Chemical Reactions BONETTRATHBURN CHEMISTRY Chemical Reactions Parts of

Chemical Reactions BONETT/RATHBURN CHEMISTRY

Chemical Reactions Parts of a chemical reaction: �Reactants – substances before reaction occurs �Products – substances produced by the reaction

Chemical Reactions Reason why reactions occur: �Increase in stability for products

Types of Reactions � Combination - two substances combine to form new substance A + B AB Ca + Cl 2 Ca. Cl 2

Types of Reactions � Combustion – substance reacts with oxygen A + O 2 AO CH 4 + O 2 CO 2 + H 2 O

Types of Reactions � Decomposition – substance breaks into two or more substances AB A + B Mn. SO 4 Mn. O + SO 3

Types of Reactions � Single-replacement – uncombined element “trades places” with an element in a compound A +BX AX + B Cd + 2 HCl Cd. Cl 2 + H 2

Types of Reactions � Double-replacement – two binary compounds “trade partners” AX +BY AY + BX Ca. CO 3 + 2 HCl Ca. Cl 2 + H 2 CO 3

Types of Reactions � Combination - two substances combine to form new substance A + B AB Ca + Cl 2 Ca. Cl 2

Types of Reactions � Combustion – substance reacts with oxygen A + O 2 AO CH 4 + O 2 CO 2 + H 2 O

Types of Reactions � Decomposition – substance breaks into two or more substances AB A + B Mn. SO 4 Mn. O + SO 3

Types of Reactions � Single-replacement – uncombined element “trades places” with an element in a compound A +BX AX + B Cd + 2 HCl Cd. Cl 2 + H 2

Types of Reactions � Double-replacement – two binary compounds “trade partners” AX +BY AY + BX Ca. CO 3 + 2 HCl Ca. Cl 2 + H 2 CO 3
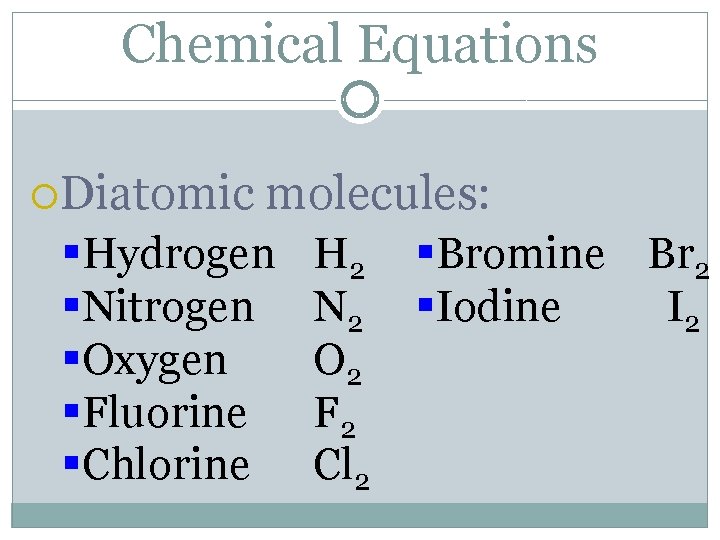
Chemical Equations Diatomic molecules: §Hydrogen §Nitrogen §Oxygen §Fluorine §Chlorine H 2 N 2 O 2 F 2 Cl 2 §Bromine Br 2 §Iodine I 2

Chemical Equations Can include heat � Endothermic – heat, from surroundings, enters the system, has positive heat of reaction (DH) value, reactants have lower potential energy than products

Chemical Equations Can include heat � Endothermic – heat, from surroundings, enters the system � Exothermic – heat exits the system and goes to surroundings, has negative heat of reaction (DH) value , reactants have higher potential energy than products
- Slides: 16