Chemistry Edition Reaction Types Formation Reactions Decomposition Reactions







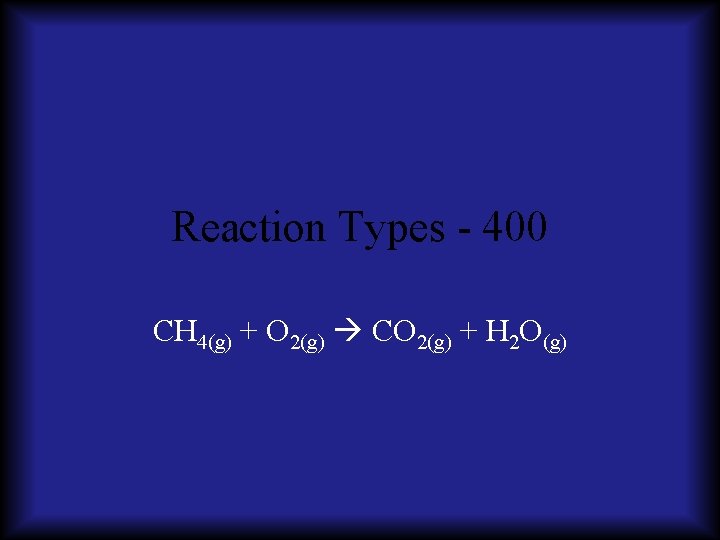











































- Slides: 52

Chemistry Edition

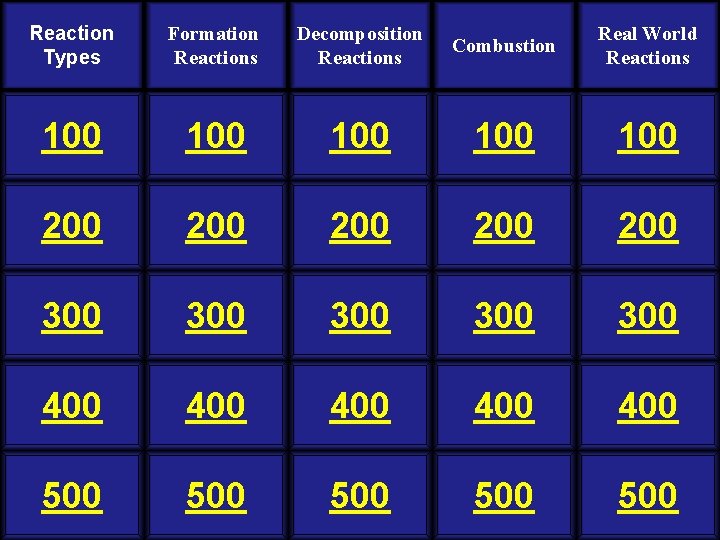
Reaction Types Formation Reactions Decomposition Reactions Combustion Real World Reactions 100 100 100 200 200 200 300 300 300 400 400 400 500 500 500

Reaction Types - 100 Na. Cl(s) Na(s) + Cl 2(g)

Reaction Types - 100 Na. Cl(s) Na(s) + Cl 2(g) A: Decomposition

Reaction Types - 200 Al(s) + S(s) Al 2 S 3(s)

Reaction Types - 200 Al(s) + S(s) Al 2 S 3(s) A: Formation

Reaction Types - 300 CO(g) + O 2(g) CO 2(g)

Reaction Types - 300 S 8(s) + O 2(g) SO 3(g) A: Formation
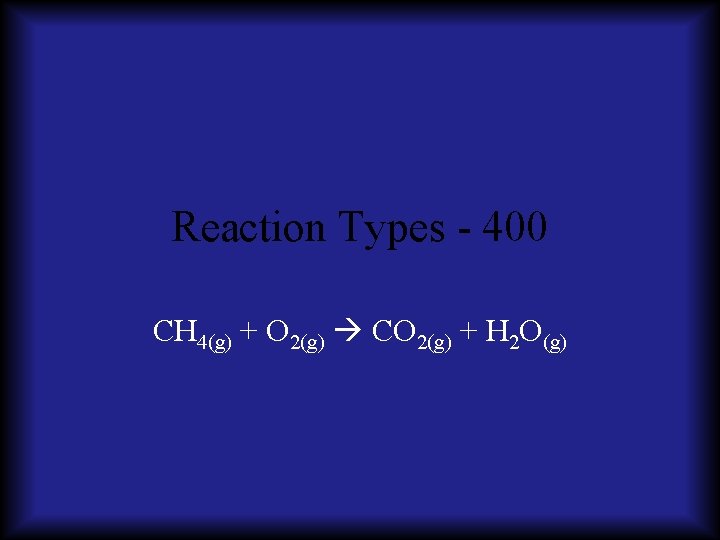
Reaction Types - 400 CH 4(g) + O 2(g) CO 2(g) + H 2 O(g)

Reaction Types - 400 CH 4(g) + O 2(g) CO 2(g) + H 2 O(g) A: Combustion

Reaction Types - 500 Hg. O(s) Hg(l) + O 2(g)

Reaction Types - 500 Hg. O(s) Hg(l) + O 2(g) A: Decomposition

Formation Reactions - 100 Metallic magnesium reacts with oxygen gas to form magnesium oxide.

Formation Reactions - 100 Metallic magnesium reacts with oxygen gas to form magnesium oxide. A: Mg(s) + O 2(g) Mg. O(s)

Formation Reactions - 200 Na(s) + Cl 2(g)

Formation Reactions - 200 Na(s) + Cl 2(g) A: Na(s) + Cl 2(g) Na. Cl(s)

Formation Reactions - 300 Solid phosphorus reacts with chlorine gas to form phosphorus chloride gas.

Formation Reactions - 300 Solid phosphorus reacts with chlorine gas to form phosphorus chloride gas. A: P(s) + Cl 2(g) PCl 3(g)

Formation Reactions - 400 N 2(g) + H 2(g)

Formation Reactions - 400 N 2(g) + H 2(g) A: N 2(g) + H 2(g) NH 3(g)

Formation Reactions - 500 Hydrogen gas reacts with chlorine gas to produce hydrogen chloride gas.

Formation Reactions - 500 Hydrogen gas reacts with chlorine gas to produce hydrogen chloride gas. A: H 2(g) + Cl 2(g) HCl(g)

Decomposition Reactions - 100 Electricity decomposes water producing hydrogen gas and oxygen gas.

Decomposition Reactions - 100 Electricity decomposes water producing hydrogen gas and oxygen gas. A: H 2 O(l) H 2(g) + O 2(g)

Decomposition Reactions - 200 Cu. O(s)

Decomposition Reactions - 200 Cu. O(s) A: Cu. O(s) Cu(s) + O 2(g)

Decomposition Reactions - 300 Solid tetraphosphorus decaoxide is heated to produce solid phosporus and oxygen gas.

Decomposition Reactions - 300 Solid tetraphosphorus decaoxide is heated to produce solid phosporus and oxygen gas. A: P 4 O 10(s) P 4(s) + O 2(g)

Decomposition Reactions - 400 Cl 2 O 5(g)

Decomposition Reactions - 400 Cl 2 O 5(g) A: Cl 2 O 5(g) Cl 2(g) + O 2(g)

Decomposition Reactions - 500 Hydrogen peroxide decomposes into water and oxygen gas.

Decomposition Reactions - 500 Hydrogen peroxide decomposes into water and oxygen gas. A: H 2 O 2(l) H 2 O(l) + O 2(g)

Combustion - 100 Rubbing alcohol (CH 3 OH) burns to produce carbon dioxide gas and water vapour.

Combustion - 100 Rubbing alcohol (CH 3 OH) burns to produce carbon dioxide gas and water vapour. A: CH 3 OH(l) + O 2(g) CO 2(g) + H 2 O(g)

Combustion - 200 CH 4(g) + O 2(g)

Combustion - 200 CH 4(g) + O 2(g) A: CH 4(g) + O 2(g) CO 2(g) + H 2 O(g)

Combustion - 300 Ethene (C 2 H 2) gas reacts with oxygen to produce carbon dioxide and water vapour.

Combustion - 300 Ethene (C 2 H 2) gas reacts with oxygen to produce carbon dioxide and water vapour. A: C 2 H 2(g) + O 2(g) CO 2(g) + H 2 O(g)

Combustion - 400 C 25 H 52(s) + O 2(g)

Combustion - 400 C 25 H 52(s) + O 2(g) A: C 25 H 52(s) + O 2(g) CO 2(g) + H 2 O(g)

Combustion - 500 Petroleum (C 4 H 10) gas burns to produce carbon dioxide gas and water vapour.

Combustion - 500 Petroleum (C 4 H 10) gas burns to produce carbon dioxide gas and water vapour. A: C 4 H 10(g) + O 2(g) CO 2(g) + H 2 O(g)

Real World Reactions - 100 Hydrogen Peroxide on a wound.

Real World Reactions - 100 A: Decomposition H 2 O 2(l) H 2 O(l) + O 2(g)

Real World Reactions - 200

Real World Reactions - 200 A: Combustion H 2(l) + O 2(l) H 2 O(g) + thermal energy

Real World Reactions - 300

Real World Reactions - 300 A: Formation Fe(s) + O 2(g) Fe. O 2(s)

Real World Reactions - 400

Real World Reactions - 400 A: Decomposition H 2 O (l) H 2(g) + O 2(g)

Real World Reactions - 500

Real World Reactions - 500 A: Combustion C 8 H 18(l) + O 2(g) CO 2(g) + H 2 O(g)