Entropy Spontaneity and Gibbs Free Energy Entropy Entropy

Entropy, Spontaneity and Gibb’s Free Energy

Entropy • Entropy is a measure of chaos • Or more scientifically. . . • Entropy is a measure of how the available energy is distributed among the particles present

• More disorder means more entropy, because there are more ways the energy can be distributed. • More disorder (i. e. more entropy) is more likely • Imagine a year 1 class sat nicely in alphabetical order. This has low disorder => low entropy and a very low chance of happening!!!!


• Entropy is given the symbol S • S⁰ is standard entropy measured at 298 K and 100 k. Pa pressure • Units are JK-1 mol-1 • NB. Joules not k. J – this will be important in calculations later

• Unlike enthalpy, we can calculate absolute values for entropy • (Using the Boltzmann constant (1. 38064852 × 10 -23 m 2 kg s-2 K-1) – no need to learn that!!) • E. g. S⁰ H 2 = 131 k. Jmol-1

• Obviously, ΔS⁰ is change is standard entropy. • If ΔS⁰ is positive, there is an increase in the ways energy can be distributed.

Predicting the sign of an entropy change • In general there is an increase in entropy • Solid liquid gas
• Examples: • N 2(g) + 3 H 2(g) 2 NH 3(g) • Entropy decrease; ΔS⁰ -ve • Ca. CO 3 (s) Ca. O(s) +CO 2(g) • Entropy increase; ΔS⁰ +ve

• Other stupid generalisations: • Entropy increases as temperature increases • Entropy increases as concentration increases • Entropy increases in mixtures relative to pure substances

Calculating entropy ∆S∅ reaction = ∑S∅ ∅ ∑S (products) (reactants) • • E. g. calculate entropy change for N 2(g) + 3 H 2(g) 2 NH 3(g) • Given S N 2 = 192; S H 2 = 131; S NH 3 = 193 (all JK-1 mol-1) • ∆S∅ = (2*193) – [192+ (3*131)] • ∆S∅ = -199 JK-1 mol-1

Predicting Spontaneity

Spontaneous reaction Moves towards completion or equilibrium under a given set of conditions without external intervention Reactions not taking place under a given set of conditions are said to be non-spontaneous

• In English – spontaneous reactions happen without any outside influence. Non-spontaneous reactions don’t!! • BUT spontaneous reactions don’t have to happen quickly. • Diamond spontaneously turns to graphite – just very, very slowly! • (it takes 100 billion years at rtp apparently, but only a few minutes at 2000⁰C)

15. 2 Entropy and Spontaneity Essential idea: A reaction is spontaneous if the overall transformation leads to an increase in total entropy (system + surroundings). The direction of a spontaneous change always increases the total entropy of the universe at the expense of energy to do useful work. This is known as the second law of thermodynamics

• Some super smart scientists managed to make this into an equation (see P 229 for a simplified derivation!!) • ∆G = ∆H - T∆S • ∆H is the change in enthalpy for the reaction; • ∆S is the change in entropy for the reaction • T is the temperature in Kelvin • I’ll say that again, and you’ll still forget it – KELVIN!!

• And G is Gibbs Free Energy

• For a reaction to be spontaneous, ΔG for the reaction must be negative. • Why? From the derivation (see P 229!) ΔG=-T ΔSuniverse • For a reaction to be spontaneous, the entropy of the universe must increase. Hence ΔG must be negative.

• Because ΔG contains a Temperature related term, spontaneity can be influenced by the temperature. Sign of the enthalpy change Sign of the entropy change Spontaneity positive (+) The reaction is spontaneous at high temperature positive (+) negative (-) The reaction is never spontaneous negative (-) The reaction is spontaneous at low temperature negative (-) positive (+) The reaction is always spontaneous

Gibbs Free Energy and Equilibrium • Consider the reaction • N 2(g) + 3 H 2(g) 2 NH 3(g) ΔG=-33 k. Jmol-1 • How can this reach equilibrium? The reverse reaction shouldn’t be spontaneous • BUT mixtures have a higher entropy (lower G) than pure substances, so a mixture of N 2, H 2 and NH 3 should have the highest entropy.

• At equilibrium, the free energy of the products is equal to the free energy of the reactants, • So G=0 at equilibrium • Any move away from this position would give a positive ΔG • So a move from equilibrium cannot happen spontaneously
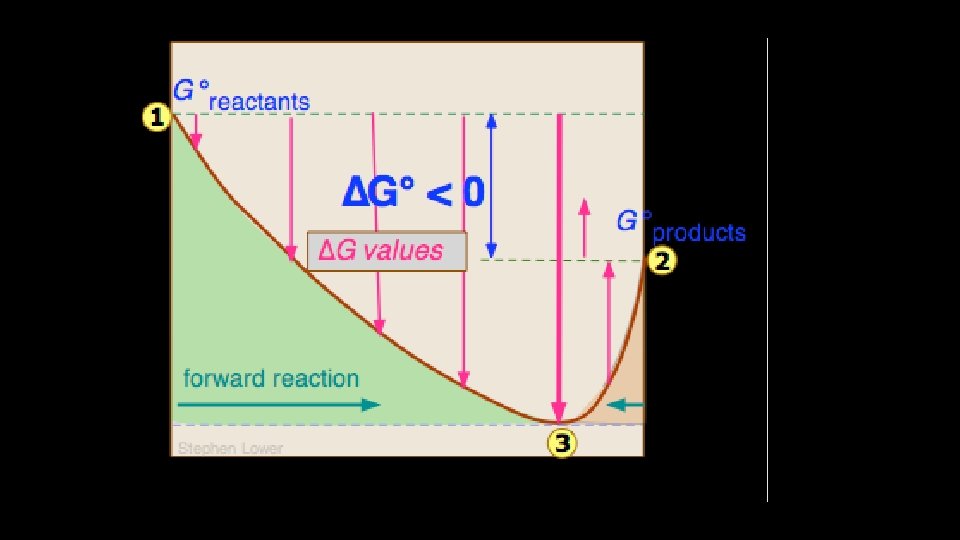

• The sign and value of ΔG also gives us information about where any equilibrium will lie. • The more –ve ΔG is the closer any equilibrium will be to pure products. • The more +ve ΔG is, the closer any equilibrium will be to pure reactants. • According to the textbook, if ΔG is more than +30 k. Jmol-1 the reaction doesn’t happen at all. • They also say that if ΔG is more negative than -30 k. Jmol-1 the reaction goes to completion. I’m not sure how to reconcile this with the Haber process. . . ?

- Slides: 24