Electrochemistry Electrochemical Cells Chemical Cells Also called voltaic















- Slides: 19

Electrochemistry

Electrochemical Cells/ Chemical Cells § § § Also called voltaic or galvanic cells A redox reaction produces electricity Occurs spontaneously

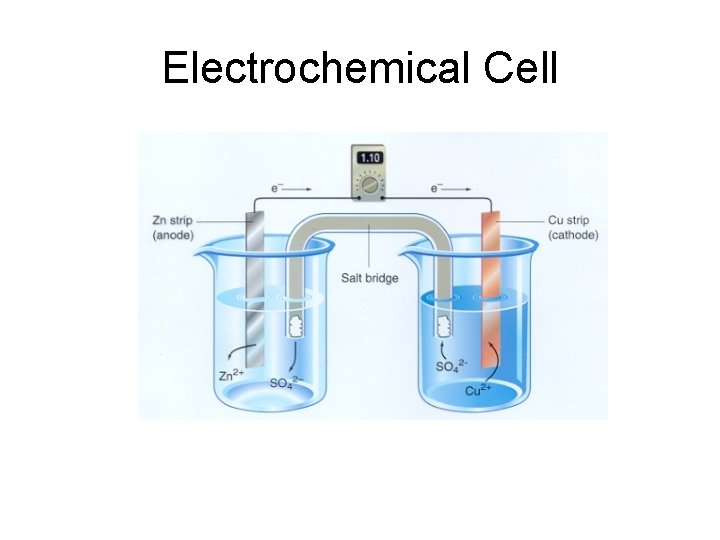
Electrochemical Cell

Half Cells Each ½ of the redox reaction occurs in a separate container – One for oxidation and one for reduction They are connected by a salt bridge – Salt Bridge: allows ions to flow between the two cells

Electrodes Metals which provide a surface for oxidation or reduction to occur – Solids – Oxidation Number = 0 – Anode – Cathode

ANODE – Oxidation occurs at the anode – Negative electrode CATHODE – Reduction occurs at the cathode – Positive electrode Red Cat – An Ox Reduction at the Cathode Oxidation at the Anode

Flow of Electrons The electrodes are connected by a wire Electrons flow from the anode to the cathode through the wire

Why does the cell produce electricity? There is a difference of electric potential between the two electrodes – Electrons will flow between the two electrodes until equilibrium is reached – At equilibrium the cell’s voltage would be zero

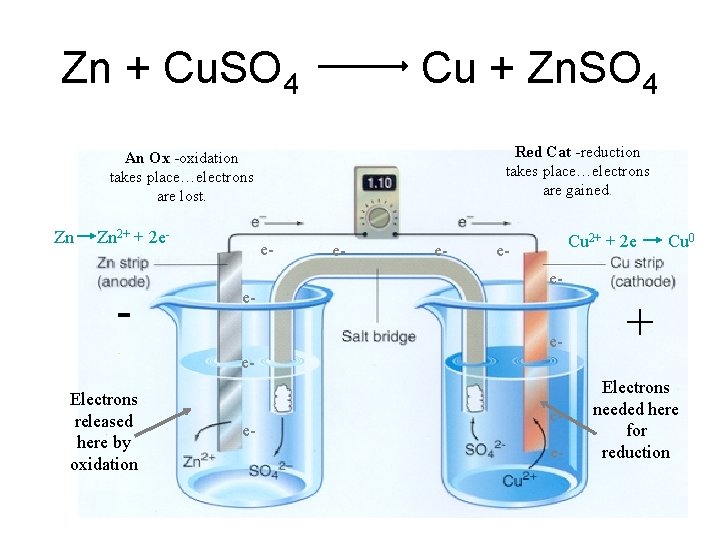
Zn + Cu. SO 4 Cu + Zn. SO 4 Red Cat -reduction takes place…electrons are gained. An Ox -oxidation takes place…electrons are lost. Zn Zn 2+ + 2 e- - ee- e- e- Cu 2+ + 2 e e- - Cu 0 ee- + e. Electrons released here by oxidation e- ee- Electrons needed here for reduction

Batteries • Use a redox reaction which produces electricity spontaneously • Batteries are recharged by reversing the reaction • Dry Cell (Acid or Alkaline), Lead Storage (Car), Rechargeable (Ni/Cd)

Corrosion Oxidation of a metal n Metal combines with element (usually oxygen) Example: 4 Fe + O 2 2 Fe 2 O 3 (rust) n

Prevention of Rust n n Cover the metal – paint, oil, another (more reactive) metal Cathodic Prevention metal is placed in contact with a more reactive metal n That metal will be oxidized (acts as the anode), the original metal acts as the cathode n n Alloys – mixture of metals n Brass, stainless steel (Fe + Cr), cast iron (C + Si)

Electrolytic Cells u Also called electrolysis u An electric current is used to produce a chemical reaction – An electric current is used to force a non -spontaneous reaction to occur

u Oxidation occurs at the anode u Reduction occurs at the cathode u Electrons flow from anode to cathode u The cathode is the negative electrode u The anode is the positive electrode – This is opposite of the chemical cell because the external current causes the polarities to switch


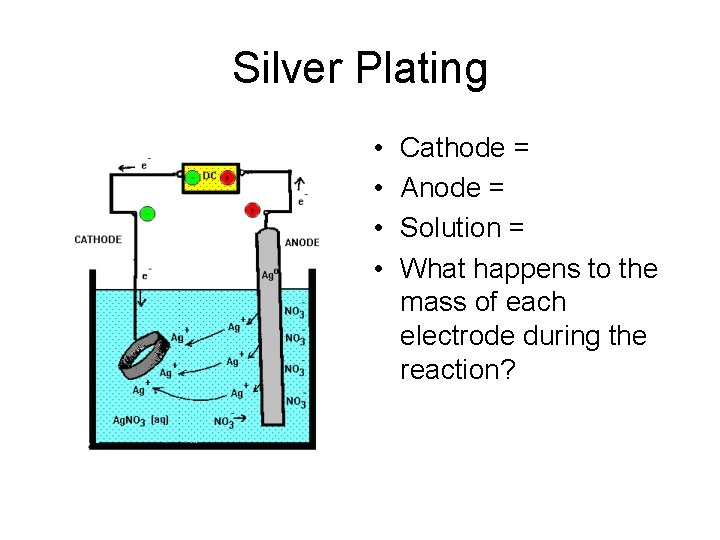
Electroplating Object to be plated is the CATHODE, negative n Metal to be plated onto the object is the ANODE, positive n Solution must contain ions of the metal to be plated n

Silver Plating • • Cathode = Anode = Solution = What happens to the mass of each electrode during the reaction?

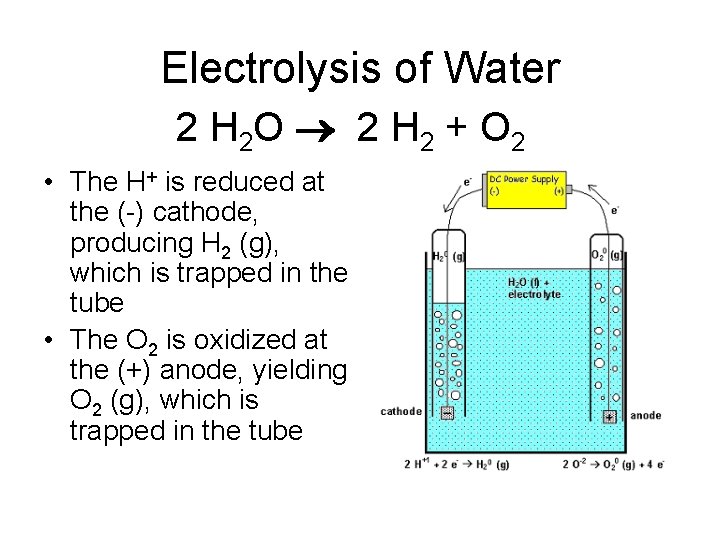
Electrolysis of Water 2 H 2 O 2 H 2 + O 2 • The H+ is reduced at the (-) cathode, producing H 2 (g), which is trapped in the tube • The O 2 is oxidized at the (+) anode, yielding O 2 (g), which is trapped in the tube

Hydrogen Fuel Cells • Uses hydrogen gas as the fuel 2 H 2 + O 2 2 H 2 O