14 2 a Voltaic Cells Basic Function Voltaic

14. 2 a Voltaic Cells Basic Function

Voltaic Cell Basics • Electrodes and electrolytes chemically react to form ions that move in or out of solution. • Anode = electrode where electrons are transferred out of the solution (ox. occurs) • Cathode = electrode where electrons are transferred into the solution (red. occurs) • Electrolytes can be a liquid or a paste • Each half-cell contains one electrode and one electrolyte

Voltaic Cells • • • device that changes chemical energy into electrical energy involves a redox reaction reducing agent transfers electrons to the oxidizing agent § oxidation: <---- e- ----loss of electrons ( ox. state) § reduction: cathode -- anode gain of electrons red/SOA ox/SRA ( ox state)

Voltaic Cells • if the two half-rxtns are combined in the same container, the electron exchange occurs directly as work and released as heat (not ideal) • to harness the energy, keep each half-rxtn in a separate container so the electron transfer occurs through a wire (half-cells) • when the metal solid of the metal ion is not a possibility then an inert electrode is used • usually carbon or platinum
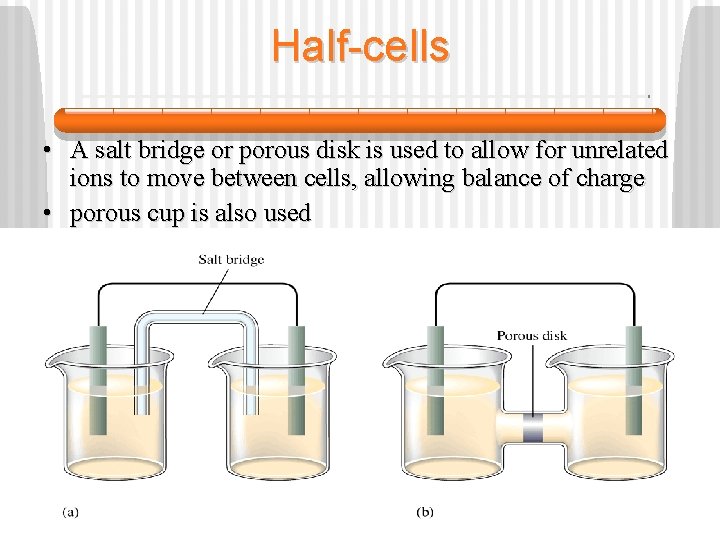
Half-cells • A salt bridge or porous disk is used to allow for unrelated ions to move between cells, allowing balance of charge • porous cup is also used
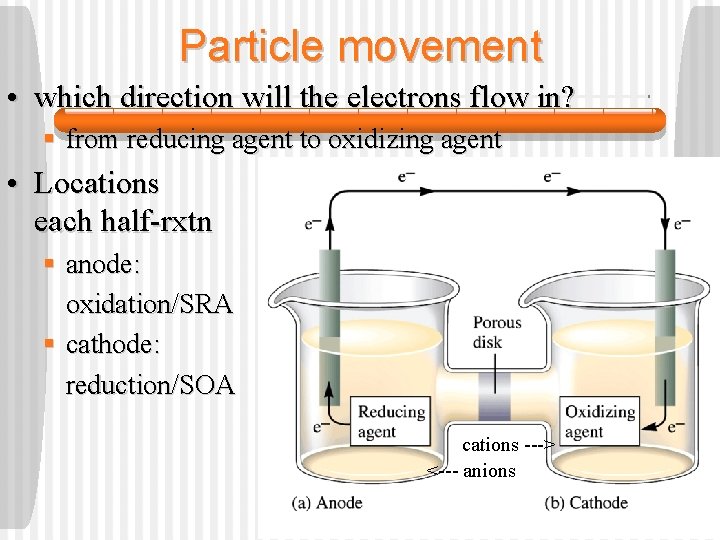
Particle movement • which direction will the electrons flow in? § from reducing agent to oxidizing agent • Locations each half-rxtn of § anode: oxidation/SRA § cathode: reduction/SOA cations ---> <--- anions

Cell Notation • • • short hand for describing cells anode is on L and cathode is on R separate anode and cathode half-rxtn with || separate phases in one half-rxtn with | electrodes go on far ends of notation Mg(s) | Mg 2+(aq) || Al 3+(aq) | Al(s) • Pt(s) | Cl. O 3 -(aq), Cl. O 4 -(aq) || Mn. O 4 -(aq), Mn 2+(aq) | Pt(s)

- flow ------------> ------------e | anode (-) | electrolyte | cathode (+) cations ---> <--- anions

Basic Concepts of Electrochemical Cells Anode Cathode

CHEMICAL CHANGE ---> ELECTRIC CURRENT With time, Cu plates out onto Zn metal strip, and Zn strip “disappears. ” • Zn is oxidized and is the reducing agent Zn(s) ---> Zn 2+(aq) + 2 e • Cu 2+ is reduced and is the oxidizing agent Cu 2+(aq) + 2 e- ---> Cu(s)

CHEMICAL CHANGE ---> ELECTRIC CURRENT • To obtain a useful current, we separate the oxidizing and reducing agents so e- transfer occurs thru an external wire. This is accomplished in a GALVANIC or. com/physsci/chemistry/essentialchemistry/flash/galvan 5. swf VOLTAIC cell. http: //www. mhhe. com/ A group of such cells is called a battery.

Zn --> Zn 2+ + 2 e- Cu 2+ + 2 e- --> Cu Oxidation Anode Negative <--Anions Cations--> Reduction Cathode Positive • Electrons travel thru external wire • Salt bridge allows anions and cations to move between half cells


Cell description • Describe a voltaic Cell: § balanced chemical eqtn. § give the direction of electron flow § assign the anode and cathode § give cell notation

Example • Describe the Galvanic cell based on the following half-reactions: § Ag+ + e- Ag § Fe 3+ + e- Fe 2+ • Write balanced equation § Fe half-rxtn is reversed since Fe 2+ is SRA § Ag+ + Fe 2+ Ag + Fe 3+

Example • Give the direction of electron flow § oxidation: Fe 2+ Fe 3+ + e§ reduction: Ag+ + e- Ag § electrons flow from Fe 2+ half-cell to Ag+ halfcell • Assign cathode and anode § anode: oxidation: Fe 2+ Fe 3+ + e§ cathode: reduction: Ag+ + e- Ag
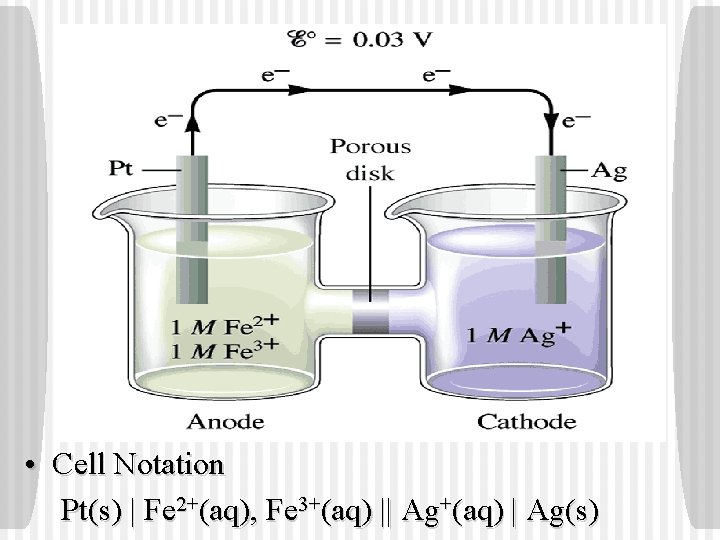
• Cell Notation Pt(s) | Fe 2+(aq), Fe 3+(aq) || Ag+(aq) | Ag(s)

Homework • Read p. 622 - 626 in textbook • Questions p. 626 #1 -8 • LSM 14. 2 C & D
- Slides: 18