Chapter Two Measurements In Chemistry Measurements in Chemistry









- Slides: 19

Chapter Two Measurements In Chemistry

Measurements in Chemistry → CO 2. 1 Measurements can never be exact; there is always some uncertainty. © Richard Hamilton Smith/Corbis Outline Copyright © Houghton Mifflin Company. All rights reserved. 2

Measurements in Chemistry cont’d ← Fig. 2. 1 Metric system units are becoming increasingly evident on highway signs. David Frazier/Photo Researchers Copyright © Houghton Mifflin Company. All rights reserved. 3

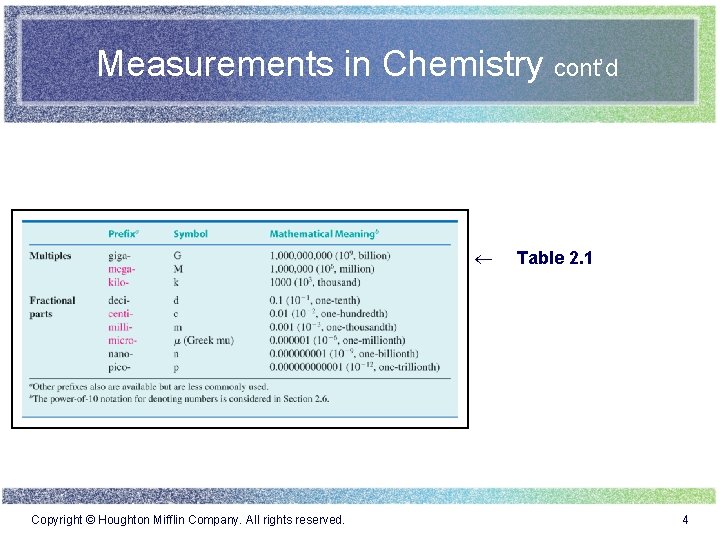
Measurements in Chemistry cont’d ¬ Copyright © Houghton Mifflin Company. All rights reserved. Table 2. 1 4

Measurements in Chemistry cont’d → Fig. 2. 2 Comparisons of the base metric system units of length, mass, and volume with common objects. E. R. Degginger Copyright © Houghton Mifflin Company. All rights reserved. 5

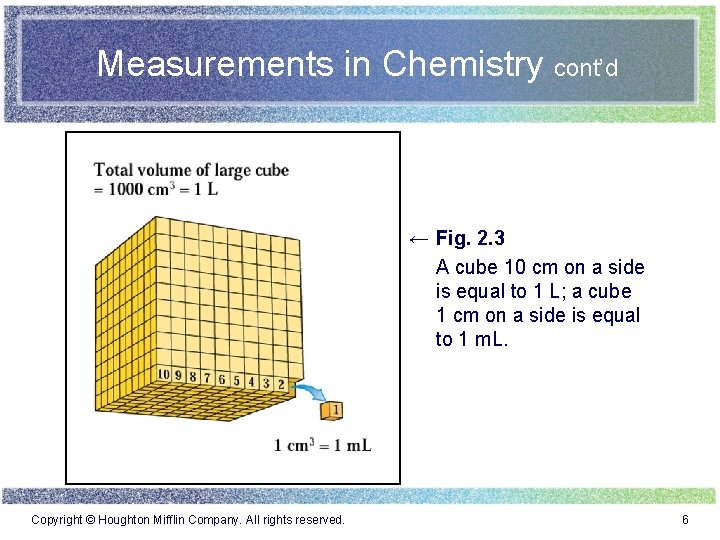
Measurements in Chemistry cont’d ← Fig. 2. 3 A cube 10 cm on a side is equal to 1 L; a cube 1 cm on a side is equal to 1 m. L. Copyright © Houghton Mifflin Company. All rights reserved. 6

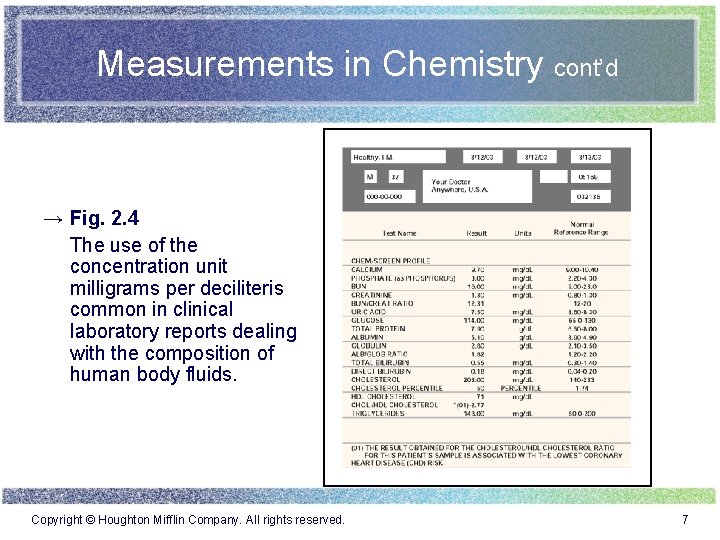
Measurements in Chemistry cont’d → Fig. 2. 4 The use of the concentration unit milligrams per deciliteris common in clinical laboratory reports dealing with the composition of human body fluids. Copyright © Houghton Mifflin Company. All rights reserved. 7

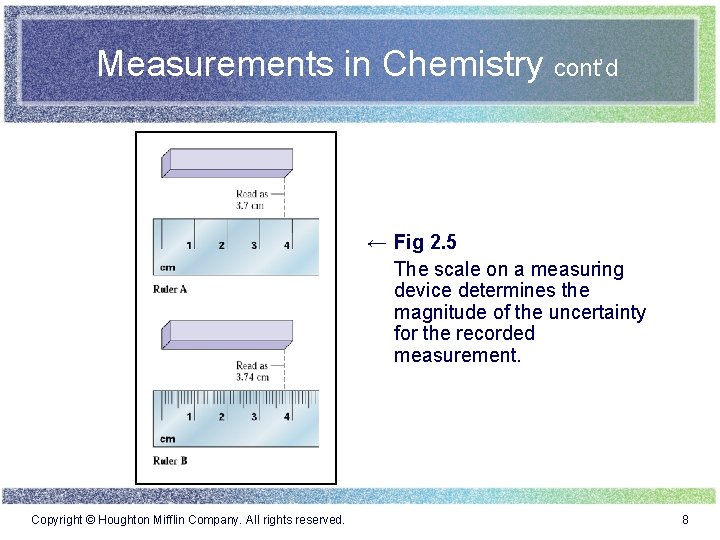
Measurements in Chemistry cont’d ← Fig 2. 5 The scale on a measuring device determines the magnitude of the uncertainty for the recorded measurement. Copyright © Houghton Mifflin Company. All rights reserved. 8

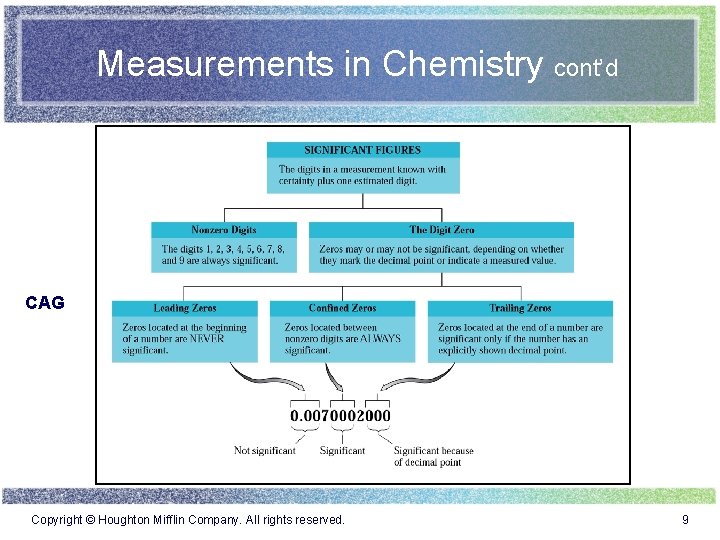
Measurements in Chemistry cont’d CAG Copyright © Houghton Mifflin Company. All rights reserved. 9

Measurements in Chemistry cont’d → Fig. 2. 6 The digital readout on an electronic calculator usually shows more digits than are needed. Copyright © Houghton Mifflin Company. All rights reserved. 10

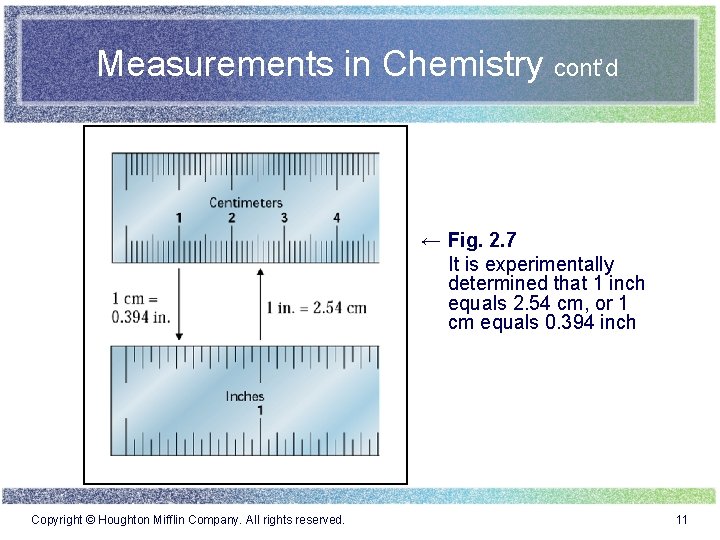
Measurements in Chemistry cont’d ← Fig. 2. 7 It is experimentally determined that 1 inch equals 2. 54 cm, or 1 cm equals 0. 394 inch Copyright © Houghton Mifflin Company. All rights reserved. 11

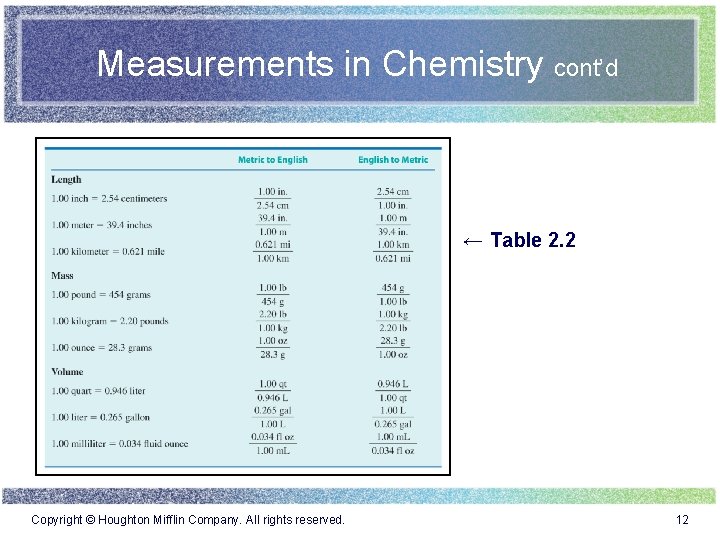
Measurements in Chemistry cont’d ← Table 2. 2 Copyright © Houghton Mifflin Company. All rights reserved. 12

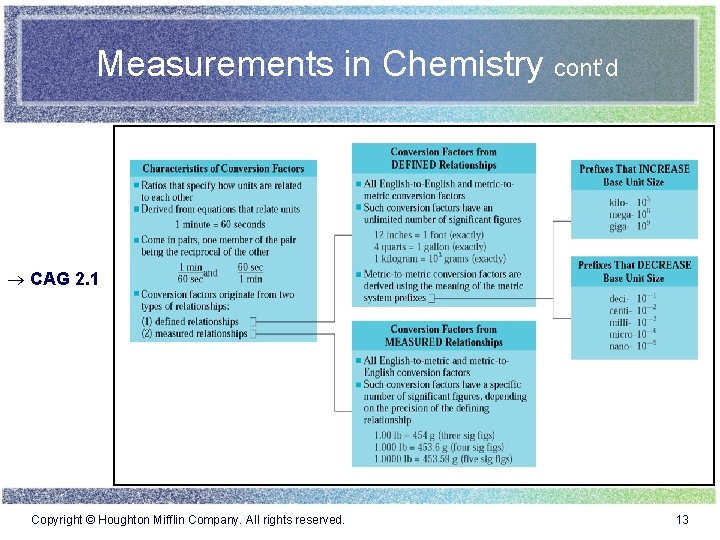
Measurements in Chemistry cont’d ® CAG 2. 1 Copyright © Houghton Mifflin Company. All rights reserved. 13

Measurements in Chemistry cont’d → Fig. 2. 8 Both of these items have a mass of 23 grams, but they have very different volumes; therefore, their densities are different as well. Copyright © Houghton Mifflin Company. All rights reserved. 14

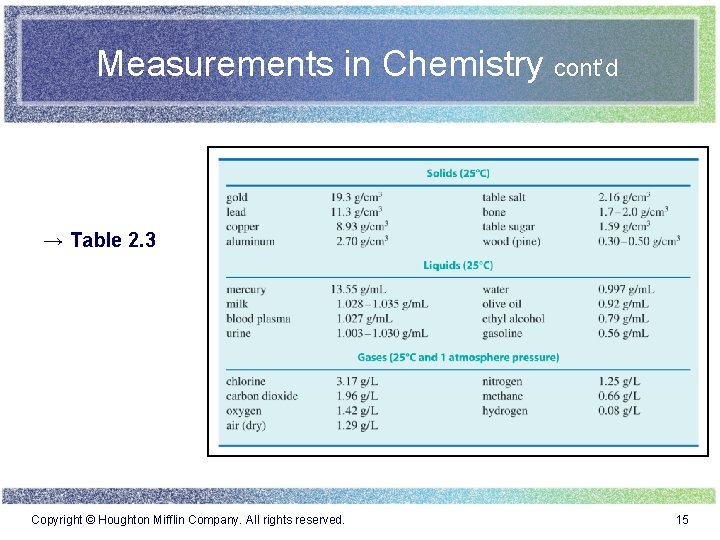
Measurements in Chemistry cont’d → Table 2. 3 Copyright © Houghton Mifflin Company. All rights reserved. 15

Measurements in Chemistry cont’d ← Fig. 2. 9 The penny is less dense than the mercury it floats on. Copyright © Houghton Mifflin Company. All rights reserved. 16

Measurements in Chemistry cont’d ® CC 2. 1 Copyright © Houghton Mifflin Company. All rights reserved. 17

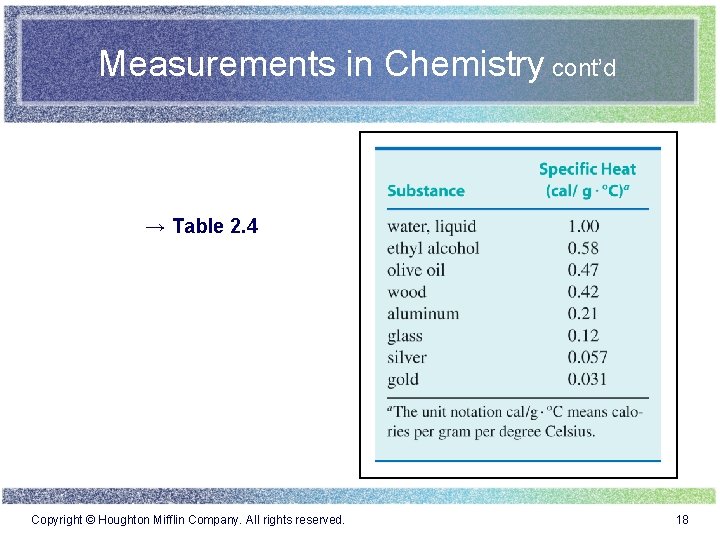
Measurements in Chemistry cont’d → Table 2. 4 Copyright © Houghton Mifflin Company. All rights reserved. 18

Measurements in Chemistry cont’d → Fig 2. 10 The relationships among the Celsius, Kelvin, and Fahrenheit temperature scales are determined by the degree sizes and the reference point values. Copyright © Houghton Mifflin Company. All rights reserved. 19