Engineering Chemistry revised edition ISBN 978 81 265

Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Corrosion in Metals and Alloys The destruction of metals or alloys by surrounding environment through chemical or electrochemical changes. Causes of Corrosion is the deterioration of materials by chemical interaction with their environment. The term corrosion is sometimes also applied to the degradation of plastics, concrete and wood, but generally refers to metals. The most familiar example of corrosion is rusting of iron exposed to the atmospheric conditions. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Loss of metal Loss of time Reduction of value of goods Effects of Corrosion Contamination of fluids due to chemical reaction Changes in surface properties Mechanical damage Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
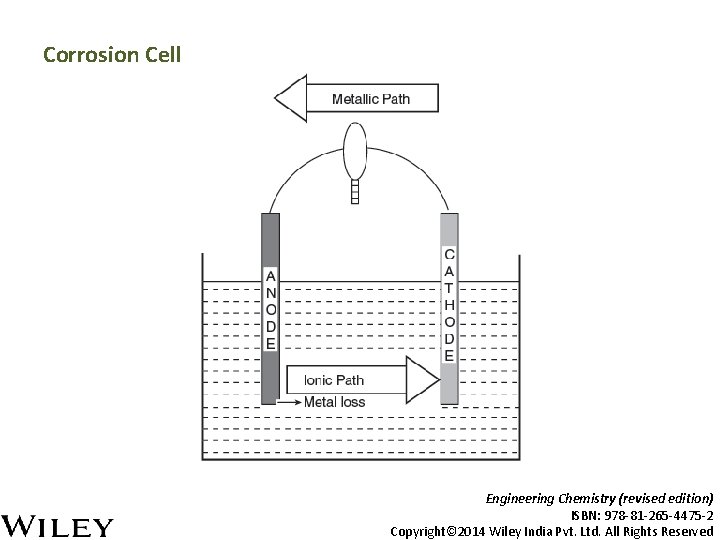
Corrosion Cell Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Types of Corrosion Dry Corrosion (Direct Chemical Attack) There are three main types of dry corrosion. 1. Oxidation corrosion (reaction with oxygen): The overall reaction can be given by Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
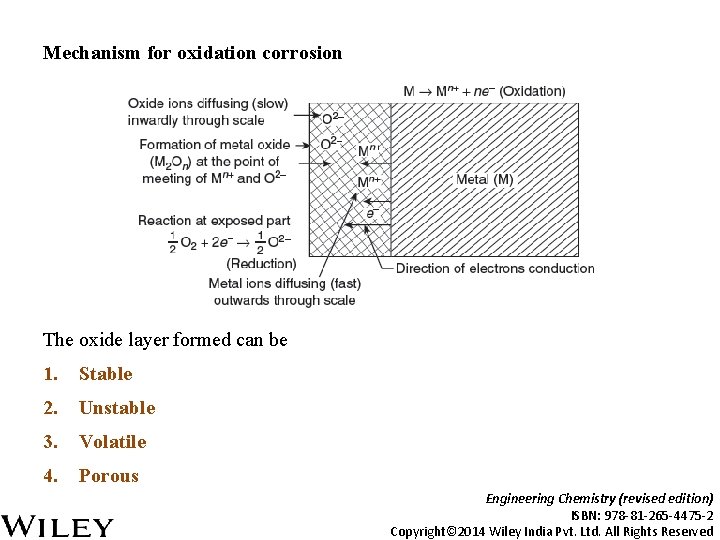
Mechanism for oxidation corrosion The oxide layer formed can be 1. Stable 2. Unstable 3. Volatile 4. Porous Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

2. Corrosion by other gases such as Cl 2, SO 2, H 2 S, NOx: In dry atmosphere, these gases react with metal and form corrosion products, which may be protective or non-protective. Dry Cl 2 reacts with Ag and forms Ag. Cl which is a protective layer, while Sn. Cl 4 is volatile. In petroleum industries at high temperatures, H 2 S attacks steel forming Fe. S scale which is porous and interferes with normal operations. 3. Liquid metal corrosion: In several industries, molten metal passes through metallic pipes and causes corrosion due to dissolution or internal penetration. For example, liquid metal mercury dissolves most metals by forming amalgams, thereby corroding them. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
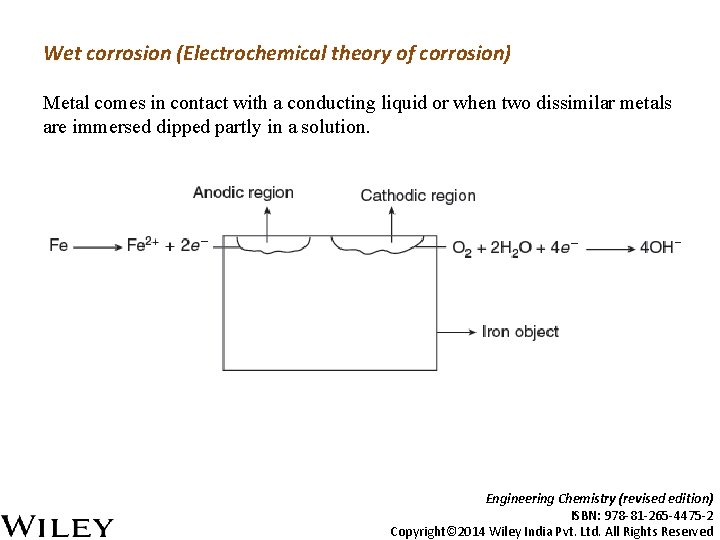
Wet corrosion (Electrochemical theory of corrosion) Metal comes in contact with a conducting liquid or when two dissimilar metals are immersed dipped partly in a solution. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

The reactions involved in the galvanic cell are as follows: 1. Anodic reaction: Metal undergoes oxidation (corrosion) with positive release of electrons: 2. Cathodic reactions: These reactions are dependent on the constituents of the corrosion medium. • If the solution is aerated and almost neutral, oxygen is reduced in presence of H 2 O to OH− ions. • If the solution is deaerated and almost neutral, H 2 is liberated along with OH− ions. • If the solution is deaerated and acidic, H+ ions are reduced to hydrogen gas Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

3. The metal ions formed at the anode combine with hydroxyl ions and form the corresponding metallic hydroxide Fe(OH)2, which further gets oxidized to hydrated ferric oxide [rust]. In the presence of limited oxygen, black rust is formed as follows: Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
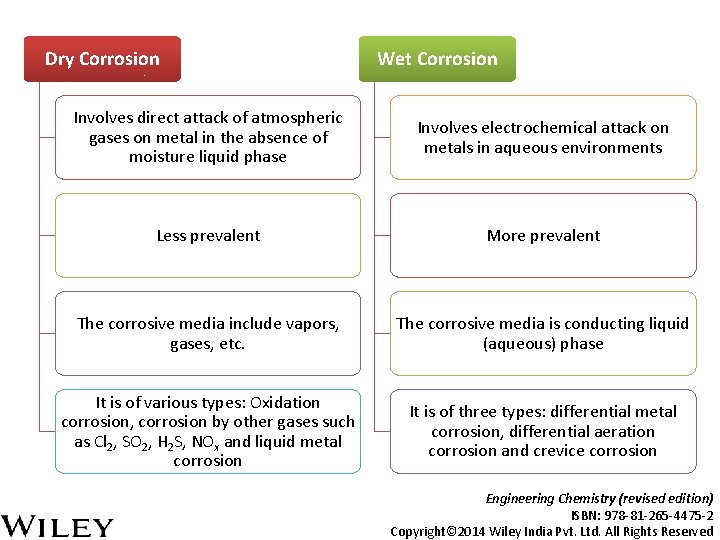
Dry Corrosion Wet Corrosion Involves direct attack of atmospheric gases on metal in the absence of moisture liquid phase Involves electrochemical attack on metals in aqueous environments Less prevalent More prevalent The corrosive media include vapors, gases, etc. The corrosive media is conducting liquid (aqueous) phase It is of various types: Oxidation corrosion, corrosion by other gases such as Cl 2, SO 2, H 2 S, NOx and liquid metal corrosion It is of three types: differential metal corrosion, differential aeration corrosion and crevice corrosion Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Types of Electrochemical Corrosion Differential Metal Corrosion (Galvanic Corrosion) When two dissimilar metals are in contact in corrosive environment, metal with lesser standard reduction potential will act as anode and will undergo oxidation (corrosion). Metal with higher standard reduction potential will act as cathode and it will provide surface for reduction of oxygen. . Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Other examples of differential metal corrosion (galvanic corrosion) include the following: 1. Buried iron pipeline connected to zinc bar. 2. Steel pipe connected to copper plumbing. 3. Steel propeller shaft in bronze bearing. 4. Zinc coating on mild steel. 5. Lead–tin solder around copper wires. Preventive Measures 1. Placing a thin layer of an insulator between two metals or materials. 2. Selecting materials having very less potential difference. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Differential Aeration Corrosion (Concentration Cell Corrosion) When metal is exposed to differential air or oxygen concentration. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

The examples of differential aeration corrosion include: 1. Half-immersed iron plate in aqueous solution. 2. Steel pipe carrying any liquid exposed to atmosphere. 3. Ocean going ships. 4. Steel storage tanks. Preventive Measures 1. Using metallic coating, electroless plating or chemical conversion methods. 2. Maintaining the material’s own protective fi lm. 3. Controlling the chemistry of fluids and using inhibitors. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
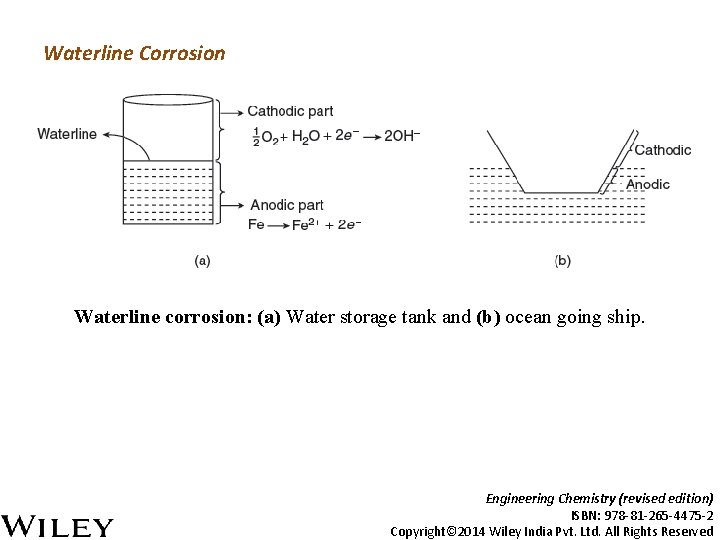
Waterline Corrosion Waterline corrosion: (a) Water storage tank and (b) ocean going ship. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
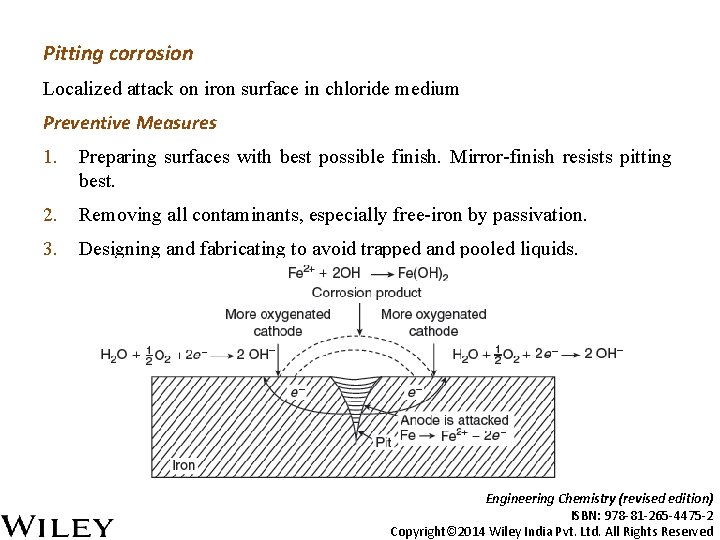
Pitting corrosion Localized attack on iron surface in chloride medium Preventive Measures 1. Preparing surfaces with best possible finish. Mirror-finish resists pitting best. 2. Removing all contaminants, especially free-iron by passivation. 3. Designing and fabricating to avoid trapped and pooled liquids. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

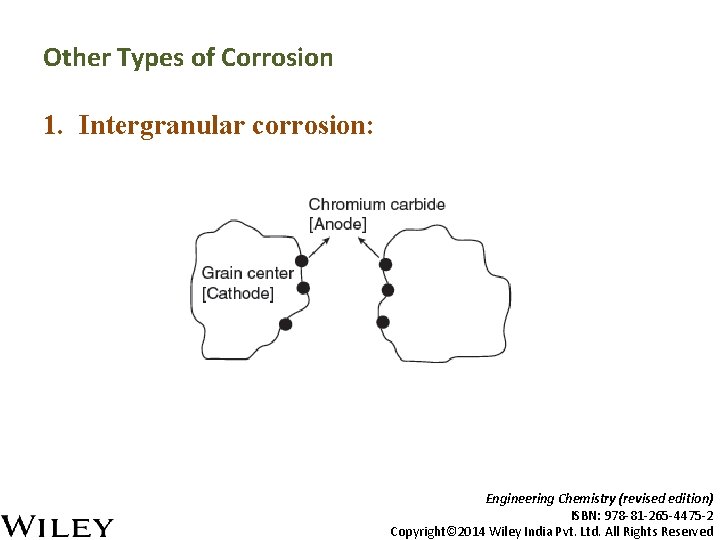
Other Types of Corrosion 1. Intergranular corrosion: Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

2. Soil corrosion: Underground pipes, cables, tank bottoms, etc. , get corroded due to moisture, p. H of soil, ionic species like chlorides and microorganisms like bacteria. It is further enhanced by differential aeration of various parts of the soil. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
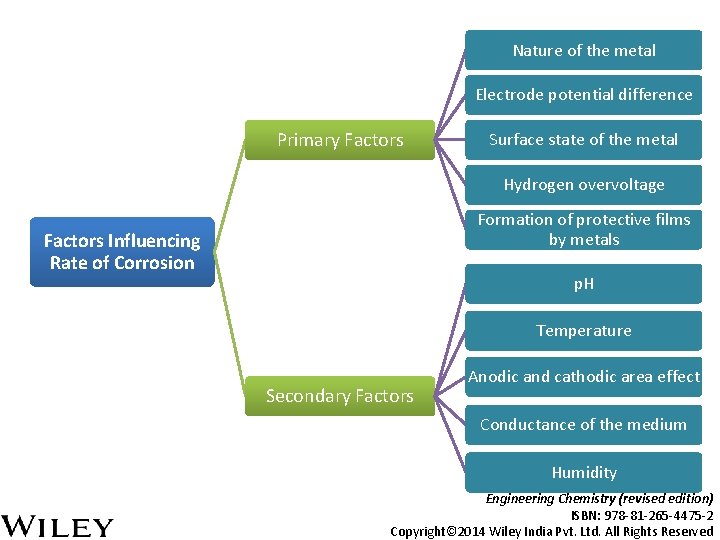
Nature of the metal Electrode potential difference Primary Factors Surface state of the metal Hydrogen overvoltage Formation of protective films by metals Factors Influencing Rate of Corrosion p. H Temperature Secondary Factors Anodic and cathodic area effect Conductance of the medium Humidity Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
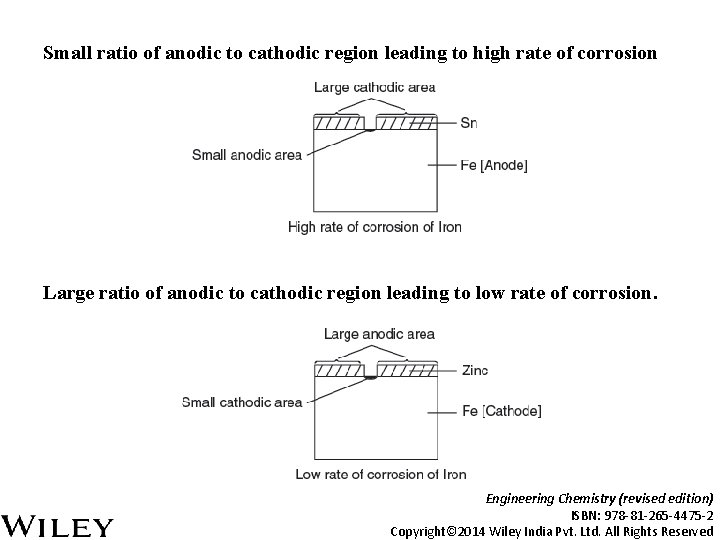
Small ratio of anodic to cathodic region leading to high rate of corrosion Large ratio of anodic to cathodic region leading to low rate of corrosion. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
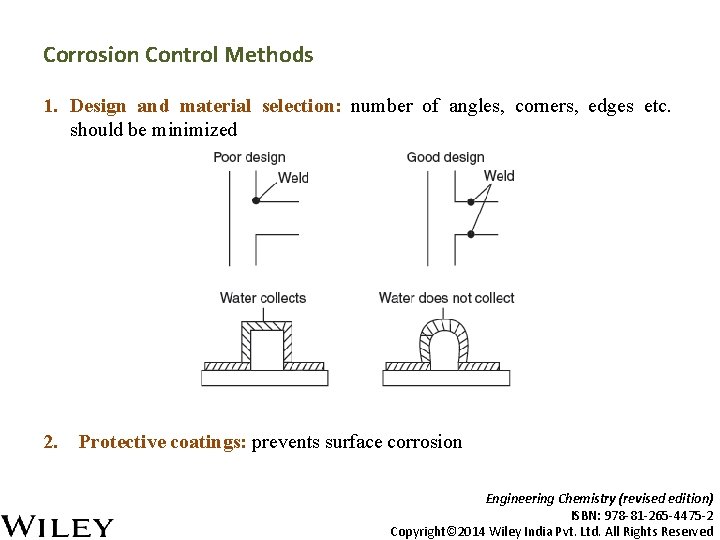
Corrosion Control Methods 1. Design and material selection: number of angles, corners, edges etc. should be minimized 2. Protective coatings: prevents surface corrosion Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
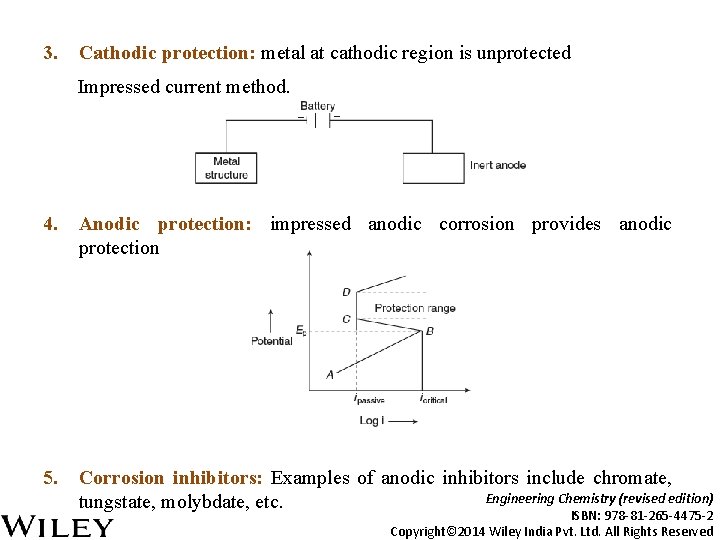
3. Cathodic protection: metal at cathodic region is unprotected Impressed current method. 4. Anodic protection: impressed anodic corrosion provides anodic protection 5. Corrosion inhibitors: Examples of anodic inhibitors include chromate, Engineering Chemistry (revised edition) tungstate, molybdate, etc. ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Corrosion: Deterioration of metal by electrochemical process e. g. Rust on iron, due to formation of hydrated ferric oxide i. e. Fe 2 O 3. x. H 2 O Tarnish on silver Green Patina on Cu ØCorrosion causes enormous damage to building, bridges, ships and cars. 25

Fundamental Process in Corrosion Conditions for rusting: Oxygen gas and water Electrochemical reaction: Anode: Metal surface Fe(s) → Fe 2+ (aq) + 2 e- E 0 red = -0. 44 V Cathode: Another region of same metal surface O 2( g) + 4 H+ + 4 e- → 2 H 2 O (E 0 red = 1. 23 V Overall reaction: 2 Fe(s) + O 2 (g) + 4 H+ (aq) → 2 Fe 2+ (aq) + 2 H 2 O E 0 cell = Ec – EA = 1. 23 – (-0. 44) = 1. 67 V 4 Fe 2+ (aq) +O 2 (g) + (4+2 x)H 2 O(l) → 2 Fe 2 O 3. x. H 2 O(s) +8 H+ 26 Rust

Corrosion Prevention Ø Barrier Protection: Coating with paint ØAlloy: Stainless steel i. e. Alloy of Fe-Cr Ø Sacrificial Protection: By covering Fe(s) with more active metal. Such as Zn and Sn e. g. Fe surface covered with Zn metal Zn → -Zn 2+(aq) + 2 e. E 0 red = -0. 76 Fe → Fe 2+ (aq) + 2 e. E 0 red = -0. 44 The Process of covering Fe with Zn is known as Galvanization. Sn → Sn 2+ + 2 e- Fe → Fe 2+ (aq) + 2 e- E 0 red = -0. 14 E 0 red = -0. 44 27

Corrosion Prevention Ø Electrical/Cathodic Protection: Fe is connected with more active metal Like Mg and Zn. e. g. Under ground water pipes Fe(s) → Fe 2+ (aq) + 2 e- E 0 red = - 0. 44 V Zn (s) → -Zn 2+(aq) + 2 e- E 0 red = - 0. 76 V Mg (s) → Mg 2+(aq) + 2 e- E 0 cell = - 2. 37 V 28

Corrosion on other metals Aluminum: Al 2 O 3 layer will formed Copper: Cu. CO 3 Silver: Ag 2 S
- Slides: 29