Scientific Measurement Chemistry Chapter 3 Measurements Are really











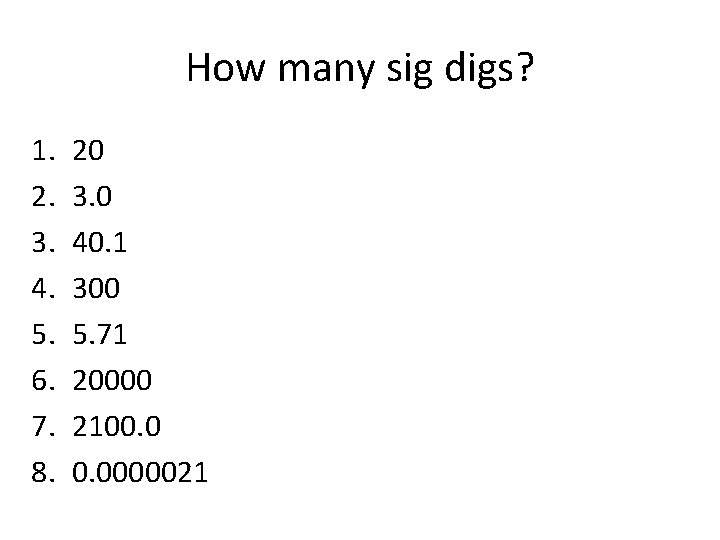
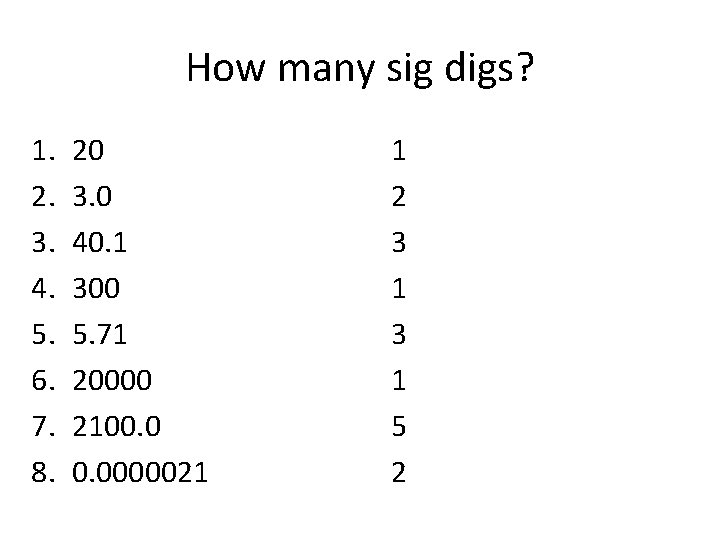



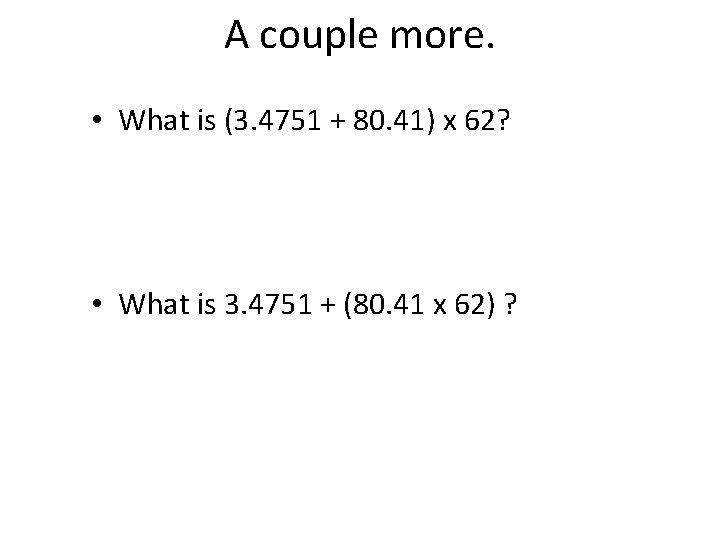








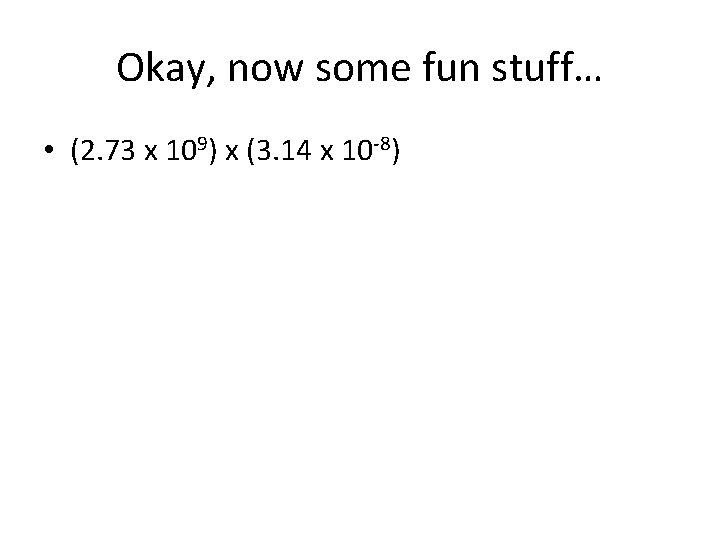




















































- Slides: 83

Scientific Measurement Chemistry Chapter 3

Measurements • Are really, really important to sciences. • Important to be able to decide if measurements are correct. • Must have two parts: – A quantity – A unit • UNIT ARE VERY, VERY IMPORTANT!

• http: //www. youtube. com/watch? v=h. Qp. Q 0 hx VNTg

Scientific Notation • Science deals with very large and very small numbers • Like: – 60200000000000 atoms – 0. 00000000250 m • But zeroes are boring and are a waste of space. • So, we use scientific notation – 1 number in front of the decimal, and all other important (significant) numbers behind the decimal – 6. 02 x 1023 – 2. 50 x 10 -17

• http: //www. youtube. com/watch? v=0 l. FQOmb 6 m. Vs

Get the meter stick, Valz!

Significant Figures (or Digits) • How well you “know” and estimate your measurement – All the digits you are confident in, plus one that is estimated • Measurements must always be reported to the correct number of significant digits. – Answers depend on the correct number of digits to determine the most “honest” answer.

Counting significant digits 1. All zeroes before the first non-zero don’t count. • 0. 0000002 only has 1 sig digs 2. All non-zero numbers count. • 21343 has 5 sig digs 3. All zeroes between non-zeroes count • 2002 has 4 sig digs 4. All zeroes AFTER non-zeroes count IF there is a decimal • • • 200. 0 has 4 sig digs 200. has 3 sig digs (decimal at the end!) 200 has 1 sig digs

A simpler way of saying it. • The zeroes in the front NEVER count. • The zeroes at the end ONLY count if there is a decimal. • That’s it.

The Pacific/Atlantic Method • If the period is present (with a p) you start in the Pacific Ocean, on the left side (at the first nonzero), and count until you’re out of numbers. • If the period is absent (with an a) you start in the Atlantic, on the right side and don’t start counting until you hit the first non-zero.

Get some, scratch paper, yo. Number it 1 to 8.
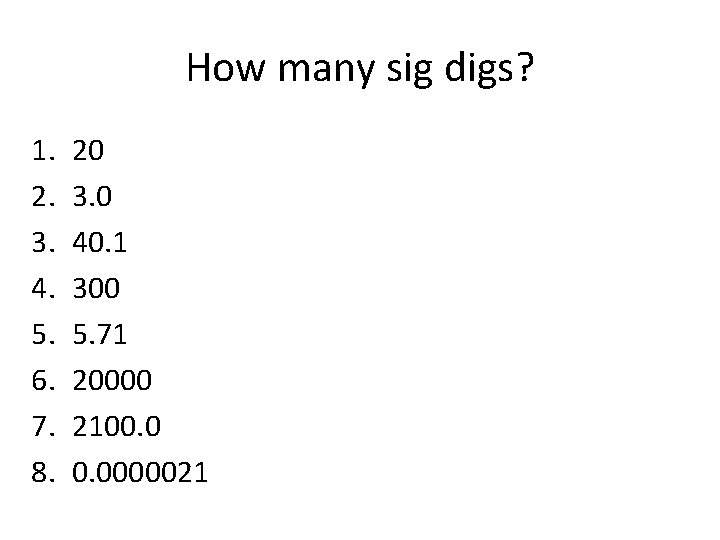
How many sig digs? 1. 2. 3. 4. 5. 6. 7. 8. 20 3. 0 40. 1 300 5. 71 20000 2100. 0000021
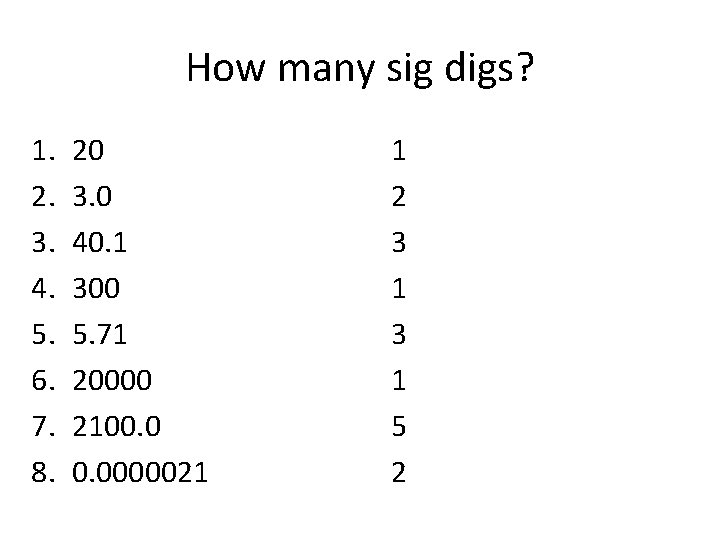
How many sig digs? 1. 2. 3. 4. 5. 6. 7. 8. 20 3. 0 40. 1 300 5. 71 20000 2100. 0000021 1 2 3 1 5 2

Rules for Math with Sig. Digs. • Multiply or Divide: – Use the LEAST number of significant digits from your measurements. • Add or Subtract – Use the LEAST significant place value (which has the highest place value). • When both: – Use whichever method gives you the least number of significant digits as the answer • When averaging: – The number of numbers is NOT a measurement, and therefore does NOT count when determining the number of significant digits.

WHY? !? • When adding or subtracting numbers, they must be from the same type of measurement. That means that the units must be the same, so the place value must be the same. • When multiplying or dividing you are changing the units or converting, so it’s just the number of significant digits that matter. – Place VALUE doesn’t matter when converting • Don’t write this down.

Let’s try some… • What is 3. 3/1. 55 in correct significant digits? • What is 3. 161 – 0. 85 in correct significant digits?
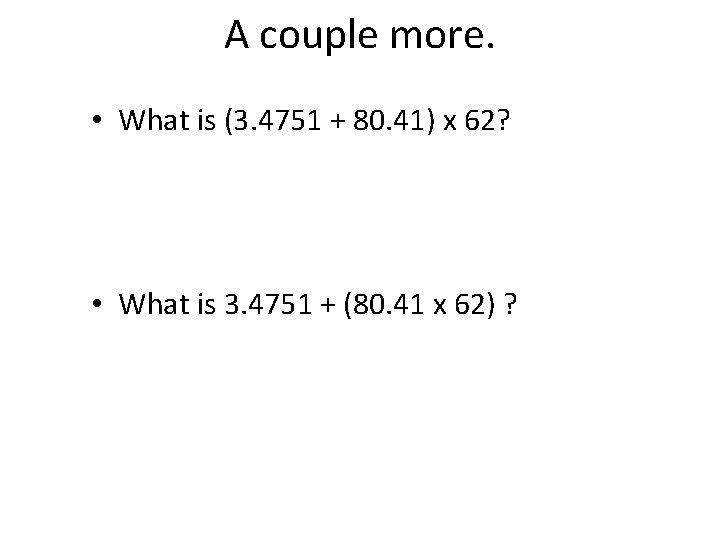
A couple more. • What is (3. 4751 + 80. 41) x 62? • What is 3. 4751 + (80. 41 x 62) ?

Back to scientific notation really quick…

Let’s try some out… • First, let’s convert 325100000 to scientific notation.

Let’s try some out… • First, let’s convert 325100000 to scientific notation. – 3. 251 x 108

Okay, how about this… • Convert 4. 78 x 10 -5 to standard notation.

Okay, how about this… • Convert 4. 78 x 10 -5 to standard notation. – 0. 0000478

Fine, do this one, smart guy. • Fix this number to proper scientific notation: 5541 x 105

Fine, do this one, smart guy. • Fix this number to proper scientific notation: 5541 x 105 – 5. 541 x 108

Doing maff with Scientific Notation • Addition/subtraction – Change the exponent on one of the numbers until they are the same – Then line up the decimals and subtract/add – Adjust the coefficients until there is again 1 digit in front of the decimal • Multiplication/division – Divide or multiply the coefficients – Divide or multiply the base 10 exponents – Adjust the coefficients again • YOU STILL FOLLOW THE RULES OF SIGNIFICANT DIGITS!!!1 ONE!1
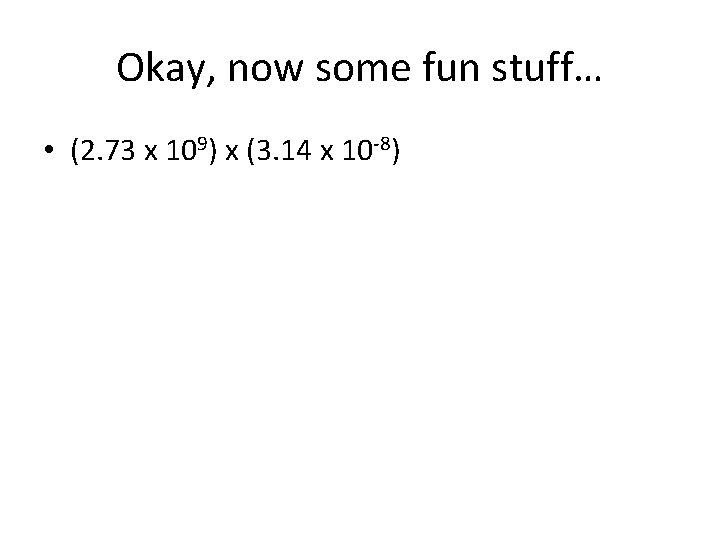
Okay, now some fun stuff… • (2. 73 x 109) x (3. 14 x 10 -8)

Okay, now some fun stuff… • (2. 73 x 109) x (3. 14 x 10 -8) – 8. 5722 x 101

Okay, now addition. • (2. 73 x 109) + (3. 14 x 108)

Okay, now addition. • (2. 73 x 109) + (3. 14 x 108) – 3. 044 x 109

Accuracy and Precision • Accuracy – How close the measurement is to the actual (true) value • Closeness to the “target” – Measured by ERROR. • Precision – How close measurements are to each other • Closeness to other measures of the same thing – Measured by the number of significant digits.

• http: //www. youtube. com/watch? v=Tz. Ln. O 04 u O 30 • http: //www. youtube. com/watch? v=q. PC 1 Km. D Xeg. Y

Determining Error • Error is the difference between a measured (experimental) value and the accepted value – Error = experimental value – accepted value – % error = |Error|*100/accepted value

Practice • You measure the world’s largest duck to be 15 m long, but it is known to actually be 14 m long. – What is the error? – What is the percent error?

Practice • You measure the world’s largest duck to be 15 m long, but it is known to actually be 14 m long. – What is the error? • 15 -14 = 1 m – What is the percent error? • • |15 -14|*100/14 7. 1 % = 7% (with sig figs)

Okay, now your turn… • You have a 17. 1 kg sample of lead. It should displace 1. 508 liters of water. But when you measure it, it displaces exactly 1. 50 liters. What is your error and percent error?

3. 1 Quiz 1. Convert 3. 2 x 104 to standard notation. 2. Convert 0. 00079 to scientific notation. 3. Put 0. 000083 x 10 -4 in proper scientific notation. 4. How many significant digits are in 3, 000? 5. Solve and answer in correct significant digits: 3. 421 x 0. 411 + 9. 62

3. 1 Quiz answers 1. 32000 -4 2. 7. 9 x 10 3. 8. 3 x 10 -9 4. 1 5. 11. 0

SI: International System of Units (System International) • The basis for all measurements made in science (and in this class) • Uses 5 most commonly used units: • Meter (distance) • Kilogram (mass) • Kelvin (temperature) • Second (time) • Mole (amount) • All measurements can be reported as some SI measurement.

Prefixes in SI • SI uses prefixes to shorten measurements (much like scientific notation) • These are put in front of measurements to give a certain place value (multiple) of the unit. – Ex) Rather than 1000 m, you can write 1 km, because kilo means 1000. – Ex) Rather than writing 0. 000001 g, you can write 1 μg or 1 microgram, because micro means 10 -6


Important SI measurements in Chemistry • Length – Meter, km, cm, mm, nm • Volume – Liter, m. L, μL – Cubic meter (m 3), cubic cm (cm 3) – 1 cm 3 = 1 m. L • Mass – Gram, kg, mg, μg – NOT the same as WEIGHT!!!! • Temperature – Celsius • Based on boiling and melting point of water – Kelvin • Absolute, based on the point at which matter stops moving • Energy – Joules, k. J, calories (stupid, based on water, don’t use)

Volume’s two “sets of units” • Volume is 3 measures of length multiplied together. – m, km, cm, or mm 3 times • m 3, km 3, cm 3, or mm 3 • Volume can also be measure in terms of liters. – 1000 L = 1 m 3 – 1 L = 1000 cm 3 – 1 m. L = 1 cm 3 • This is the one that you NEED to know.

How to convert cubic units to liter units • Convert your original number to m. L or cm 3. • Then it is a 1 to 1 conversion (same number, just change the unit) • Then convert to the unit you want. • Ex) 40 L to mm 3 – 40 L = 40, 000 m. L – 40, 000 m. L = 40, 000 cm 3 – 1 cm = 10 mm • 1 cm = 103 mm 3 = 1000 mm 3 – 40, 000 cm 3 = 40, 000 mm 3

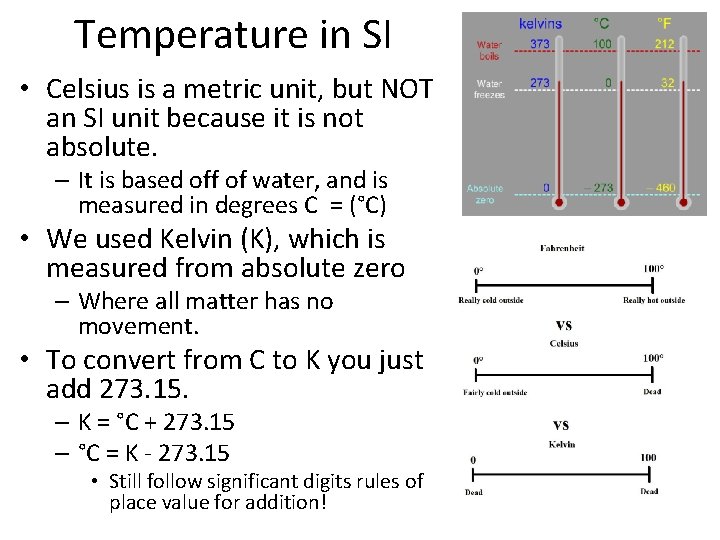
Temperature in SI • Celsius is a metric unit, but NOT an SI unit because it is not absolute. – It is based off of water, and is measured in degrees C = (°C) • We used Kelvin (K), which is measured from absolute zero – Where all matter has no movement. • To convert from C to K you just add 273. 15. – K = °C + 273. 15 – °C = K - 273. 15 • Still follow significant digits rules of place value for addition!

A problem? You’re the problem! • Convert 27°C to Kelvin.

A problem? You’re the problem! • Convert 27°C to Kelvin. – 273. 15 + 27 = 300. 15 K • 300. K with correct SD

A problem? You’re the problem! • Convert 27°C to Kelvin. – 273. 15 + 27 = 300. 15 K • 300. K with correct SD • Convert 395. 17 K to °C

A problem? You’re the problem! • Convert 27°C to Kelvin. – 273. 15 + 27 = 300. 15 K • 300. K with correct SD • Convert 395. 17 K to °C – 395. 17 – 273. 15 = 122. 02°C

Don’t worry about the calorie to Joule conversion. • Calories are a stupid unit of measurement. – If I want you to do a stupid conversion, I’ll give you the conversion and the units.

3. 2 Quiz 1. What is the SI unit of length? 2. What is the name of the “before” measurement that is used to shorten SI measurements. – What is the “k” in “km” called (or the “c” in “cm”)? 3. What is the SI prefix for 1000? 4. What is larger m. L or mm 3? 5. Convert 95°C to K, using correct significant digits.

3. 2 Quiz 1. 2. 3. 4. 5. Meter (m) prefix Kilo m. L 368 K

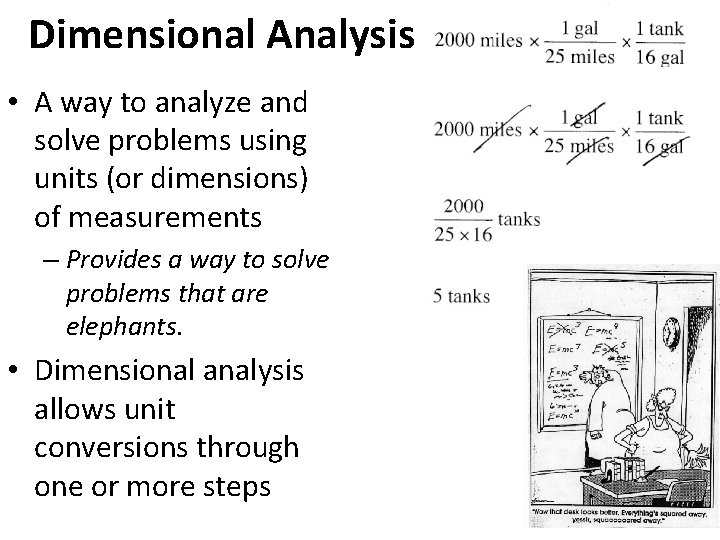
Dimensional Analysis • Also called “factor-label conversions” • Uses conversion factors to convert one measurement to an equal one using different units • Conversion factors are a ratio of two EQUAL measurements with different numbers – Ex) 1 mile = 1760 yards • Therefore 1 mi =1 1760 yards This is a stack of pennies. It has nothing to do with dimensional analysis, but when I Googled “dimensional analysis” this image came up, and it’s pretty awesome.

Conversion Factors • Conversion factors must always be equal to one – Will not change the actual value when multiplied by a measurement – Only changes the unit label to give you a different “looking” measurement • But nothing really changes

Another example problem. • Convert 1600 km/hr (the airspeed of a fighter jet) to m/s.

Another example problem. • Convert 1600 km/hr (the airspeed of a fighter jet) to m/s. – 444. 44… m/s – Rounds to 440 m/s with sig. figs.

• https: //youtu. be/h. Qp. Q 0 hx. VNTg? t=3 m 7 s

Dimensional Analysis • A way to analyze and solve problems using units (or dimensions) of measurements – Provides a way to solve problems that are elephants. • Dimensional analysis allows unit conversions through one or more steps

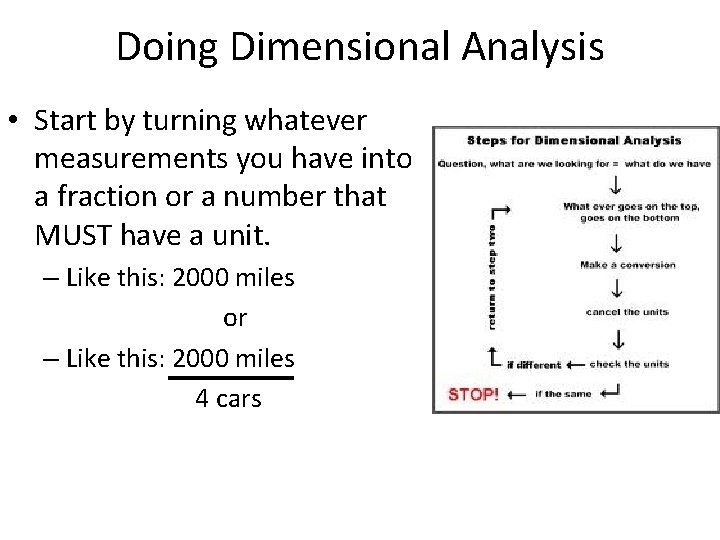
Doing Dimensional Analysis • Start by turning whatever measurements you have into a fraction or a number that MUST have a unit. – Like this: 2000 miles or – Like this: 2000 miles 4 cars

Completing Dimensional Analysis • Multiply by conversion factors until you have an answer in the units you want • Okay, lets do this: • Convert 1. 4 miles into km. – 1. 62 km/1 mi

Completing Dimensional Analysis • Multiply by conversion factors until you have an answer in the units you want • Okay, lets do this: • Convert 1. 4 miles into km. – 1. 62 km/1 mi • 2. 268 km. – But only 2 sig figs, so 2. 3 km, right?

When sig digs matter. • Significant Digits only matter in MEASUREMENTS (which are estimates), not for conversions (which are definite). – This doesn’t count for converting from SI to non-SI, though.

Now the BIG problem. • The sink in the fume hood has been dripping since 1999. How much water in L has gone the drain since then?

On your own now… • I have 3. 12 x 103 papers to grade – I can grade 3 papers per hour – I can grade 8 hours per day – How many days will it take to grade them? – Present your answer in standard notation, with the correct number of significant digits.

Converting in SI • 2 methods of conversion – Dimensional Analysis method • Difficult, slow. – Lazy Mr. V method • Easy, quick. • DOES NOT WORK WITH NON-SI CONVERSIONS!!!

Practice Problems • Convert 1. 75 m into cm

Practice Problems • Convert 1. 75 m into cm – 0 – (-2) = 2 – Move decimal 2 places to the RIGHT.

Practice Problems • Convert 1. 75 m into cm – 0 – (-2) = 2 – Move decimal 2 places to the RIGHT. • Answer: 175 cm

On your own • Convert 678µg to kg.

On your own • Convert 678µg to kg. – µ = -6 to kg = 3 – -6 – 3 = -9 – 9 spots to the LEFT – 0. 000000678

Okay, how about this one? • 3 cm 3 to mm 3 – If you have cubic units, you must do the conversion 3 times. – (IF you have square units, you must do the conversion 2 times. )

Okay, how about this one? • 3 cm 3 to mm 3 – If you have cubic units, you must do the conversion 3 times. – (IF you have square units, you must do the conversion 2 times. ) – cm = -2 – mm= -3 – -2 – (-3) = 1 – 1 * 3 = 3, so 3 spots to the right – 3 cm 3 = 3000 mm 3

Try this one on for size: • Convert 3. 71 cm 3 to m 3. – Remember, it’s an elephant, but you can just eat it one bite at a time!

3. 3 Quiz 1. Convert 80. 0 k. L to m. L, and put in correct scientific notation with correct significant digits. 2. Convert 211 m/s into km/hr. – Answer in correct sig figs. 3. A conversion factor is always equal to (or the same as) what number? 4. T/F: A conversion factor’s significant digits matter when determining the final answer’s number of significant digits. – Assume that the conversion is an absolute, and not just a measured value. 5. Convert 30. 0 miles per gallon to km per liter if 1 mi=1. 61 km, and 3. 78541 L=1 gallon

3. 3 Quiz Answers 1. 8. 00 x 107 m. L 2. 760. km/hr 3. 1 4. F 5. 12. 8 km/L

Density • Density is an easily measured intensive property. – Does not matter how much matter you have. • Density is a ratio of mass divided by volume. • How much “stuff” fits in a certain space. • Density range: – Hydrogen (0. 000084 g/m. L) – Water (1. 000 g/m. L) – Osmium (22. 57 g/m. L)

Determining Density • Density = mass volume • Okay, so let’s determine the density of an object with a mass of 37. 2 g, that requires a volume of 4. 5 m. L.

Determining Density • Density = mass volume = 37. 2 g = 8. 3 g/m. L 4. 5 m. L • Okay, so let’s determine the density of an object with a mass of 37. 2 g, that requires a volume of 4. 5 m. L.

So, what is it? • Density is 7. 9 g/m. L, rounded to 2 significant digits. Which metal is it? – Are you SURE? Density (g/m. L)

Density used to calculate mass or volume • If you KNOW the material you can determine the mass or volume. – If D = m/V – What is m? – What is V? • If lead has a density of 11. 34 g/m. L, and you need 100. 0 g of it, how much space will it occupy?

Determining Mass from Measurements • I have a fish aquarium with a height of 20. 0 cm, a width of 13. 0 cm, and a length of 16. 5 cm. If I fill it all the way up with water at 4°C (D = 1. 00 g/m. L) what will be the mass of the aquarium?

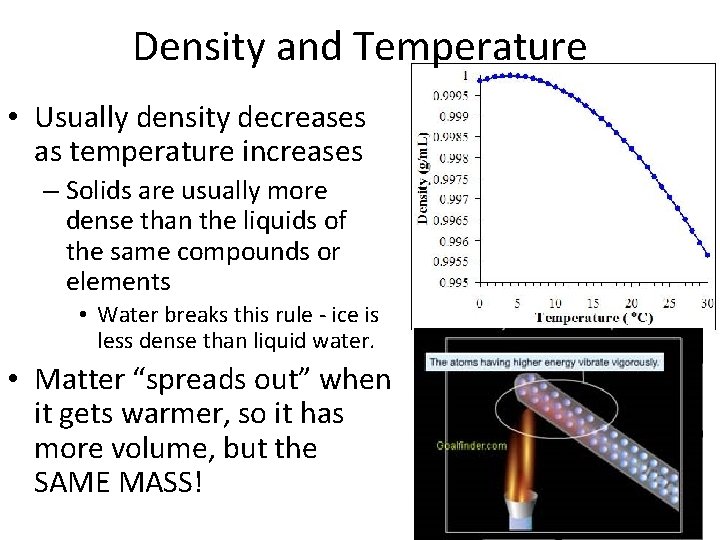
Density and Temperature • Usually density decreases as temperature increases – Solids are usually more dense than the liquids of the same compounds or elements • Water breaks this rule - ice is less dense than liquid water. • Matter “spreads out” when it gets warmer, so it has more volume, but the SAME MASS!

3. 4 Quiz 1. Write the equation for calculating density. 2. When temperature decreases, density usually _______. 3. Write an equation to determine the mass of an object based on its density and volume. 4. If an object has a mass of 36 g, and a volume of 6. 0 m. L, what is its density in g/m. L? 5. If an object has a mass of 3. 00 g, and a density of 39. 30 g/m 3, what is its volume?

3. 4 Quiz 1. D = m/V 2. increases 3. m=Dx. V 4. 6. 0 g/m. L 3 5. 0. 0763 m