Remember you cannot predict the sign of entropy

Remember you cannot predict the sign of entropy unless the reaction involves a change in the number of moles of gas N 2(g) + 3 H 2(g) 2 NH 3(g) ΔSo. Rx = Σn. So. Products - Σn. So. Reactants = (2 moles NH 3)(193 J/mole x K) - (1 mole N 2)(191. 5 J/mole x K) - (3 moles H 2)(130. 6 j/mole x K) = - 197 J/K

Entropy Change in the Surroundings Second Law of Thermodynamics- All real processes occur spontaneously in the direction that increases the entropy of the universe. For spontaneous reactions decreases in the entropy of the system can occur only if the increases in the entropy of the surroundings outweigh them. The surroundings either adds or removes heat from the system. The surroundings is a heat sink

The surroundings is a heat sink Exothermic Change • • • Heat lost by the system = heat gain by the surroundings Increase in freedom of motion of particles in the surroundings Particle energy is dispersed in the surroundings More kinetic energy in surroundings Slight temperature increase. Not noticeable qsystem< 0 • qsurroundings> 0 • ΔSsurroundings > 0 •

The surroundings is a heat sink Endothermic Change • • • Heat gain by the system = heat lost by the surroundings Decrease in freedom of motion of particles in the surroundings Particle energy is more localized in the surroundings Less kinetic energy in surroundings Slight temperature decrease. Not noticeable qsystem> 0 • qsurroundings< 0 • ΔSsurroundings < 0 •

Remember you cannot predict the sign of entropy unless the reaction involves a change in the number of moles of gas N 2(g) + 3 H 2(g) 2 NH 3(g) ΔSo. Rx = Σn. So. Products - Σn. So. Reactants ΔSo. System = (2 moles NH 3)(193 J/mole x K) - (1 mole N 2)(191. 5 J/mole x K) - (3 moles H 2)(130. 6 j/mole x K) = - 197 J/K < 0 Predicts non-spontaneous Rx

N 2(g) + 3 H 2(g) 2 NH 3(g) ΔSo. System = - 197 J/K • ΔSUniverse > 0 Spontaneous Rx thus ΔSsurroundings > +197 J/K • ΔHsystem = ΔHReaction = (2 mole NH 3)(-45. 9 KJ/mole NH 3) - (3 moles H 2)(0 KJ/mole H 2) - (1 mole N 2)( 0 KJ/mole N 2) • ΔSsurroundings = - qreversible/T = - 91. 8 KJ = -ΔHReaction/T

• ΔSSurroundings = (91. 8 KJ/mole)(1000 J/KJ) = 308 J/K (298 K) • ΔSUniverse = ΔSSystem + ΔSSurroundings = -197 J/K + 308 J/K = 111 J/K • ΔSUniverse > 0 Rx Spontaneous at 298 K

Temperature affects ΔSo. Surroundings • At 20 K - very little random motion • At 298 K – the surroundings already has a relatively large quantity of dispersed energy. Less effect ΔSo. Surroundings = - q. System = - ΔHSystem T T

Reactions at Equilibrium • ΔSUniverse > 0 Rx is Spontaneous • ΔSUniverse = ΔSSystem + At equilibrium Entropy ΔSSurroundings = 0 ΔSSystem = - ΔSSurroundings H 2 O(l) H 2 O(g) ΔSSystem = ΔSproducts - ΔSReactants = 195. 9 J/K - 86. 8 J/K = 109. 1 J/K = 188. 72 J/K – 69. 94 J/K = 118. 78 J/K @ 373 K @ 298 K
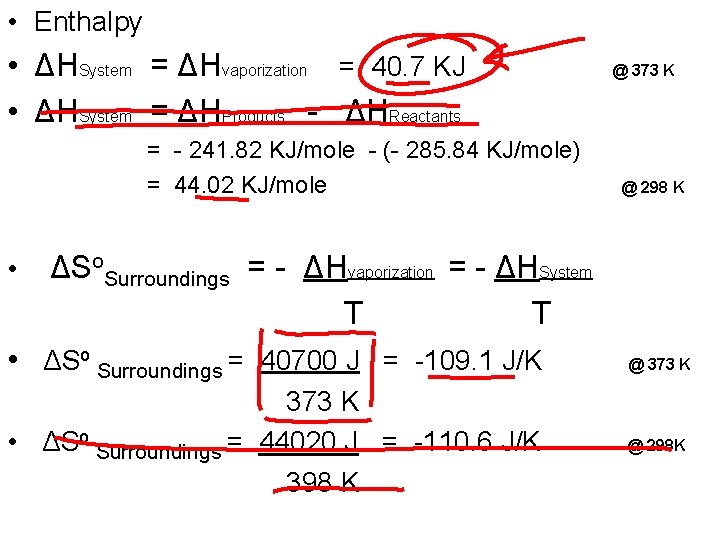
• Enthalpy • ΔHSystem = ΔHvaporization = 40. 7 KJ • ΔHSystem = ΔHProducts - ΔHReactants = - 241. 82 KJ/mole - (- 285. 84 KJ/mole) = 44. 02 KJ/mole • @ 373 K @ 298 K ΔSo. Surroundings = - ΔHvaporization = - ΔHSystem T T • ΔSo Surroundings = 40700 J = -109. 1 J/K @ 373 K • ΔSo Surroundings = 44020 J = -110. 6 J/K 398 K @ 298 K

• ΔSUniverse = ΔSSystem + ΔSSurroundings = 109. 1 J/K + (-109. 1 J/K) = 0 @ 373 K = 118. 8 J/K + (-110. 6 J/K) = 8. 2 J/K @ 298 K At equilibrium the entropy change of the forward reaction is equal in magnitude but opposite in sign to the entropy change of the reverse reaction. When a system reaches equilibrium , neither the forward nor the reverse reaction is spontaneous.

Spontaneous Endothermic & Exothermic Reactions No matter what it’s entropy change, a reaction occurs because the total entropy of the reacting system and its surroundings ( the universe) increases
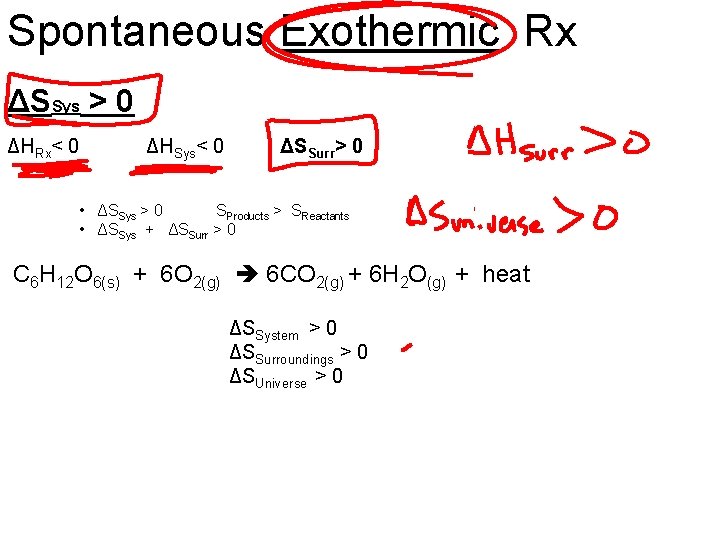
Spontaneous Exothermic Rx ΔSSys > 0 ΔHRx< 0 ΔHSys< 0 ΔSSurr> 0 • ΔSSys > 0 SProducts > SReactants • ΔSSys + ΔSSurr > 0 C 6 H 12 O 6(s) + 6 O 2(g) 6 CO 2(g) + 6 H 2 O(g) + heat ΔSSystem > 0 ΔSSurroundings > 0 ΔSUniverse > 0
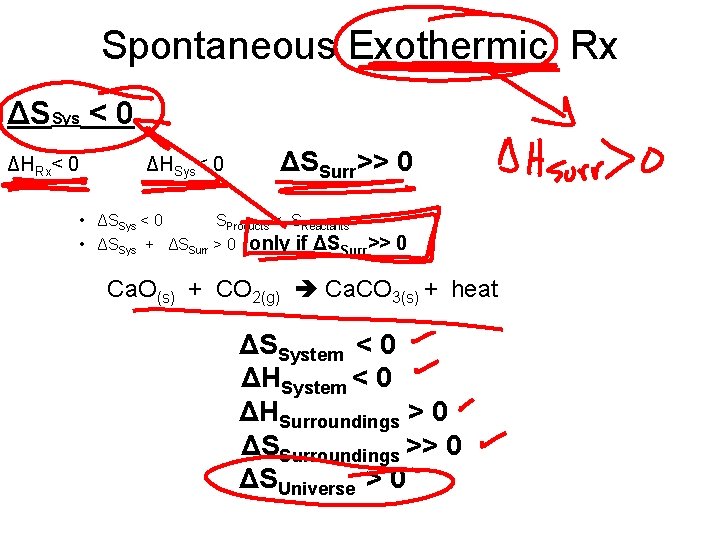
Spontaneous Exothermic Rx ΔSSys < 0 ΔHRx< 0 ΔHSys< 0 ΔSSurr>> 0 • ΔSSys < 0 SProducts < SReactants • ΔSSys + ΔSSurr > 0 only if ΔSSurr>> 0 Ca. O(s) + CO 2(g) Ca. CO 3(s) + heat ΔSSystem < 0 ΔHSurroundings > 0 ΔSSurroundings >> 0 ΔSUniverse > 0

Spontaneous Endothermic Rx • ΔHSystem > 0 ΔSSurr = - ΔHSys < 0 T • Solution process of ionic compounds • Heat is absorbed to form the solution • Crystalline solids become free moving ions ΔSSystem >> 0 ΔSSystem > - ΔSSurroundings ΔSUniverse >> 0

Spontaneous Endothermic Rx • ΔHSystem > 0 ΔSSurr = - ΔHSys < 0 T If ΔSSys is also negative then the reaction is non-spontaneous.
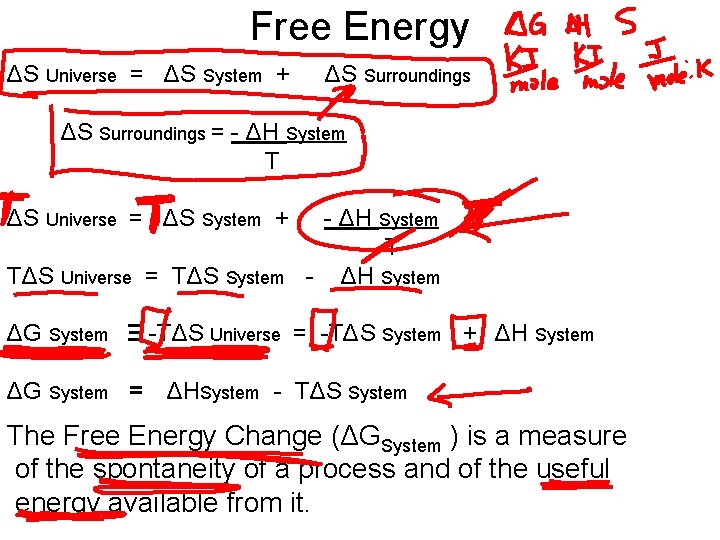
Free Energy ΔS Universe = ΔS System + ΔS Surroundings = - ΔH System T ΔS Universe = ΔS System + TΔS Universe = TΔS System - ΔH System T - ΔH System ΔG System Ξ -TΔS Universe = -TΔS System + ΔH System ΔG System = ΔHSystem - TΔS System The Free Energy Change (ΔGSystem ) is a measure of the spontaneity of a process and of the useful energy available from it.

ΔGSystem Ξ -TΔSUniverse = -TΔSSystem + ΔHSystem ΔSuniverse > 0 ΔSuniverse < 0 ΔSuniverse = 0 Spontaneous Process Non-spontaneous Process at equilibrium The Absolute Temperature is always positive thus for a spontaneous process -TΔSUniverse < 0 ΔGuniverse > 0 ΔGuniverse = 0 Spontaneous Process Non-spontaneous Process at equilibrium

ΔGSystem Ξ -TΔSUniverse = -TΔSSystem + ΔHSystem Free Energy and Equilibrium and Rx Direction a. A + b. B c. C + d. D Q<K Q>K Q=K Q/K < 1 Q/K > 1 Q/K = 1 Rx moves Right Rx moves Left Rx @ Equilibrium K = [C]c[D]d = Q [A] a[B]b Ln(Q/K) < 0 Ln(Q/K) > 0 Ln(Q/K) = 0 ΔG < 0 ΔG > 0 ΔG = RTLn(Q/K) = RTLn(Q) - RTLn(K) @ Standard State Conditions Concentrations = 1 M ΔGo = RTLn(1) - RTLn(K) ΔGo = - RTLn(K) ΔG = ΔGo - RTLn(Q)

A process non-spontaneous in one direction (ΔG > 0 ) is spontaneous in the opposite direction (ΔG < 0 ) ΔGSystem is used to predict the spontaneity rather than ΔSSystem and ΔSSurroundings ΔGSystem Hess’s Law = ΔHSystem - TΔSSystem ΔGo. Rx = Σn. Gfo. Products - Σn. Gfo. Reactants
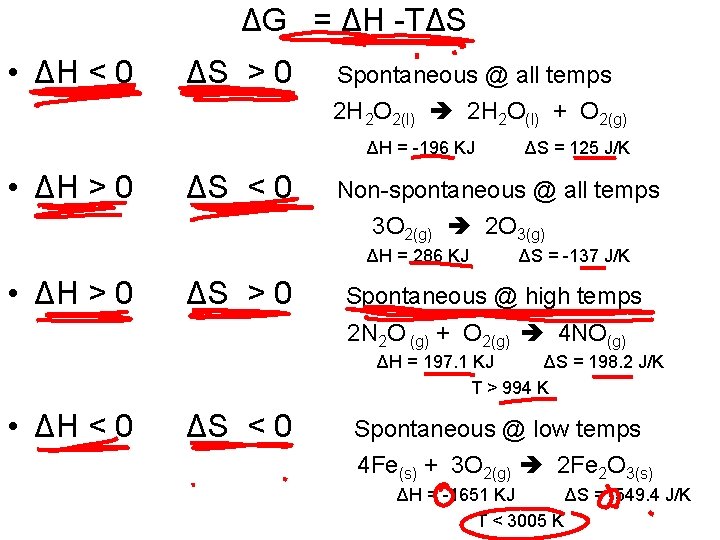
ΔG = ΔH -TΔS • ΔH < 0 ΔS > 0 Spontaneous @ all temps 2 H 2 O 2(l) 2 H 2 O(l) + O 2(g) ΔH = -196 KJ • ΔH > 0 ΔS < 0 ΔS = 125 J/K Non-spontaneous @ all temps 3 O 2(g) 2 O 3(g) ΔH = 286 KJ • ΔH > 0 ΔS = -137 J/K Spontaneous @ high temps 2 N 2 O (g) + O 2(g) 4 NO(g) ΔH = 197. 1 KJ ΔS = 198. 2 J/K T > 994 K • ΔH < 0 ΔS < 0 Spontaneous @ low temps 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) ΔH = -1651 KJ ΔS = -549. 4 J/K T < 3005 K

- Slides: 22