Maximum Entropy Maximum Entropy Production and their Application











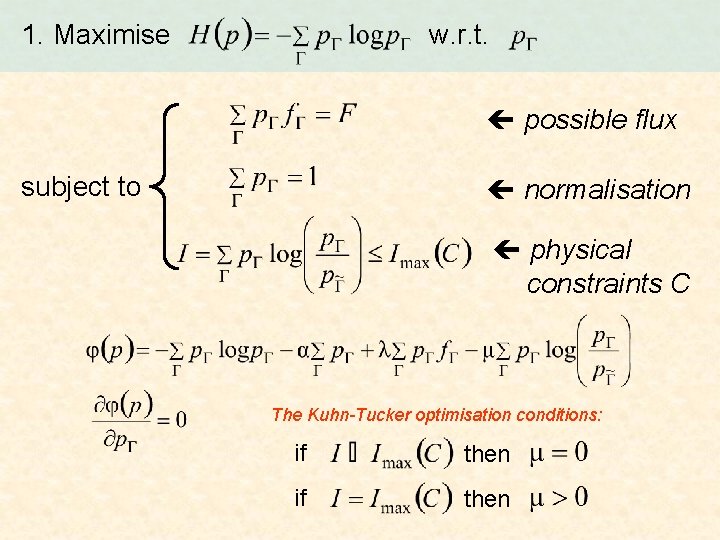








- Slides: 28

Maximum Entropy, Maximum Entropy Production and their Application to Physics and Biology Prof. Roderick C. Dewar Research School of Biological Sciences The Australian National University

Part 1: Maximum Entropy (Max. Ent) – an overview Part 2: Applying Max. Ent to ecology Ø Part 3: Maximum Entropy Production (MEP) Part 4: Applying MEP to physics & biology Dewar & Maritan (in preparation)

Part 3: Maximum Entropy Production (MEP) • The problem: to predict non-equilibrium fluxes from given constraints • The solution: apply Max. Ent to microscopic paths (Jaynes) - irreversibility in information-theoretical terms - Max. Ent implies maximum irreversibility • Max irreversibility MEP (steady states) • Max irreversibility Fokker-Planck equation (dynamics)

Part 3: Maximum Entropy Production (MEP) • The problem: to predict non-equilibrium fluxes from given constraints • The solution: apply Max. Ent to microscopic paths (Jaynes) - irreversibility in information-theoretical terms - Max. Ent implies maximum irreversibility • Max irreversibility MEP (steady states) • Max irreversibility Fokker-Planck equation (dynamics)

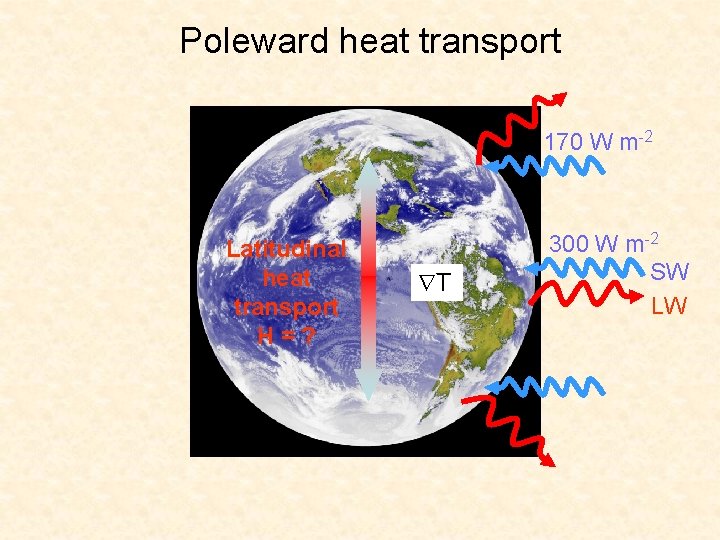
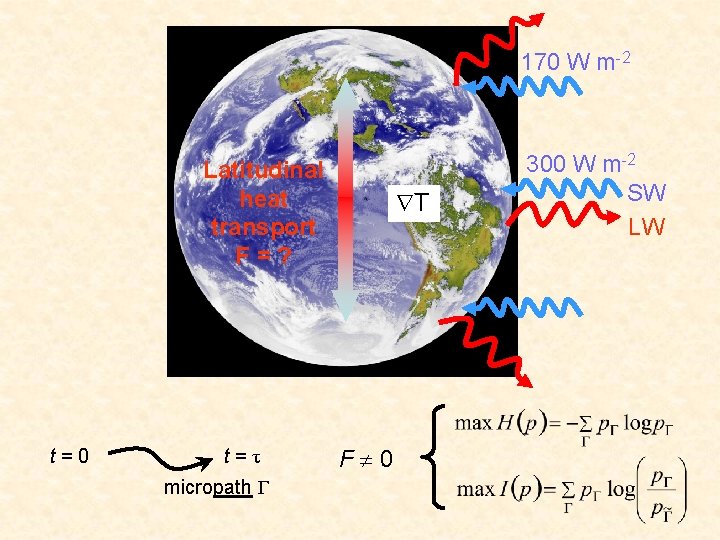
Poleward heat transport 170 W m-2 Latitudinal heat transport H=? T 300 W m-2 SW LW

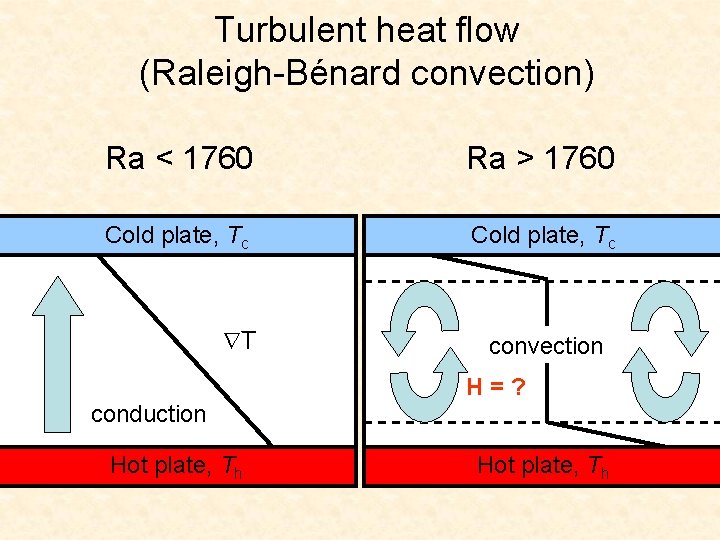
Turbulent heat flow (Raleigh-Bénard convection) Ra < 1760 Ra > 1760 Cold plate, Tc T convection H=? conduction Hot plate, Th

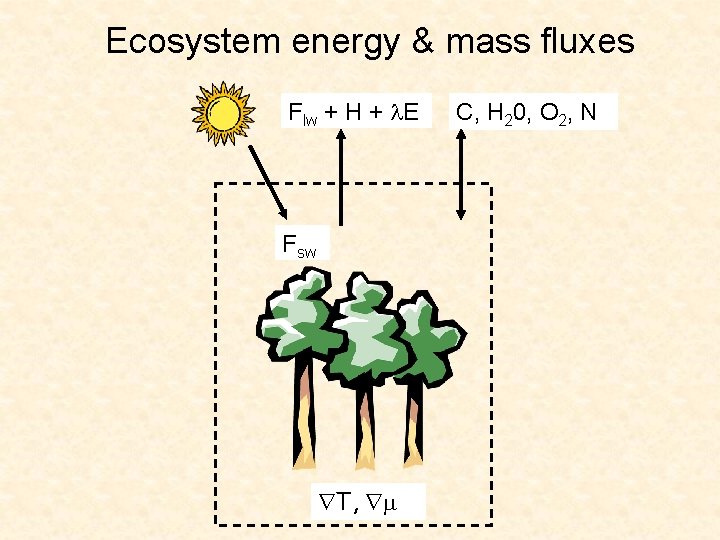
Ecosystem energy & mass fluxes Flw + H + E Fsw T, C, H 20, O 2, N

From among all those possible flux patterns compatible with the constraints, which one is reproducibly selected?

Part 3: Maximum Entropy Production (MEP) • The problem: to predict non-equilibrium fluxes from given constraints • The solution: apply Max. Ent to microscopic paths (Jaynes) - irreversibility in information-theoretical terms - Max. Ent implies maximum irreversibility • Max irreversibility MEP (steady states) • Max irreversibility Fokker-Planck equation (dynamics)

Γ = microscopic path of system + environment = path Γ followed in reverse Path entropy t=0 t=τ

Γ = microscopic path of system + environment t=0 t=τ = path Γ followed in reverse Path entropy Irreversibility (equilibrium)

Γ = microscopic path of system + environment t=0 t=τ = path Γ followed in reverse Path entropy Irreversibility (equilibrium) Physical constraints C I(p) is finite, i. e. macroscopic fluxes can run in reverse

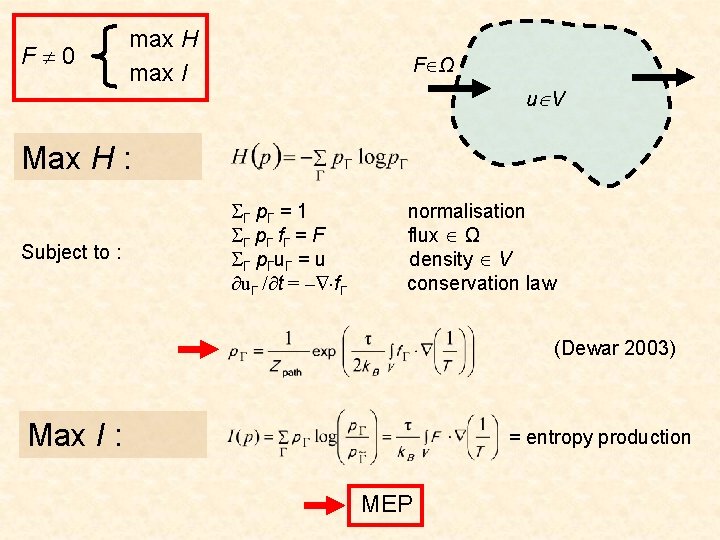
To select F, maximise H in two steps … 1. Maximise w. r. t. possible flux subject to normalisation physical constraints C Step 1 2. Maximise w. r. t. F reproducible flux

Step 1
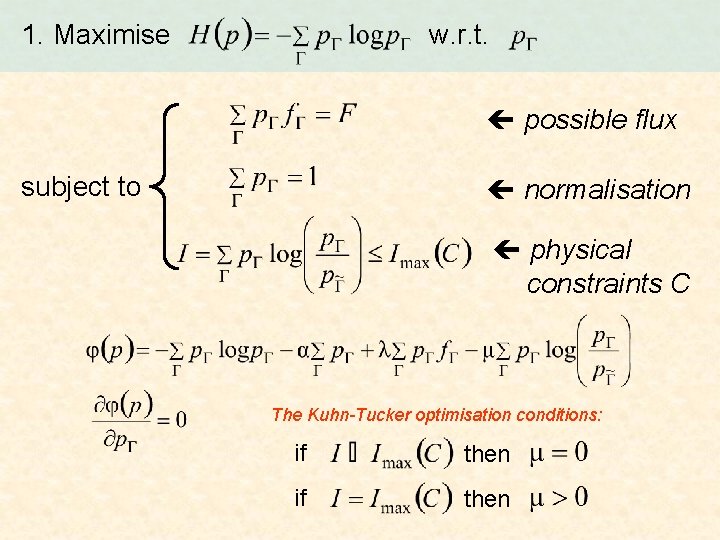
1. Maximise w. r. t. possible flux subject to normalisation physical constraints C The Kuhn-Tucker optimisation conditions: if then

Step 2

2. Maximise w. r. t. F reproducible F When μ = 0 we get (cf. Boltzmann) λ=F=0 i. e. equilibrium Therefore non-equilibrium systems (F 0) must satisfy μ > 0 i. e. maximum irreversibility F 0 max S max I

Part 3: Maximum Entropy Production (MEP) • The problem: to predict non-equilibrium fluxes from given constraints • The solution: apply Max. Ent to microscopic paths (Jaynes) - irreversibility in information-theoretical terms - Max. Ent implies maximum irreversibility • Max irreversibility MEP (steady states) • Max irreversibility Fokker-Planck equation (dynamics)

170 W m-2 Latitudinal heat transport F=? t=0 t=τ micropath Γ T F 0 300 W m-2 SW LW

F 0 max H max I F Ω u V Max H : Subject to : Γ pΓ = 1 Γ pΓ f Γ = F Γ pΓuΓ = u uΓ / t = – fΓ normalisation flux Ω density V conservation law (Dewar 2003) Max I : = entropy production MEP

Thermodynamic EP emerges from constraint of energy & mass balance. . EPΓ = V Σ fluxΓ force heat flow (1/T) stress strain mass flow (-μ/T) reaction rate affinity from energy balance from mass balance . . and this is why MEP is so general Dewar (2003)

Part 3: Maximum Entropy Production (MEP) • The problem: to predict non-equilibrium fluxes from given constraints • The solution: apply Max. Ent to microscopic paths (Jaynes) - irreversibility in information-theoretical terms - Max. Ent implies maximum irreversibility • Max irreversibility MEP (steady states) • Max irreversibility Fokker-Planck equation (dynamics)

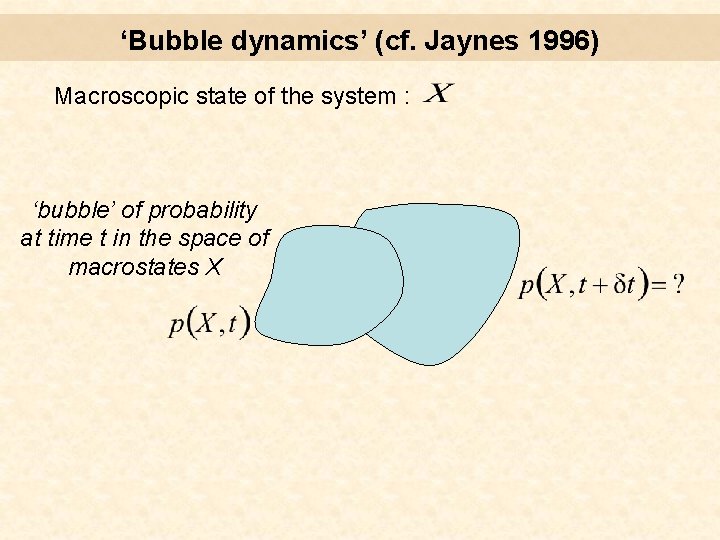
‘Bubble dynamics’ (cf. Jaynes 1996) Macroscopic state of the system : ‘bubble’ of probability at time t in the space of macrostates X

Maximum irreversibility macroscopic dynamics System state with probability at time t Path probability: Conditional state velocity : Probability current: Conservation of probability : can be expressed in terms of p(X, t) and v(X, t) Max I w. r. t. v(X, t) subject to constraints reproducible v(X, t) temporal evolution of p(X, t) under C

Example: Gaussian fluctuations (near equilibrium) t +dt t Maximise I w. r. t. v(X, t) subject to Non-eq. forcing Onsager drift diffusion Fokker-Planck equation: p/ t = - (pv)

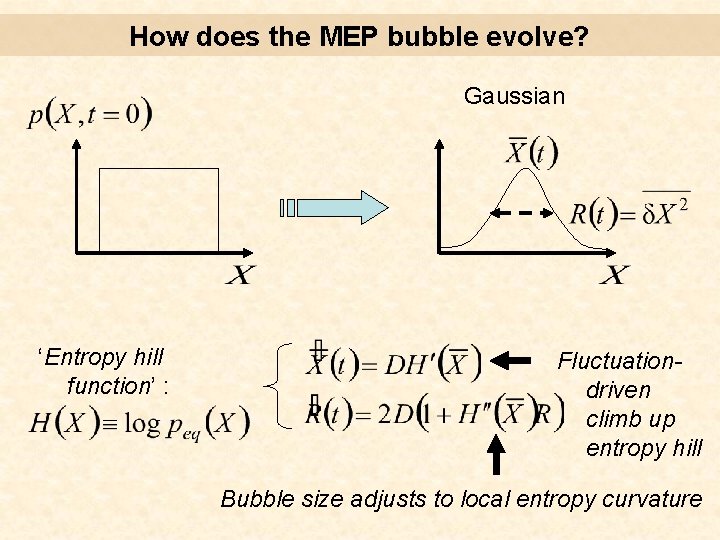
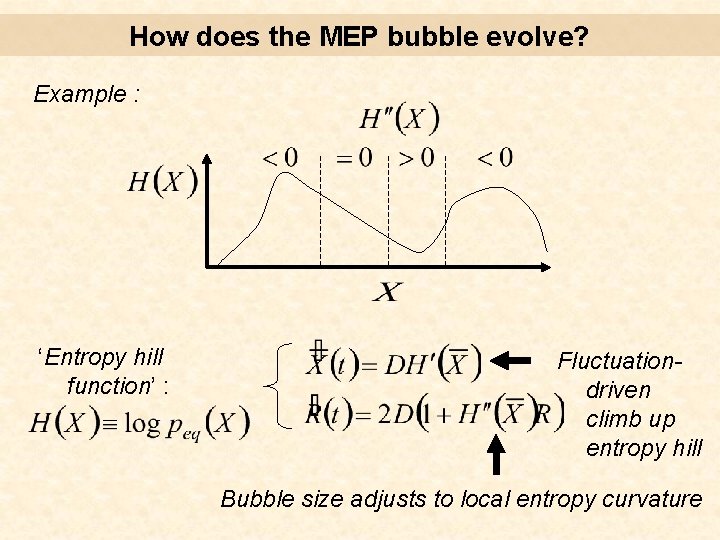
How does the MEP bubble evolve? Gaussian ‘Entropy hill function’ : Fluctuationdriven climb up entropy hill Bubble size adjusts to local entropy curvature

How does the MEP bubble evolve? Example : ‘Entropy hill function’ : Fluctuationdriven climb up entropy hill Bubble size adjusts to local entropy curvature

Summary of Lecture 3 … Boltzmann Gibbs Shannon Jaynes • Max. Ent (reproducible behaviour) systems arbitrarily far from equilibrium obey maximum irreversibility (Max I) • Max I governs selection of nonequilibrium steady states (MEP) and macroscopic dynamics (e. g. Fokker-Planck)