Materials of Electrochemical Equipment Their degradation and Corrosion


















- Slides: 26

Materials of Electrochemical Equipment, Their degradation and Corrosion Summer school on electrochemical engineering, Palic, Republic of Serbia Prof. a. D. Dr. Hartmut Wendt, TUD

Material Choices • Metals (steels) as conventional selfsupporting materials for electrodes, electrolyzer troughs, gas – pipes and bipolar plates • Ionomers for diaphragms • Polymers as insulating materials

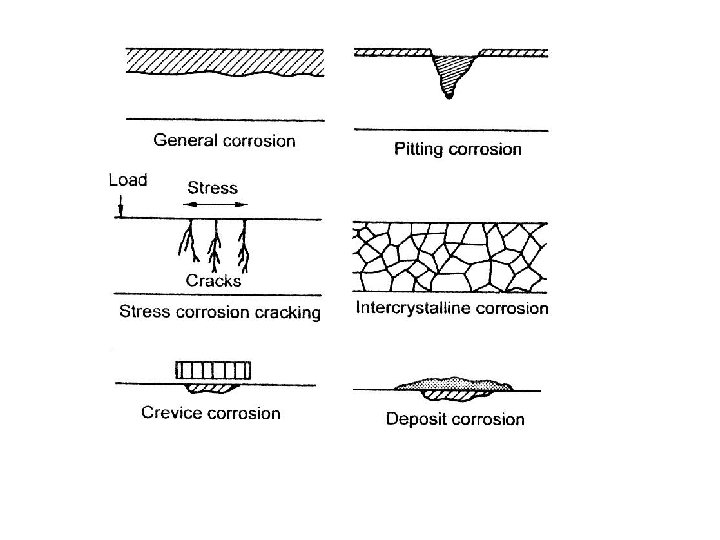
Metals • CORROSION • Mechanical wear and erosion • High temperature sintering and granule growth • High temperature surface oxidation and internal oxidation of non noble constituents

Polymers and Ionomers • Bon breaking by oxidation (oxygen and peroxides) • Reduction ( lower valent metal ions, hydrogen) • Solvolysis (preferentially hydrolysis) by acids and bases. • Particular for Ionomer membranes (MEAs) is delamination

Carbon A special story of its own

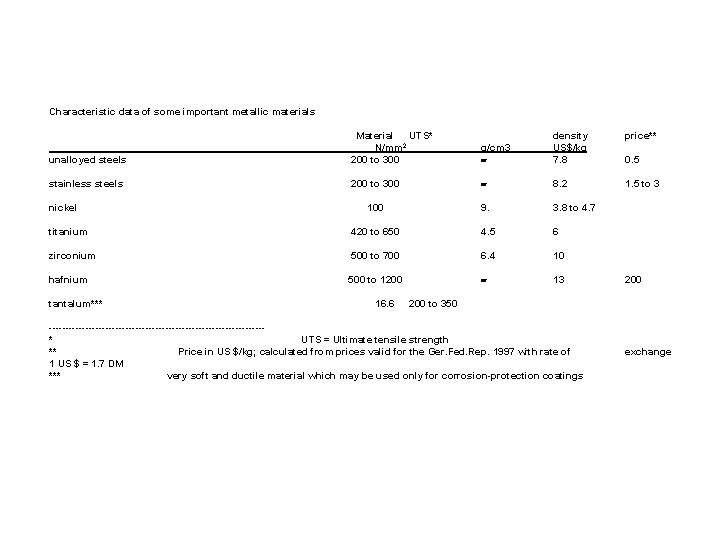
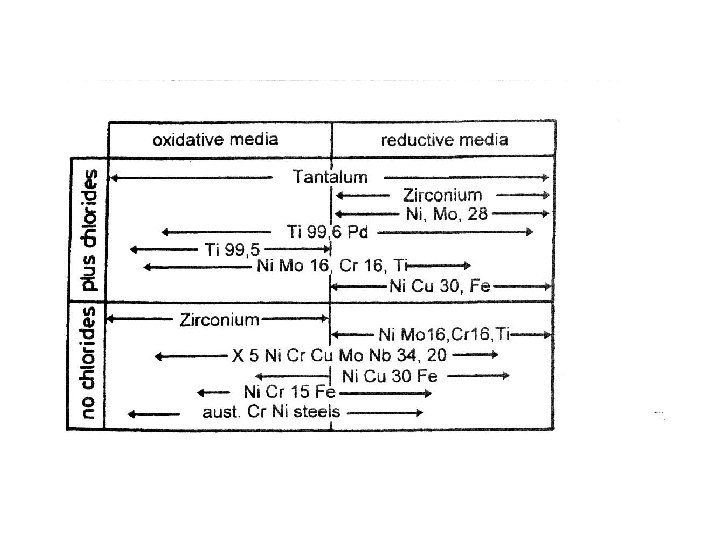
Characteristic data of some important metallic materials g/cm 3 density US$/kg 7. 8 price** unalloyed steels Material UTS* N/mm 2 200 to 300 stainless steels 200 to 300 8. 2 1. 5 to 3 100 9. 3. 8 to 4. 7 titanium 420 to 650 4. 5 6 zirconium 500 to 700 6. 4 10 hafnium 500 to 1200 13 nickel tantalum*** 16. 6 0. 5 200 to 350 --------------------------------* UTS = Ultimate tensile strength ** Price in US $/kg; calculated from prices valid for the Ger. Fed. Rep. 1997 with rate of 1 US $ = 1. 7 DM *** very soft and ductile material which may be used only for corrosion-protection coatings exchange

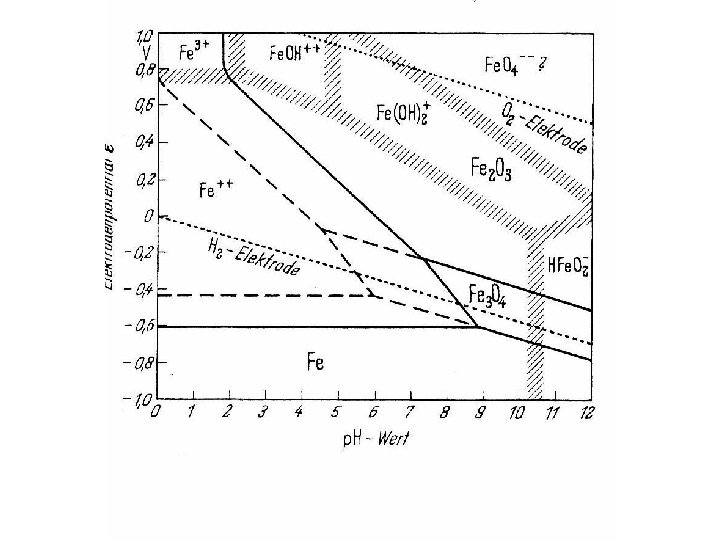
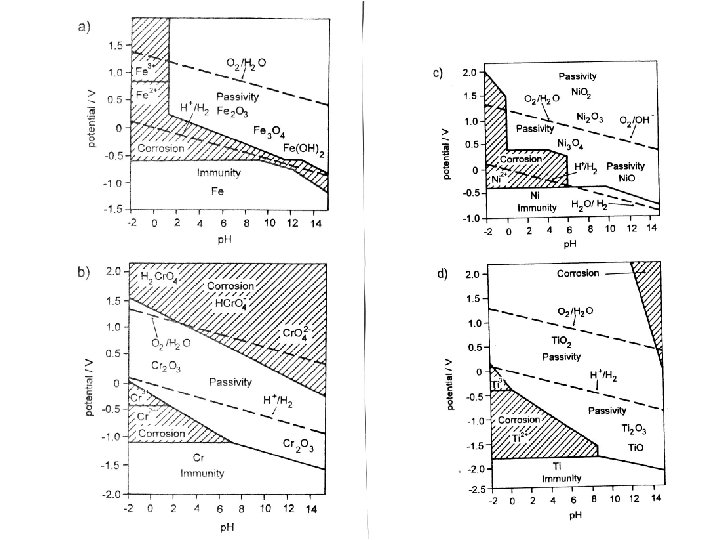
p. H-potential (Pourbaix) diagrams A diagnostic thermodynamic tool Identifying existing phases as Condition for potential passivity



What tells the Pourbaix diagram ? • Iron might become passive at O 2 – potential and at p. H beyond 2. It will never be immune. • Nickel is immune at p. H greater 8 in presence of hydrogen, but there is only a reserve of 80 m. V • Chromium (and steels with Cr) is never immune but might become passive • Titanium is never immune but might become passive over total p. H – range and potentials more positive than RHE.



High temperatures and Metals • High temperatures (> 600 o. C), and longterm exposure in HT – fuel cells would lead to total oxidation on oxygen side (exception is only gold). • Fe-containing alloys might become passive because of formation of protective oxide layers from alloy components (W, Mo, Cr. Al and other). • Internal oxidation by oxygen diffusion into metals and preferential oxidation of non-noble components can change internal structure (dispersion hardening) • On hydrogen side there might occur hydrogenembrittlement (Ti, Zr)

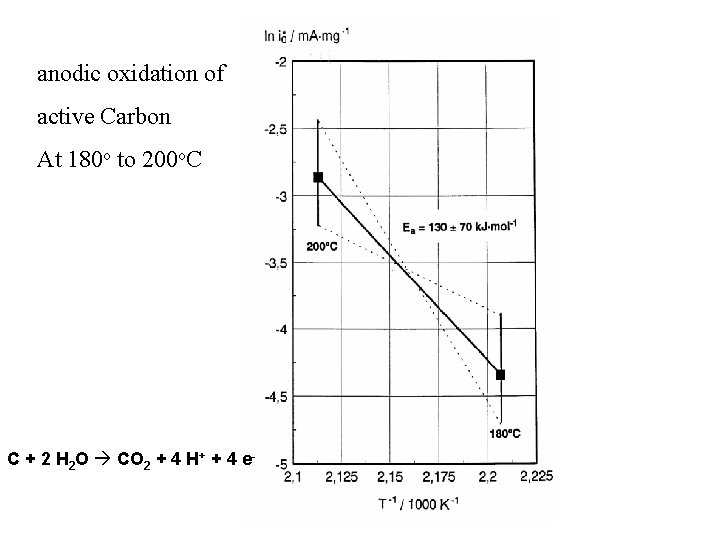
Carbon in Fuel Cells • The element carbon is not nobler than hydrogen. • It is unstable against atmospheric and anodic oxidation in particular at enhanced temperature (PAFC: 220 o. C) • At still higher temperature it also becomes unstable towards steam (C+H 20 ->CO+H 2)

anodic oxidation of active Carbon At 180 o to 200 o. C C + 2 H 2 O CO 2 + 4 H+ + 4 e-

Polymers and Ionomers Properties and deterioration

* Price in Germany mid 1997. Rate of exchange: 1 US $ equal 1. 7 DM, Source: Kunststoff Information (KI), D - 61350 Bad Homburg

Non – Fluorinated Polymers • May only be used with non – oxidizing electrolytes and atmospheres • Very often need glass-fiber enforcement • Chlorinated and perchlorinated polymers are chemically more stable than non-chlorinated polymers • Polyesters and amides are sensitive against hydrolysis in strongly acid and caustic electrolyte • They are cheaper than fluorinated polymers Polystyrenes are not acceptable for Fuel cells and electrolyzers

Fluorinated Polymers • Perfluorinated Polymers (Teflon. TM) are most stable polymers • They are soft and tend to creep and flow • Polyvinyliden-fluoride tends to stresscorrosion-cracking at elevated temperature in contact to acid soltutions (For details look at DECHEMA- WERKSTOFFTABELLEN)

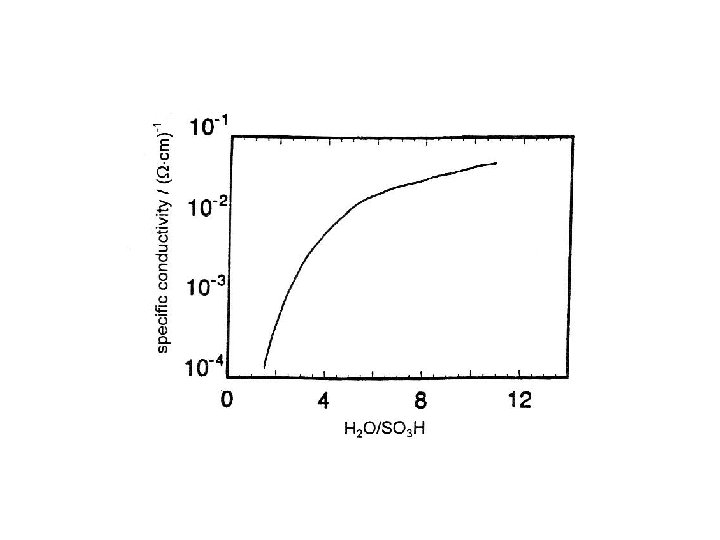
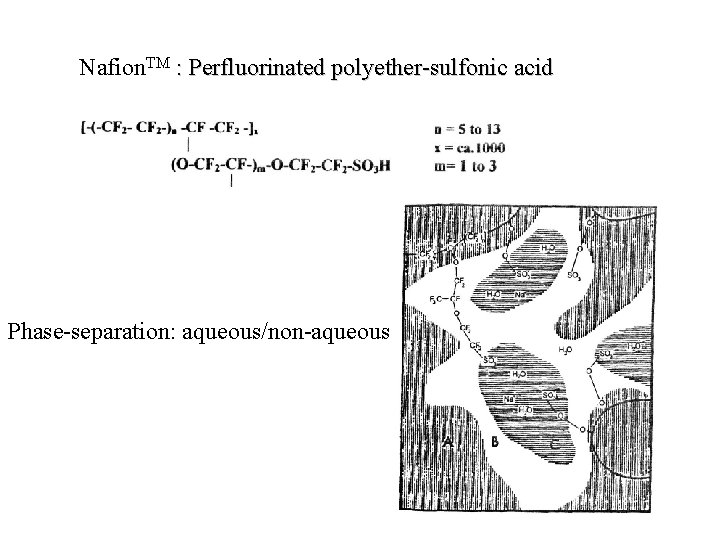
Ionomers – Ion-exchange membranes • In batteries non-fluorinated ion-exchange membranes are sometimes used as separators – but are usually too expensive • Nafion. TM had been developed for the cloroalkali electroysis and had become the material of choice for fuel cells (PEMFC) • Weakness: High water transfer; at least 4 H 2 O per H+ transferred (also methanol)


Nafion. TM : Perfluorinated polyether-sulfonic acid Phase-separation: aqueous/non-aqueous

Anion exchange membranes are chemically less stable

Delamination of MEAs • Reason: Weak contact between prefabricated PEM and PEM-bonded elctrocatalyst layer • Lifetime of MEAs can be extended steady fuel cell operation, because repeated hydration/dehydration with subsequent change of degree of swelling exerts stress on the bond between membrane and catalyst

NEW membrane materials • Aim: reduce swelling, water and methanol or ethanol transport, improve durability of contact between membrane and catalyst layer • Sulfonated polyaryls, polyethetherketones (PEEKs) and Polyaryl-sulfones (all new PEM-materials are sulfonic acids)

Summary The electrochemical engineer needs not to be an expert in material science but he needs to know when to go and ask material scientists