
Hydroxylation
- Slides: 30