Spontaneous Irreversible Irreversible Processes The nd 2 Law

Spontaneous (Irreversible) Irreversible Processes & The nd 2 Law of Thermodynamics

• From Statistical Arguments we’ve Quantitatively Defined Entropy: S k. Bln( ) k. B Boltzmann’s constant (E) Number of Microstates at a Given Energy
• We’ve also discussed the fact that Entropy is a measure of the amount of Disorder in a system.

Spontaneous Processes & Entropy Spontaneous Processes • Processes that can proceed with no outside intervention. • Entropy can be viewed as a measure of randomness or disorder in the atoms & molecules in a system. The 2 nd Law of Thermodynamics says that Total Entropy always increases in a spontaneous process! • So, Microscopic Disorder also always increases in a spontaneous process!
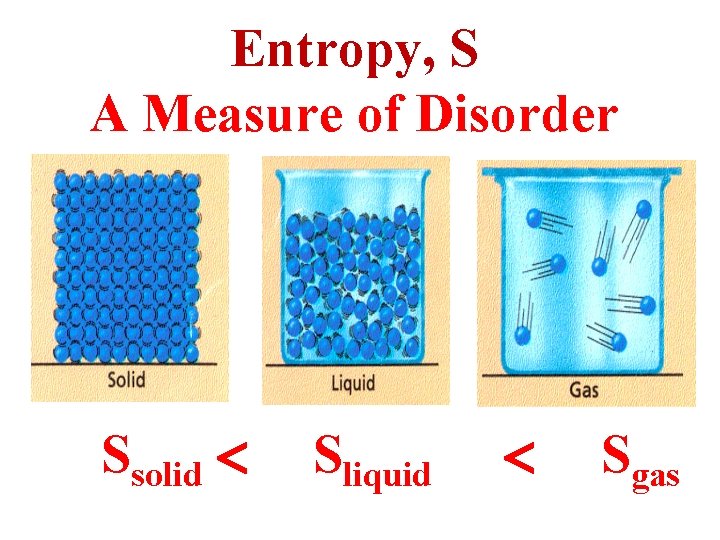
Entropy, S A Measure of Disorder Ssolid Sliquid Sgas
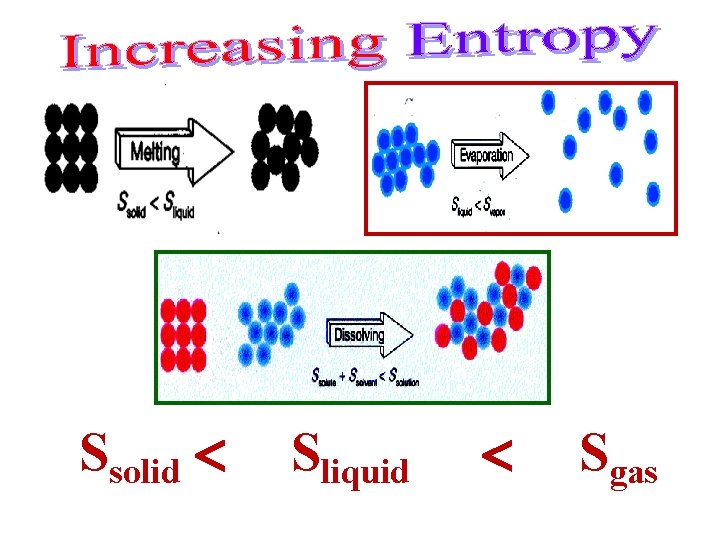
Ssolid Sliquid Sgas
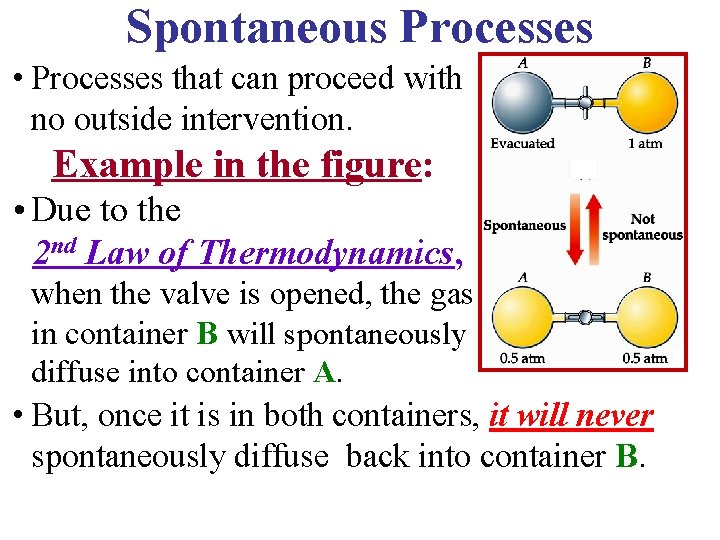
Spontaneous Processes • Processes that can proceed with no outside intervention. Example in the figure: • Due to the 2 nd Law of Thermodynamics, when the valve is opened, the gas in container B will spontaneously diffuse into container A. • But, once it is in both containers, it will never spontaneously diffuse back into container B.
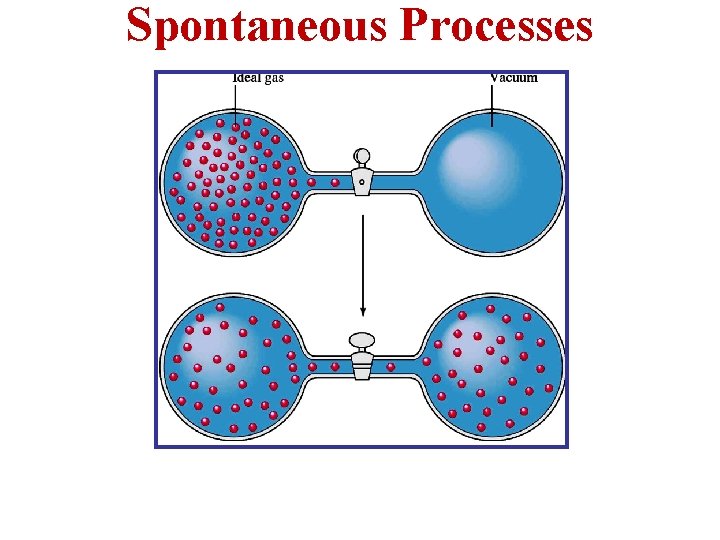
Spontaneous Processes

The 2 nd Law of Thermodynamics • Processes that are spontaneous in one direction aren’t spontaneous in the reverse direction. • Example in the figure: Due to the 2 nd Law of Thermodynamics the shiny nail at the top will, over a long time, rust & eventually look as at the bottom. But (obviously), if the nail is rusty, it will not ever spontaneously become shiny again!!

• Processes that are spontaneous at one temperature may be non-spontaneous at other temperatures. Example in the figure: • For T > 0 C the ice will melt spontaneously. • For T < 0 C, the reverse process is spontaneous.
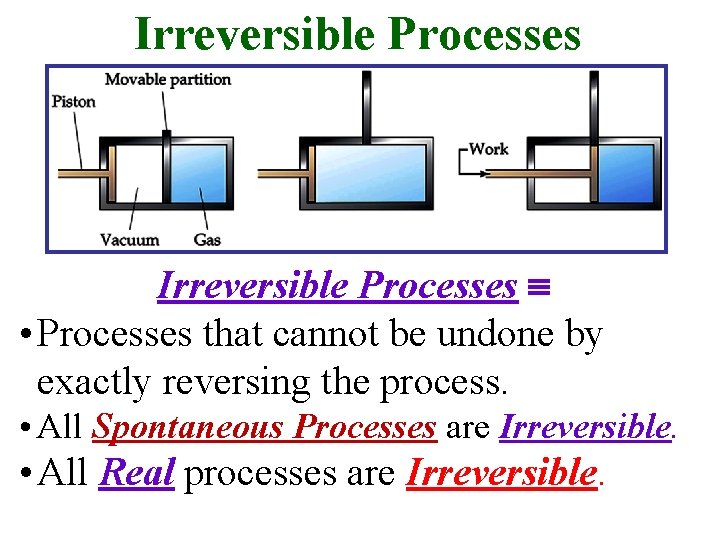
Irreversible Processes • Processes that cannot be undone by exactly reversing the process. • All Spontaneous Processes are Irreversible. • All Real processes are Irreversible.

Spontaneous Processes: Always occur on their own, without outside intervention. Always have a definite direction. The reverse process is never spontaneous. • Temperature can have an impact on spontaneity. Examples: • Ice melting or forming • Hot metal cooling at room temperature.

• Whenever a chemical system is in equilibrium, a reaction can go reversibly to reactants or products. • (Example: water vapor at 100 º C). • In a Spontaneous Process, the path between reactants and products is irreversible.

The reverse of a spontaneous process is not spontaneous. “Scrambled eggs don’t unscramble!”

Spontaneous, Irreversible Processes: More Examples 1. Due to friction, mechanical work changes into heat automatically. 2. Gas inflates toward vacuum. • Note!! The 2 nd Law of Thermo says that the opposite processes of these can’t proceed automatically. To take a system back to it’s initial state, External work must be done on it.

Spontaneous, Irreversible Processes: More Examples 3. Heat transfers from a high temperature object to a low temperature object. 4. 2 solutions of different concentrations are put together and mixed uniformly. • Note!! The 2 nd Law of Thermo says that the opposite processes of these can’t proceed automatically. To take a system back to it’s initial state, External work must be done on it.

Spontaneous Processes (Changes): • Once such a process begins, it proceeds automatically without the need to do work on the system. • The opposite of every Spontaneous Process is a Non-Spontaneous Process that can only proceed if external work is done on the system.

Reversible Processes (Idealizations!) • In a Reversible Process, the system undergoes changes such that the system plus it’s surroundings can be put back in their original states by exactly reversing the process. • In a Reversible Process, changes proceed in infinitesimally small steps, so that the system is infinitesimally close to equilibrium at every step. This is clearly an idealization & can never happen in a real system!

Other Statements of the 2 nd Law of Thermodynamics • “The entropy of the universe does not change for Reversible Processes” and also: • “The entropy of the universe increases for Spontaneous Processes” (“You can’t break even”). • For Reversible (ideal) Processes: • For Irreversible (real, spontaneous) Processes:

Still Other Statements of the 2 nd Law of Thermodynamics 1. “In any spontaneous process, there is always an increase in the entropy of the universe. ”

Still Other Statements of the 2 nd Law of Thermodynamics 1. “In any spontaneous process, there is always an increase in the entropy of the universe. ” Or 2. “The Total Entropy S of the Universe has the property that, for any spontaneous process ∆S ≥ 0 ”.

Example: Entropy of the Universe Increases • 1200 J of heat flows spontaneously through a copper rod from a hot reservoir at TH = 650 K to a cold reservoir at TC = 350 K. • Calculate the amount by which this irreversible process changes the entropy of the universe. (Assuming no other changes occur).
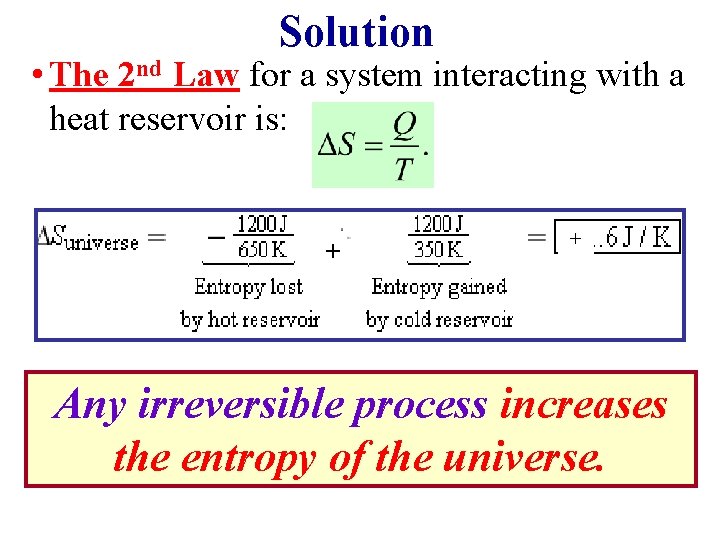
Solution • The 2 nd Law for a system interacting with a heat reservoir is: + + Any irreversible process increases the entropy of the universe.

More Examples of Spontaneous Processes Free Expansion of a Gas • The container on the right is filled with gas. • The container on the left is vacuum. The valve between them is closed. Now, imagine that the valve is opened. Valve Vacuum Closed Gas
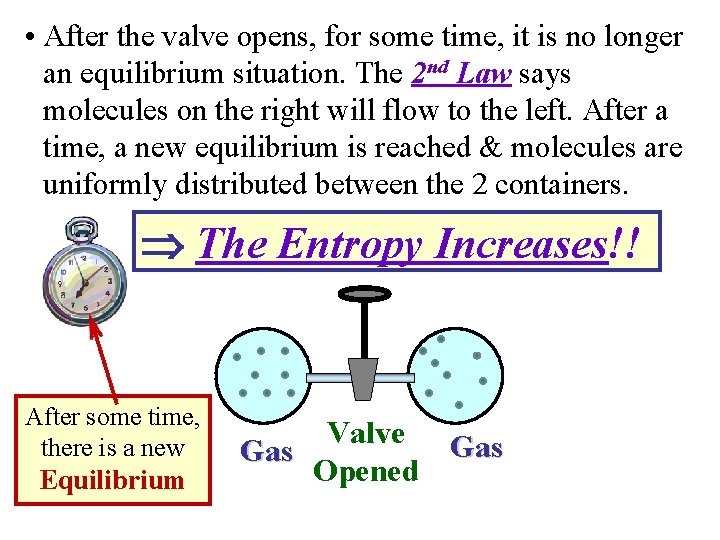
• After the valve opens, for some time, it is no longer an equilibrium situation. The 2 nd Law says molecules on the right will flow to the left. After a time, a new equilibrium is reached & molecules are uniformly distributed between the 2 containers. The Entropy Increases!! After some time, there is a new Equilibrium Valve Gas Opened

Thermal Conduction • A hot object (red) is brought into thermal contact with a colder object (blue). The 2 nd Law says that heat đQ will flow from the hot object to the colder object. Hot đQ Cold

• After the objects are in thermal contact, for some time, by the 2 nd Law, heat đQ flows from the hot object to the cold object. During that time, it isn’t an equilibrium situation. After a time, a new equilibrium is reached & the 2 objects are at the same temperature. The Entropy Increases!! After some time, there is a new Equilibrium Warm
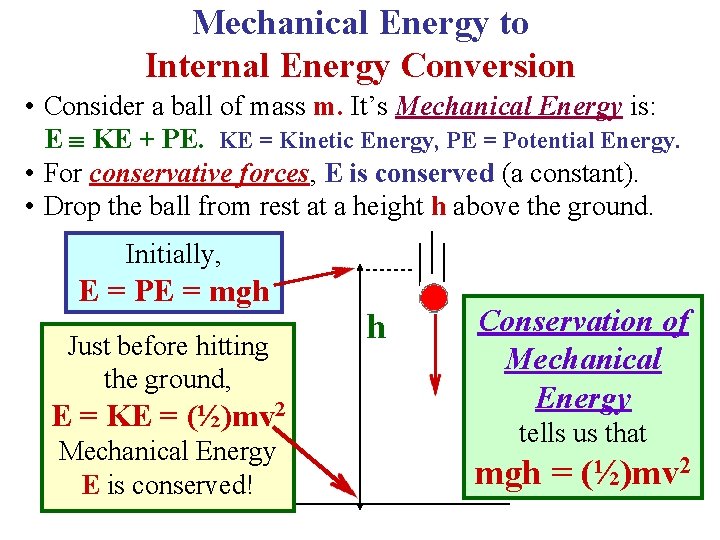
Mechanical Energy to Internal Energy Conversion • Consider a ball of mass m. It’s Mechanical Energy is: E KE + PE. KE = Kinetic Energy, PE = Potential Energy. • For conservative forces, E is conserved (a constant). • Drop the ball from rest at a height h above the ground. Initially, E = PE = mgh Just before hitting the ground, E = KE = (½)mv 2 Mechanical Energy E is conserved! h Conservation of Mechanical Energy tells us that mgh = (½)mv 2
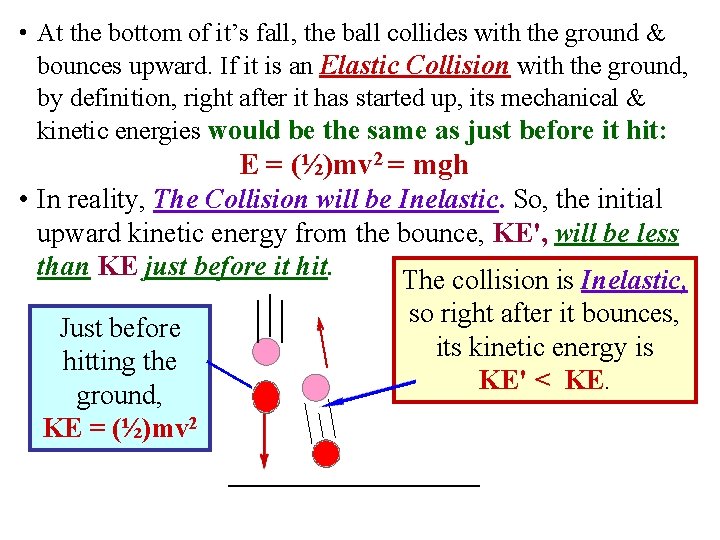
• At the bottom of it’s fall, the ball collides with the ground & bounces upward. If it is an Elastic Collision with the ground, by definition, right after it has started up, its mechanical & kinetic energies would be the same as just before it hit: E = (½)mv 2 = mgh • In reality, The Collision will be Inelastic. So, the initial upward kinetic energy from the bounce, KE', will be less than KE just before it hit. The collision is Inelastic, Just before hitting the ground, KE = (½)mv 2 so right after it bounces, its kinetic energy is KE' < KE.
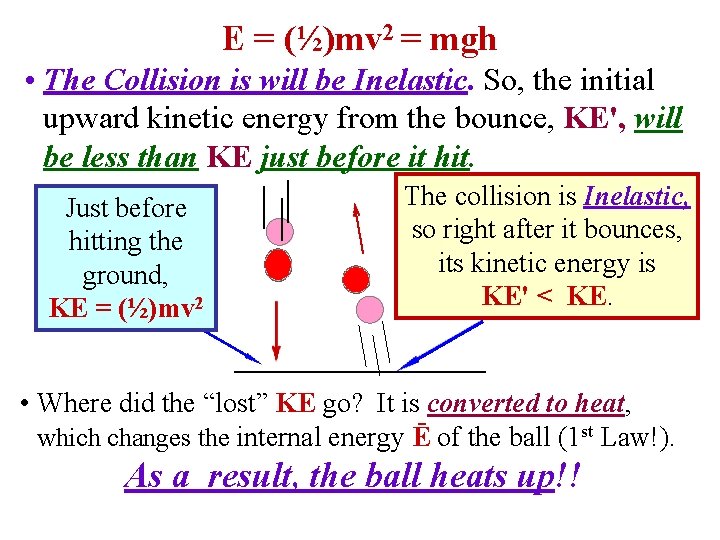
E = (½)mv 2 = mgh • The Collision is will be Inelastic. So, the initial upward kinetic energy from the bounce, KE', will be less than KE just before it hit. Just before hitting the ground, KE = (½)mv 2 The collision is Inelastic, so right after it bounces, its kinetic energy is KE' < KE. • Where did the “lost” KE go? It is converted to heat, which changes the internal energy Ē of the ball (1 st Law!). As a result, the ball heats up!!

• The ball’s has an inelastic collision with the ground, so it loses some kinetic energy: KE' < KE. • The lost kinetic energy is converted to heat, which changes the ball’s internal energy Ē. So, the ball gets warmer!! • In Ch. 4, we’ll show that, for quasi-static process in which an object heats up, changing its temperature by an amount T, it’s internal energy change is Ē = mc. V T so the ball heats up! m ≡ ball mass c. V ≡ specific heat at constant volume. KE' < KE KE = (½)mv 2
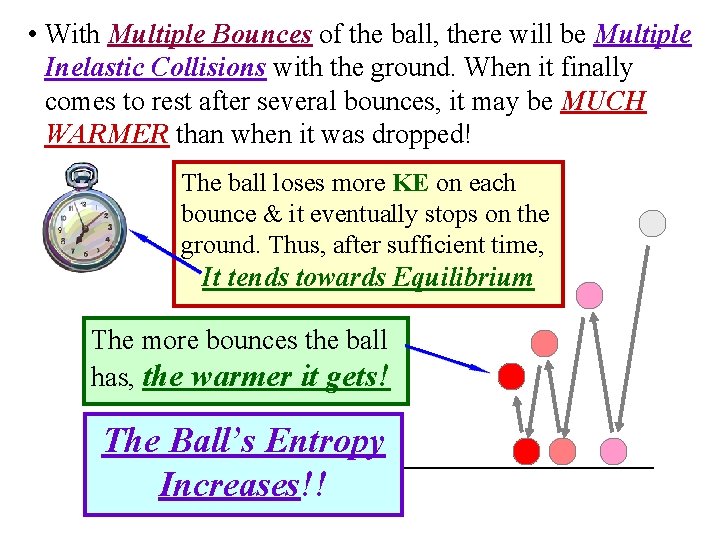
• With Multiple Bounces of the ball, there will be Multiple Inelastic Collisions with the ground. When it finally comes to rest after several bounces, it may be MUCH WARMER than when it was dropped! The ball loses more KE on each bounce & it eventually stops on the ground. Thus, after sufficient time, It tends towards Equilibrium The more bounces the ball has, the warmer it gets! The Ball’s Entropy Increases!!

Irreversible (Spontaneous) Processes • An object can slide down an incline if friction is overcome. • But it can’t spontaneously move up the incline of its own accord. The conversion of mechanical energy to thermal energy by friction as it slides is irreversible. • In the figure, an ice cube at T = 0 C sits for some time, gradually melting. Eventually, only a puddle of water will remain.

More Examples of Spontaneous Processes • Spontaneous processes occur in a system left to itself. • No action from outside the system is necessary to bring the change about.

More Examples of Spontaneous Processes • Spontaneous processes occur in a system left to itself. No action from outside the system is necessary to bring the change about. Example: Dissolving a Solid in a Liquid (like salt in water) • Ions have more entropy (more states) than the water, • But, some water molecules have less entropy (they’re grouped around ions). There must be an overall increase in entropy.

More Examples of Spontaneous Processes • Spontaneous processes occur in systems left to themselves. No action from outside the system is necessary to cause the change. • Question: Water put into a freezer spontaneously turns to ice. (So, the water entropy decreases!) • Entropy always increases, so, how can we account for this?

• Question: Water put into a freezer spontaneously turns to ice. (So, the water entropy decreases!) Why? • Doesn’t this violate the 2 nd Law of Thermo?

• Question: Water put into a freezer spontaneously turns to ice. (So, the water entropy decreases!) Why? • Doesn’t this violate the 2 nd Law of Thermo? Answers • The compressor does work on the ice + freezer. • This causes evaporation & condensation of the refrigerant. This also causes warming of the air around the container As a result of these effects, the entropy of the universe will increase.

Some Processes That Lead to an Increase in Entropy (Spontaneous Processes) 1. Melting of a solid. 2. Dissolving of a solid in a solution. 3. A solid or a liquid becomes a gas. 4. The temperature of a substance increases. 5. A chemical reaction produces more molecules.

FYI: Brief Discussion of “Impossible Processes” • Impossible Processes are those which would be Allowed by the 1 st Law of Thermo but which Can’t Occur Naturally because they would violate the 2 nd Law of Thermo.

Brief Discussion of “Impossible Processes” • Impossible Processes are those which would be Allowed by the 1 st Law of Thermo but which Can’t Occur Naturally because they would violate the 2 nd Law of Thermo. • Any process which would take a system from an equilibrium state to a nonequilibrium state without work being done on the system would violate the 2 nd Law & thus would be an “Impossible Process”!
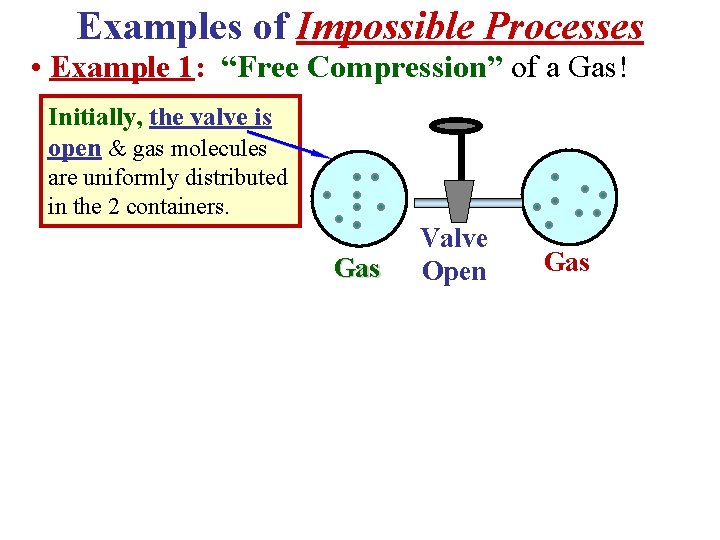
Examples of Impossible Processes • Example 1: “Free Compression” of a Gas! Initially, the valve is open & gas molecules are uniformly distributed in the 2 containers. Gas Valve Open Gas
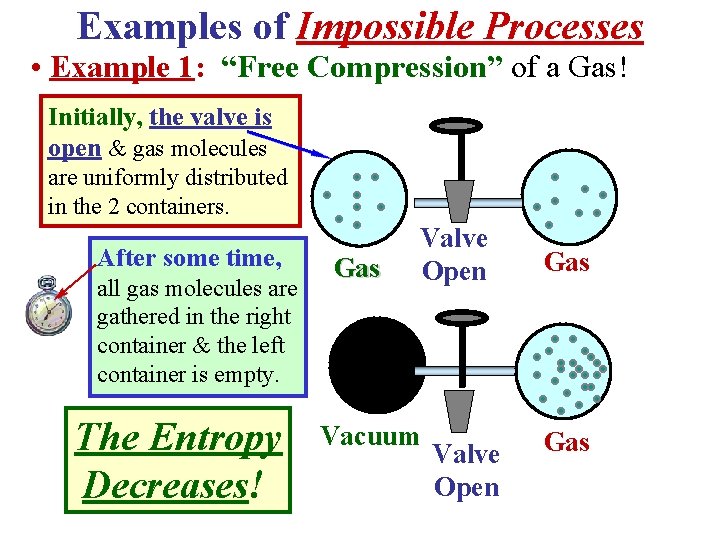
Examples of Impossible Processes • Example 1: “Free Compression” of a Gas! Initially, the valve is open & gas molecules are uniformly distributed in the 2 containers. After some time, all gas molecules are gathered in the right container & the left container is empty. The Entropy Decreases! Gas Valve Open Vacuum Valve Open Gas
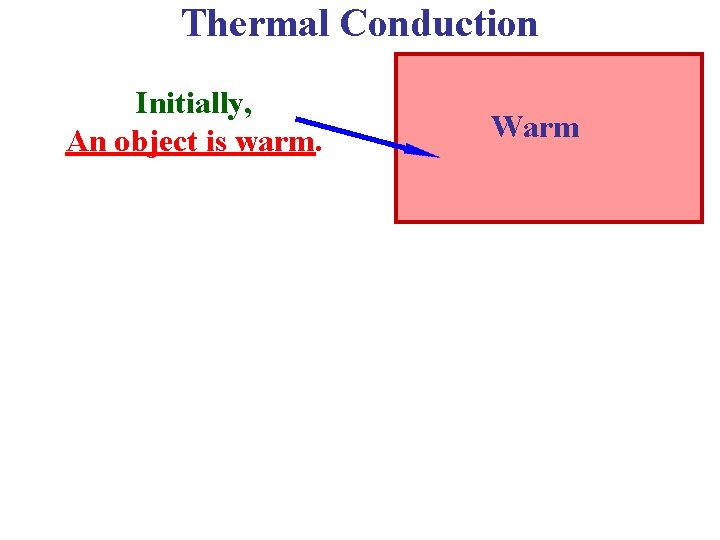
Thermal Conduction Initially, An object is warm. Warm
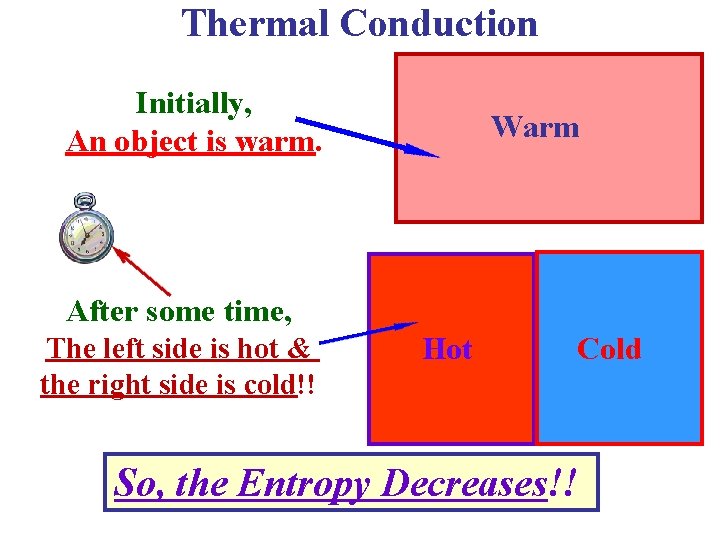
Thermal Conduction Initially, An object is warm. Warm After some time, The left side is hot & the right side is cold!! Hot Cold So, the Entropy Decreases!!
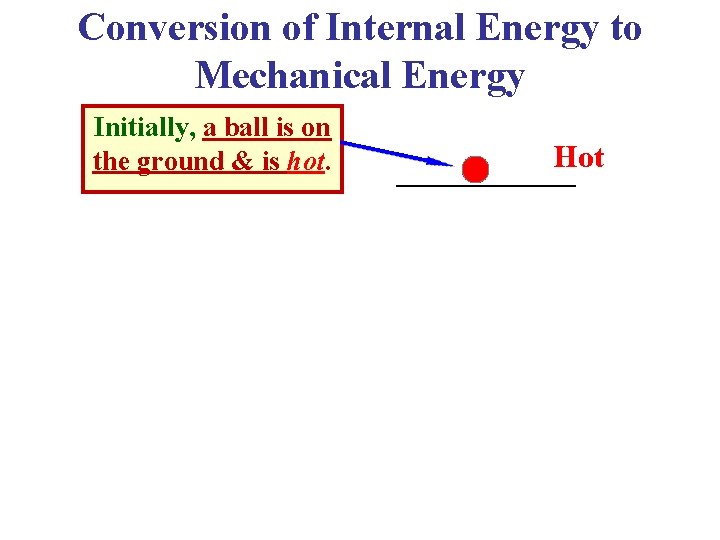
Conversion of Internal Energy to Mechanical Energy Initially, a ball is on the ground & is hot. Hot
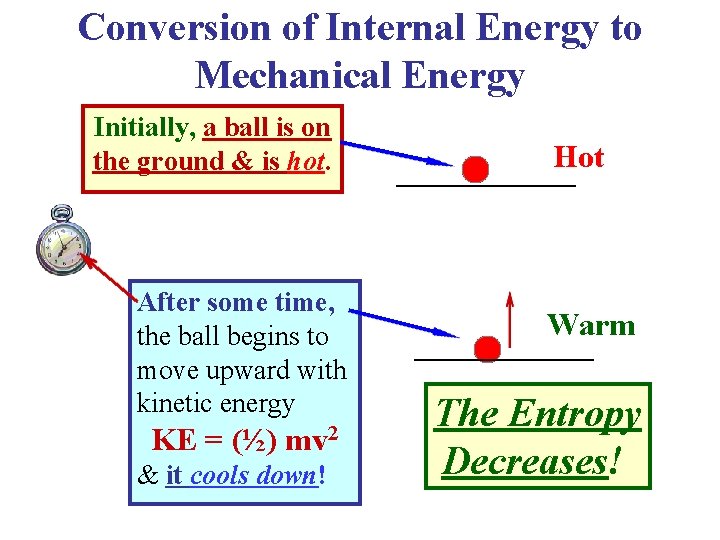
Conversion of Internal Energy to Mechanical Energy Initially, a ball is on the ground & is hot. After some time, the ball begins to move upward with kinetic energy KE = (½) mv 2 & it cools down! Hot Warm The Entropy Decreases!

“Impossible Processes” Cannot occur without the input of work đW
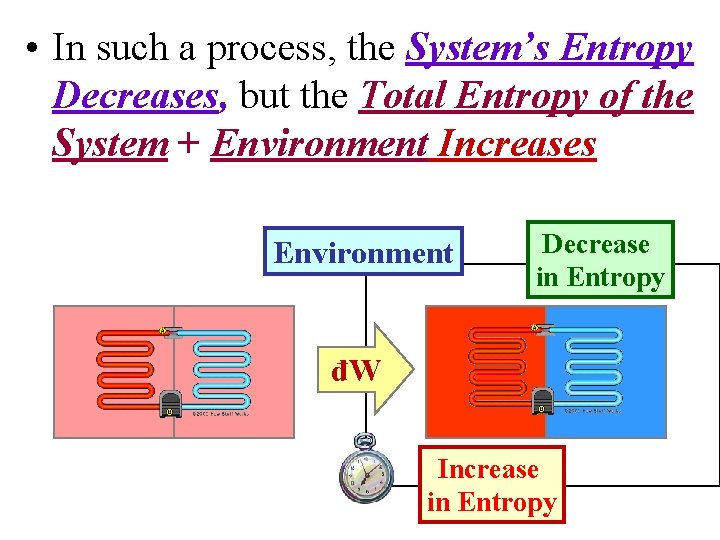
• In such a process, the System’s Entropy Decreases, but the Total Entropy of the System + Environment Increases Environment Decrease in Entropy đW Increase in Entropy

FYI: Very Brief Discussion: “Perpetual Motion” • We’ve said, “Impossible Processes” are processes which are Allowed by the 1 st Law of Thermo but which Can’t Occur Naturally because they would violate the 2 nd Law of Thermo.

FYI: Very Brief Discussion: “Perpetual Motion” • We’ve said, “Impossible Processes” are processes which are Allowed by the 1 st Law of Thermo but which Can’t Occur Naturally because they would violate the 2 nd Law of Thermo. • Any process which takes a system from an equilibrium state to a non-equilibrium state without work being done on the system Would violate the 2 nd Law of Thermo & thus Would be an Impossible Process!

• “Impossible Processes” are processes which are Allowed by the 1 st Law of Thermo but which Can’t Occur Naturally because they would violate the 2 nd Law of Thermo. • As examples which we sometimes hear about, the so-called “Perpetual Motion” or “Free Energy” machines rely on such “Impossible Processes” & thus are Always Bogus!

What is “Perpetual Motion”? • That term describes hypothetical machines that operate or produce useful work indefinitely &, more generally, hypothetical machines that produce more work or energy than they use. • There is an undisputed scientific consensus that Perpetual motion would violate either the 1 st Law or the 2 nd Law of Thermodynamics, OR BOTH! • Such machines rely on “Impossible Processes” & thus are Always Bogus!

“Perpetual Motion” • Describes a theoretical machine that, without any losses due to friction or other forms of dissipation of energy, would continue to operate indefinitely at the same rate without any external energy being applied to it. • Machines which comply with both the 1 st & 2 nd Laws of Thermodynamics but access energy from obscure sources are also sometimes referred to as “Perpetual Motion” machines, though they do not meet the standard criteria for the name.

“Perpetual Motion Machines” & the US Patent & Trademark Office • For over 100+ years now, the US PTO has had a standing rule: If a patent application is filed which claims to be about a “Perpetual Motion Machine” or a “Free Energy Machine”, etc. , it will be rejected without further review.
Evidence: Amazon Search: “Free Energy"

Brief Overview: Classification of “Perpetual Motion” Machines! 1. “Perpetual Motion” Machine of the 1 st Kind 2. “Perpetual Motion” Machine of the 2 nd Kind 3. “Perpetual Motion” Machine of the 3 rd Kind

The 1 st Kind • A “perpetual motion” machine of the first kind produces work without the input of energy. It thus violates the 1 st Law of Thermodynamics: the Law of Conservation of Energy. • The law of conservation of energy is an empirical law of physics. It states that the total amount of energy in an isolated system remains constant over. • A consequence of this law is that energy can neither be created nor destroyed: it can only be transformed from one state to another. So, It is clearly impossible for a machine to do work indefinitely without consuming energy.

The 2 nd Kind • A “perpetual motion” machine of the 2 nd kind is a machine which spontaneously converts thermal energy into mechanical work. • When thermal energy is equivalent to the work done, this does not violate the law of conservation of energy. However, it does violate the 2 nd Law of Thermo! • The signature of a perpetual motion machine of the second kind is that there is only one heat reservoir involved, which is being spontaneously cooled without involving a transfer of heat to a cooler reservoir. This conversion of heat into useful work, without any side effect, is impossible, according to the second law of thermodynamics.

The 3 rd Kind • A “perpetual motion” machine of the 3 rd kind is a machine which relies on the Complete Elimination of Friction & other dissipative forces, to maintain motion forever. • Technically, it is not so much an energy generating machine so much as a machine that operates without consuming energy. It is not possible to move with zero friction, so although low friction devices are real, zero friction devices are not. ch as an energy storage device.
- Slides: 60