CORROSION Mr Kisan Raghunath Bobe Assistant Professor Pharmaceutics

CORROSION Mr. Kisan Raghunath Bobe Assistant Professor (Pharmaceutics) JSPM’S Jayawantrao Sawant College of Pharmacy and Research, Pune.

INTRODUCTION • Corrosion is a process of reaction between metallic surface and its environment. • It is an oxidation process. • It causes loss of metal. • Hence, disintegration of a metal by its surrounding chemicals through a chemical reaction on the surface of the metal is called corrosion

• Example: Formation of rust on the surface of iron, formation of green film on the surface of copper. • The responsible factors for the corrosion of a metal the environmental chemicals, temperature and the design. 3

DIFFERENT THEORIES OF CORROSION There are three theories of corrosion: • Dry or chemical corrosion theory • Wet or galvanic or electrochemical theory 4

DRY CORROSION THEORY • According to this theory, corrosion on the surface of a metal is due to direct reaction of gases like atmospheric oxygen, halogens, oxides of sulphur, oxides of nitrogen, hydrogen sulphide and fumes of chemicals with metal. • Depends on chemical affinity of the metal towards reactive gas. 5
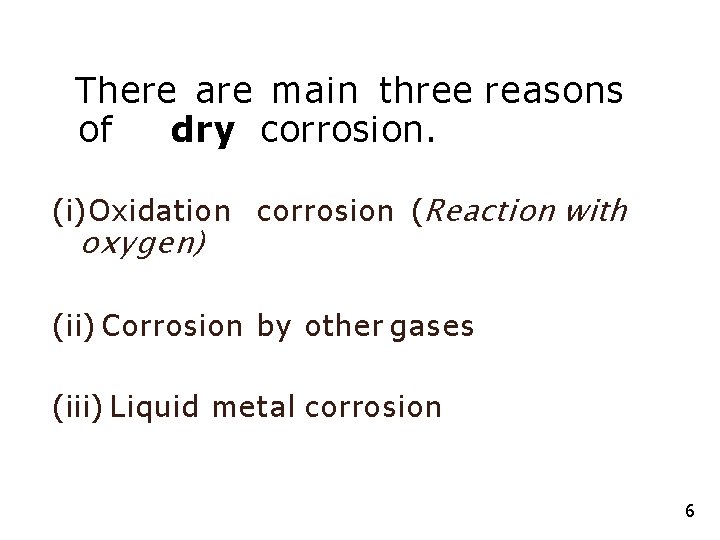
There are main three reasons of dry corrosion. (i) Oxidation corrosion (Reaction with oxygen) (ii) Corrosion by other gases (iii) Liquid metal corrosion 6

(i) Oxidation corrosion (Reaction with oxygen) • Some of the metals directly react with oxygen in the absence of moisture. • Alkali and alkaline earth metals react with oxygen at room temperature and form corresponding oxides, while some metals react with oxygen at higher temperature. • Metals like Ag, Au and Pt are not oxidized as they are noble metals. 7

• During oxidation of a metal, metal oxide is formed as a thin film on the metallic surface which protects the metal from further corrosion. • If diffusion of either o xygen or metal occur across this layer, further corrosion is possible.

(ii) Corrosion by other gases (Cl 2 , SO 2 , H 2 S) • In dry atmosphere, these gases react with metal and form corrosion products which m a y be protective or nonprotective. • In petroleum industries at high temperatures, H 2 S attacks steel forming Fe. S scale which is porous and interferes with normal operations.

(iii) Liquid - metal corrosion • In several industries, molten metal passes through metallic pipes and causes corrosion due to dissolution or due to internal penetration. • For example, liquid metal mercury dissolves most metals by forming amalgams, thereby corroding them. 1 0

WET CORROSION THEORY • It is a common type of corrosion of metal in aqueous corrosive environment. • This type of corrosion occurs when the contact with a metal comes in conducting liquid • According to this theory, there is the formation of a galvanic cell on the surface of metals. • Some parts of the metal surface act as anode and rest act as cathode. 1 1

• The chemical in the environment acts as an electrolyte. • Oxidation of anodic part takes place and it results in corrosion at anode, while reduction takes place at cathode. • Gain of electron is reduction 1 2
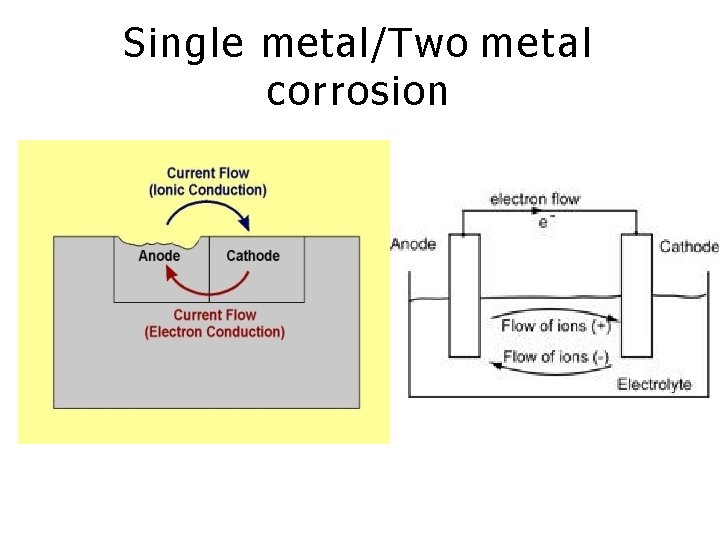
Single metal/Two metal corrosion

Differences between Dry corrosion • Corrosion occurs in the absence of moisture. Wet corrosion • Corrosion occurs in presence of conducting medium. • It involves direct attack • It involves formation of of chemicals on the metal electrochemical surface. cells. • It is a rapid process. • The process is slow. • Corrosion products are • Corrosion occurs at produced at the site of anode but rust is deposited at cathode. corrosion. • The process of corrosion • It depends on the size is uniform. of the anodic part of metal.

Factors Influencing Rate of Corrosion • The nature and extent of corrosion depend on the metal and the environment(i) Nature of the metal (purity) (ii) Environment (p. H, oxidising agents) (iii) Temperature (iv) Motion (it m a y cause erosion) 1 5

TYEPS OF CORROSION There are basically eight types of corrosion I. Uniform corrosion II. Pitting corrosion III. Intergranular (Intercrystalline ) corrosion IV. Stress corrosion V. Crevice corrosion VI. Galvanic corrosion VII. Erosion 1 6

I. Uniform corrosion • This type of corrosion results in a uniform and continuous decrease in thickness over the entire surface area of the metal. • The rate of uniform corrosion can be easily determined by measuring the mass loss, or the quantity of released hydrogen 1 7

II. Pitting corrosion • When there is break in protective oxidative layer, impurities. • This localized form of corrosion is characterized by the formation of irregularly shaped cavities on the surface of the metal. • Their diameter and depth depend on several parameters related to the metal, the medium and service conditions. • It is non uniform corrosion, the intensity and rate of pitting corrosion can not be assessed by released hydrogen. 1 8

• Pitting corrosion can be assessed using three criteria : the density, i. e. the number of pits per unit area, the rate of deepening and the probability of pitting • It is caused by Chloride containing ion.

III. Intergranular (weld decay) corrosion • Boundries in metal alloy • Boundries contain material which shows electrode potential more than other. • Impoverished (depleted on one component) is anodic wrt other. • Steel can be stabilized by titanium

i. V. Stress corrosion • This type of corrosion results from the combine action of a mechanical stress (bending, tension) and thermal. • Unequal rate of cooling for different parts • Stress induced by bolts 2 1

V. Crevice corrosion • Crevice corrosion is a localized corrosion in recesses : • overlapping zones for riveting, bolting or welding, zones under joints and under various deposits. • These zones also called crevices • Metal with more concentration of oxygen become anode • This type of corrosion is also known as deposit attack. 2 2

VI. Galvanic corrosion • When two dissimilar metals with large difference in electrochemical potential, are in direct contact in a conducting liquid • One of the two m a y corrode. • This is called galvanic corrosion. • The other metal will not corrode; it m a y even be protected in this way. • Protective oxy layer reduce corrosion 2 3

VIII. Erosion • Corrosion because of attrition or abrasion of fluid on metal. • Corrosion by erosion occurs in moving media. • This rate of corrosion is related to the flow speed of the fluid. • It leads to local thinning of the metal, which results in scratches, which are always oriented in the same direction, of the flow direction. • Insulation can reduce corrosion 2 4

Prevention (Combat) of corrosion • Material selection • Proper fabrication / designing • Alteration of environment • Cathodic (metal which is to be protected is made cathode) • Anodic protection • Surface coatings

Material selection • Electrochemical potential • Strength • Thermal expansion coefficient

Designing • Joints • Crevices • Drainage • Maintainance

Alteration of environment Reduce • Temperature • Moisture • Turbulance

Cathodic/Anodic Protection

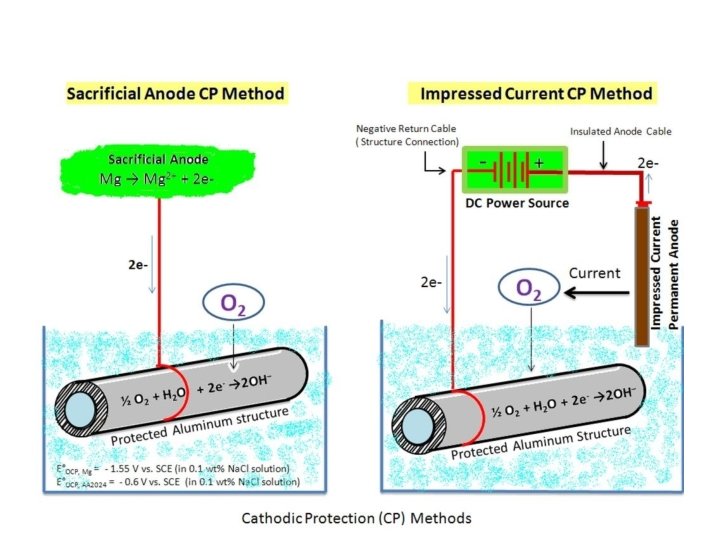
Cathodic protection • • • This is method of reducing or preventing corrosion of a metal by making it a cathode in the electrolytic cell. This can be achieved by means of an externally impressed current or sacrificial anode. An electrolyte is needed to ensure the passage of current through the part to be protected. This is effective only in soils or aqueous media where part to be protected is immersed. It is not effective in the atmosphere.

Contd. . . (1) Impressed –current method an external DC power supply is connected to the metal be protected. • Impressed emf means current which is greater than corrosion current is applied. • The negative terminal of power supply is connected to the part to be protected and the positive to an Auxiliary or inert anode eg: graphite. Steel scrap, Al, Si-Fe are also can be used. Si-Fe and graphite are suitable for ground-beds-buried • Applications: pipe-lines, underground cables of Al, Pb; storage tanks, heatexchangers, steel-gates exposed to sea water, hulls of ships, highways and bridges.

Contd. . . (2) Sacrificial anode this metal which has more negative electrode potential than the structure to be protected is connected electrically to the part or structure to be protected. • The structure is protected at the sacrifice of another metal. Mg alloys, Zn, Al-5% are widely used. These anodes are replaced as soon as consumed. • Applications: under-water parts of ships, ship hull, underground pipes, steel water tanks, water heaters, condenser tubes, oil-cargoballest tanks. Galvanized sheet is sacrificial protection of steel (Zn on steel).

Contd. . . • With sacrificial anodes, the protected metal acts as the cathode, while a more reactive metal is attached to the protected metal as the anode. • The redox reactions will occur spontaneously. While the oxidation reaction takes place at the anode, consuming • Thus, protection of the metal will have been achieved by successfully shifting its corrosion to the anode. • The materials used for the anodes are either pure highly reactive metals like magnesium and zinc

Anodic protection • Metal to be protected is made anode. • This cause formation of a protective film (oxide layer) on metals by current. • An external current is initially applied on the metal so as to passivate it. • Then the current density is reduced to at the value to ensure that the passive film does not dissolve this is passivating current. • Passivating current is minnimum protective current for passivation (at this point there is minnimum currosioin) • In presence of steel titanium become passive. • A potentiostat is used to maintain the metal at a constant potential w. r. t a reference electrode. • The primary advantage is its applicability in extreme corrosive environments with low current requirements.

Thank You
- Slides: 37