CHAPTER NOS LESSON 1 LESSON 1 UNDERSTANDING SCIENCE











































- Slides: 52

CHAPTER NOS LESSON 1

LESSON 1 UNDERSTANDING SCIENCE

KEY CONCEPTS • Recognize that the basic process used in scientific investigations involves questioning, observing, and recording and sharing results • Identify ways that sciences processes can be used to make informed decisions in the community, state, and nation • Recognize examples of evidence that

WHAT IS SCIENCE?

SCIENCE • The investigation and exploration of natural events and of the new information that results from those investigations • Uses: – Reasoning – Creativity – Skepticism

BRANCHES OF SCIENCE Life science Earth Science Physical Science

LIFE SCIENCE • Biology (life science) is the study of all living things

EARTH SCIENCE • The study of the Earth, including its landforms, rocks, soil, and forces that shape Earth’s surface

PHYSICAL SCIENCE • The study of chemistry and physics

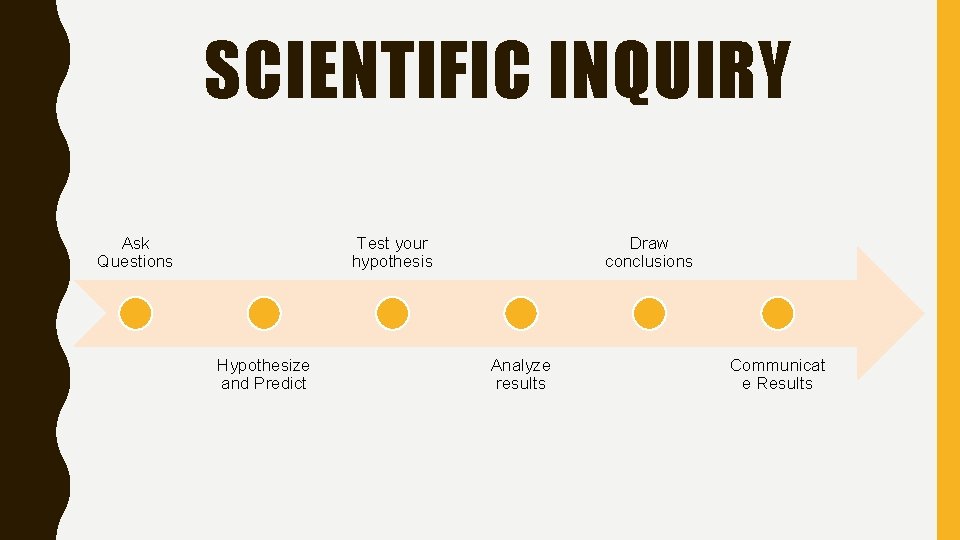
SCIENTIFIC INQUIRY Ask Questions Test your hypothesis Hypothesize and Predict Draw conclusions Analyze results Communicat e Results

1. ASK QUESTIONS • Make observations and inferences • An observation is using one or more of your senses to gather information and take note of what occurs • An inference is a logical explanation of an observation that is drawn from prior knowledge or experience

2. HYPOTHESIZE AND PREDICT • A hypothesis is a possible explanation about an observation that can be tested by scientific investigations + =

3. TEST YOUR HYPOTHESIS • Testing your predictions

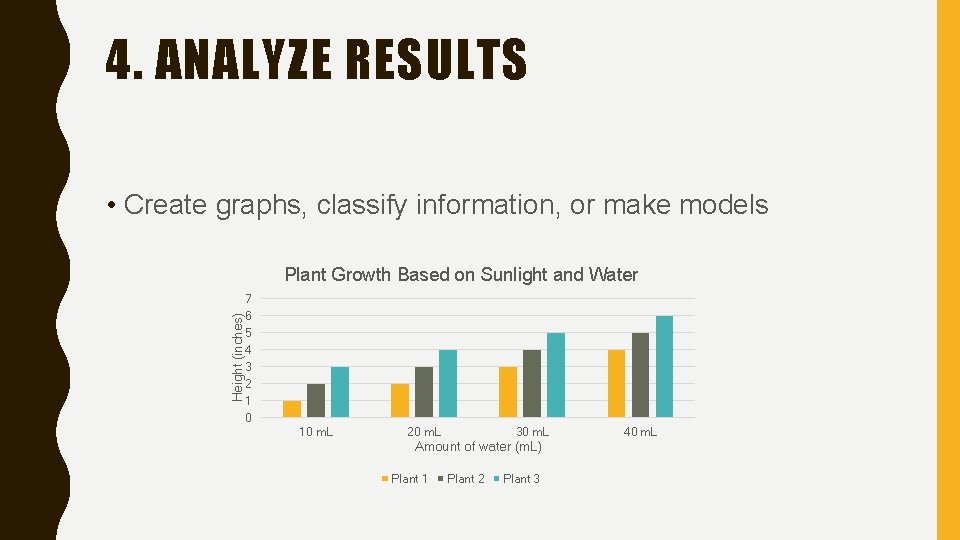
4. ANALYZE RESULTS • Create graphs, classify information, or make models Height (inches) Plant Growth Based on Sunlight and Water 7 6 5 4 3 2 1 0 10 m. L 20 m. L 30 m. L Amount of water (m. L) Plant 1 Plant 2 Plant 3 40 m. L

SCIENTIFIC MODELS A scientific model represents objects, phenomena, and physical processes in a consistent and logical ways.

5. DRAW CONCLUSIONS • A conclusion is a summary of the information gained from testing a hypothesis The more sunlight and water a plant receives the taller it will grow.

6. COMMUNICATE RESULTS • Writing scientific articles, speaking at conferences, or exchanging information.

WHAT ARE THE SIX STEPS OF SCIENTIFIC INQUIRY? 1. Ask Questions 2. Hypothesize and Predict 3. Test Your Hypothesis 4. Analyze Results 5. Draw Conclusions 6. Communicate Results

SCIENTIFIC THEORY AND SCIENTIFIC LAWS SCIENTIFIC THEORY SCIENTIFIC LAW • Explanation of observations or events based on knowledge gained from MANY observations and investigations • Describes a pattern or an event in nature that is ALWAYS true • Is an explanation on why or how an event occurred • States that an event will occur

EXAMPLES OF SCIENTIFIC THEORIES AND LAWS THEORY • Big Bang Theory LAW • Theory of General Relativity • Universal Law of Gravitation • Theory of Plate Tectonics • Newton’s Laws of Motion • Theory of Evolution • Law of Segregation • Atomic Theory • Ideal Gas Law • Cell Theory • Ohm’s Law

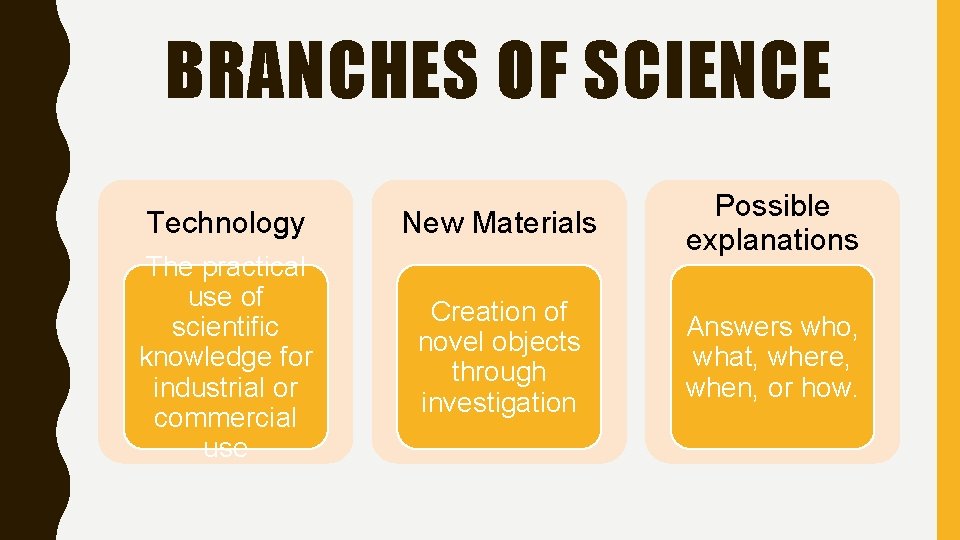
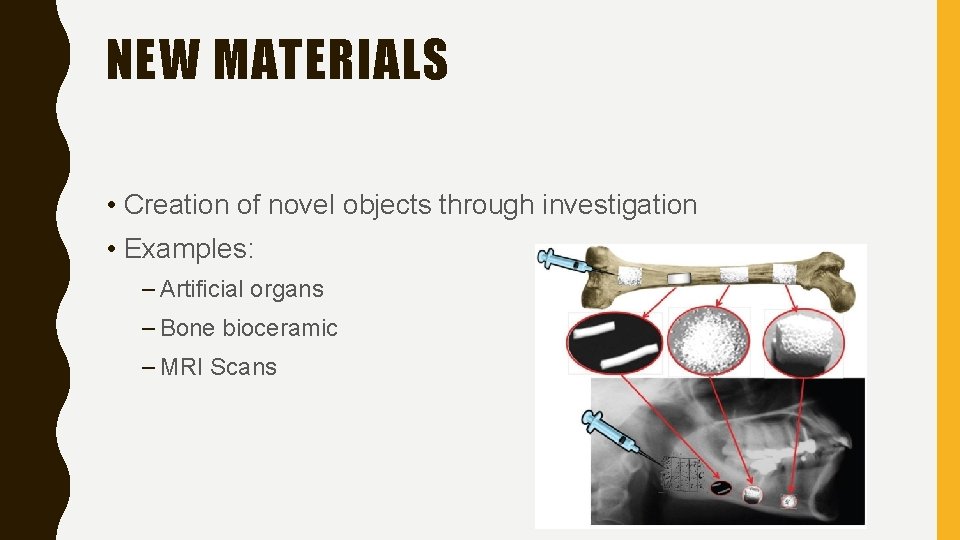
BRANCHES OF SCIENCE Technology New Materials The practical use of scientific knowledge for industrial or commercial use Possible explanations Creation of novel objects through investigation Answers who, what, where, when, or how.

TECHNOLOGY • The practical use of scientific knowledge for industrial or commercial use

NEW MATERIALS • Creation of novel objects through investigation • Examples: – Artificial organs – Bone bioceramic – MRI Scans

POSSIBLE EXPLANATIONS • Answers who, what, where, when, or how.

SKEPTICISM IN THE MEDIA

SKEPTICISM IN THE MEDIA

CAN SCIENCE ANSWER ALL QUESTIONS? NOPE

SAFETY IN SCIENCE • ALWAYS wear appropriate safety equipment • LISTEN to teacher’s instructions • LEARN to recognize potential hazards • KNOW the meaning of safety Symbols • READ more about science laboratory safety ETHICS

LESSON 2 MEASUREMENT AND SCIENTIFIC TOOLS

KEY CONCEPTS • Why did scientists create the International System of Units (SI)? • Why is scientific notation a useful tool for scientists? • How can tools, such as graduated cylinders and triple-beam balances, assist physical scientists?

DESCRIPTION AND EXPLANATION • A description is a spoken or written summary of observations. • An explanation is an interpretation of observations.

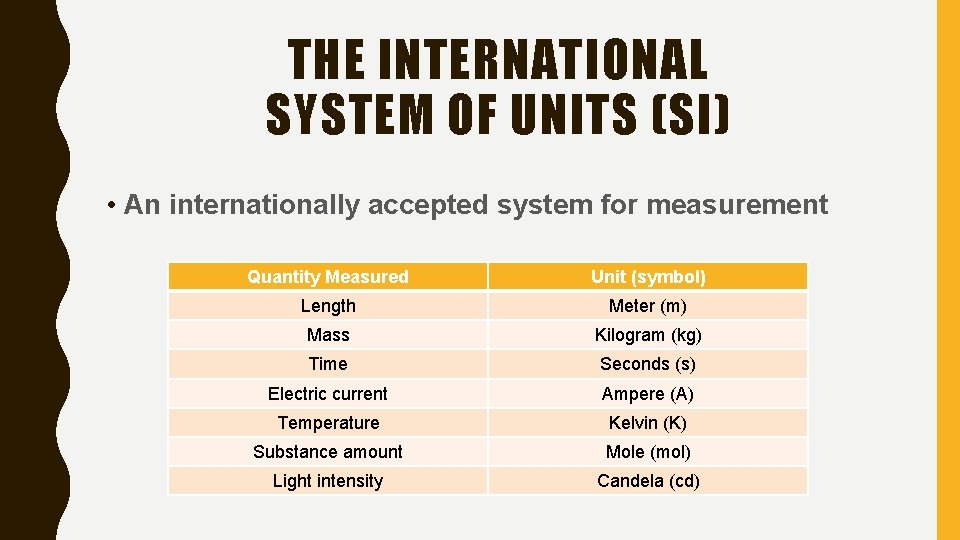
THE INTERNATIONAL SYSTEM OF UNITS (SI) • An internationally accepted system for measurement Quantity Measured Unit (symbol) Length Meter (m) Mass Kilogram (kg) Time Seconds (s) Electric current Ampere (A) Temperature Kelvin (K) Substance amount Mole (mol) Light intensity Candela (cd)

WHAT ARE THE SEVEN SI BASE UNITS? • LENGTH – METERS • MASS – KILOGRAMS • TIME – SECONDS • ELECTRIC CURRENT – AMPERE • TEMPERATURE – KELVIN • SUBSTANCE AMOUNT – MOLE • LIGHT INTENSITY - CANDELA

KELVIN T(K) = T(°C) + 273 Convert 10 degrees Celsius to Kelvin: T(K) = 10°C + 273 = 283 K Boiling temperature for water = 100° C

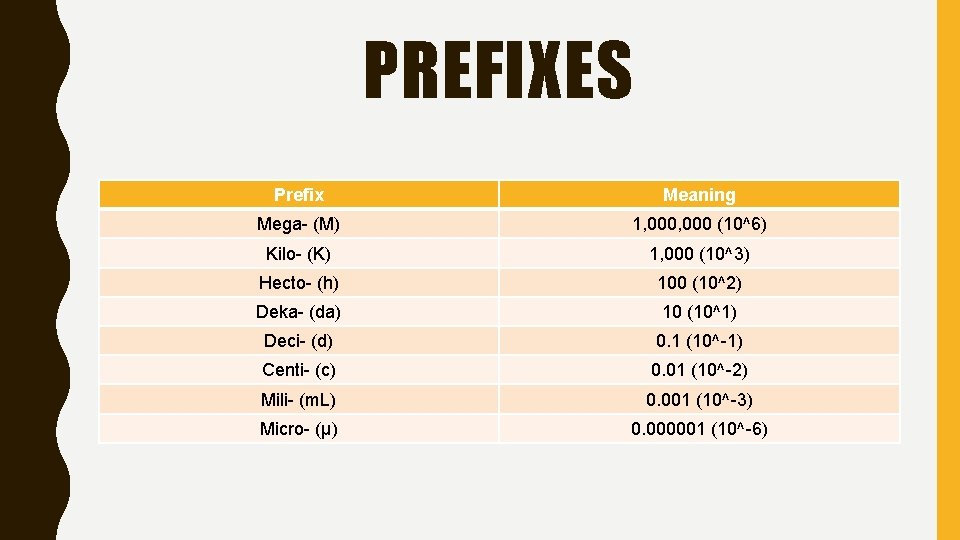
PREFIXES Prefix Meaning Mega- (M) 1, 000 (10^6) Kilo- (K) 1, 000 (10^3) Hecto- (h) 100 (10^2) Deka- (da) 10 (10^1) Deci- (d) 0. 1 (10^-1) Centi- (c) 0. 01 (10^-2) Mili- (m. L) 0. 001 (10^-3) Micro- (μ) 0. 000001 (10^-6)

King Henry Died Merrily Drinking Chocolate Milk Laughingly Gradually Kilo Hecto Deka Meter Deci Centi Milli Liter Gram

CONVERSION 1. 360 cm = _____mm 2. 5812 grams = ______ kg 3. 5000 dekaliters = ____ centiliters 4. 5. 06 cm = ______ m 5. 0. 345 kg = ______ dg

CONVERSION 1. A biologist measures an Emperor goose in the field. Her triple-beam balance shows the good has a mass of 2. 8 kg. How many grams is the Emperor goose? 2. Gaby is measuring water in a graduated cylinder and finds a volume of 2. 7 L. How many m. L is in the graduated cylinder? 3. A geologist is measuring the diameter of a circular rock and finds it to be 0. 5 mm. How many cm is the diameter of the rock?

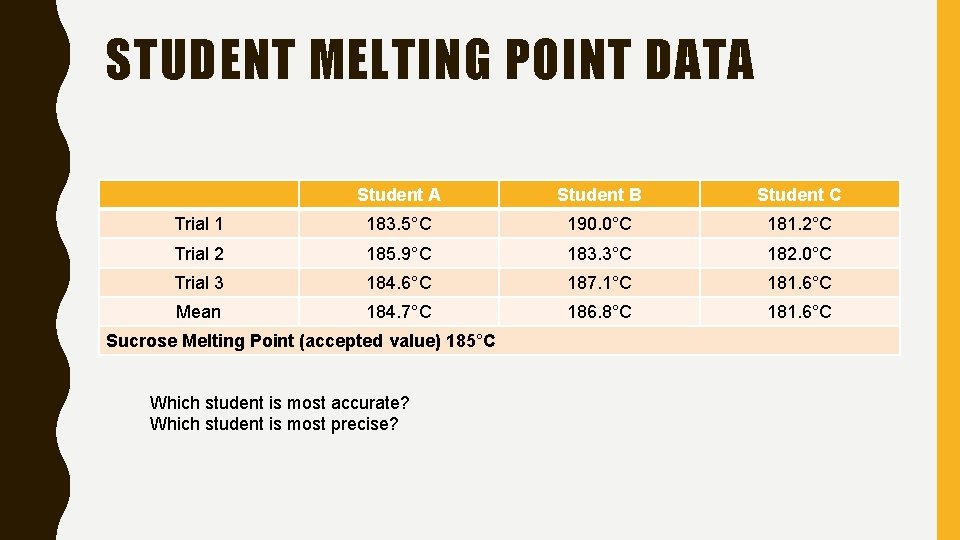
PRECISION AND ACCURACY • Accuracy is a description of how close a measurement to an accepted or true value. • Precision is a description of how similar or close measurements are to each other

STUDENT MELTING POINT DATA Student B Student C Trial 1 183. 5°C 190. 0°C 181. 2°C Trial 2 185. 9°C 183. 3°C 182. 0°C Trial 3 184. 6°C 187. 1°C 181. 6°C Mean 184. 7°C 186. 8°C 181. 6°C Sucrose Melting Point (accepted value) 185°C Which student is most accurate? Which student is most precise?

HOW TO WRITE IN SCIENTIFIC NOTATION 1. Write the original number. 2. Move the decimal point to the right or to the left to make the number between 1 and 10. Count the number of decimal places moved and note the direction. 3. Rewrite the number deleting all extra zeros to the right or to the left of the decimal point. 4. Write a multiplication symbol and the number 10 with an exponent. The exponent should equal the number of places that you moved the decimal point in step 2. If you moved the decimal point to the left, the exponent is positive. If you moved the decimal point to the right, the exponent is negative.

SCIENTIFIC TOOLS • Science Journal • Balances • Thermometer • Glassware • Compound microscope • Computers (hardware and software)

TOOLS USED BY LIFE SCIENTISTS • Magnifying Lens • Slide • Dissecting tools • Pipette • Calculators

BUNSEN BURNER

EVAPORATING DISH

FLASK

STIRRING ROD

SCUPULA

LESSON 3 CASE STUDY

DESIGNING A CONTROLLED EXPERIMENT • A variable is any factor in an experiment that can have more than one value • The dependent variable (responding variable) is the factor measured or observed during an experiment • The independent variable (manipulated variable) is the factor that is tested; it is changed by the investigator to observe how it affects a dependent variable • Constants are the factors in an experiment that remain the same

INDEPENDENT VS. DEPENDENT

DESIGNING A CONTROLLED EXPERIMENTAL GROUP CONTROL GROUP Used to study how a change in the independent variable changes the dependent variable. Contains the same factors as the experimental group, but the independent variable is NOT changed