Chapter 11 Metal Alloys Applications and Processing ISSUES













- Slides: 33

Chapter 11: Metal Alloys Applications and Processing ISSUES TO ADDRESS. . . • How are metal alloys classified and how are they used? • What are some of the common fabrication techniques? • How do properties vary throughout a piece of material that has been quenched, for example? • How can properties be modified by post heat treatment? Chapter 11 - 1

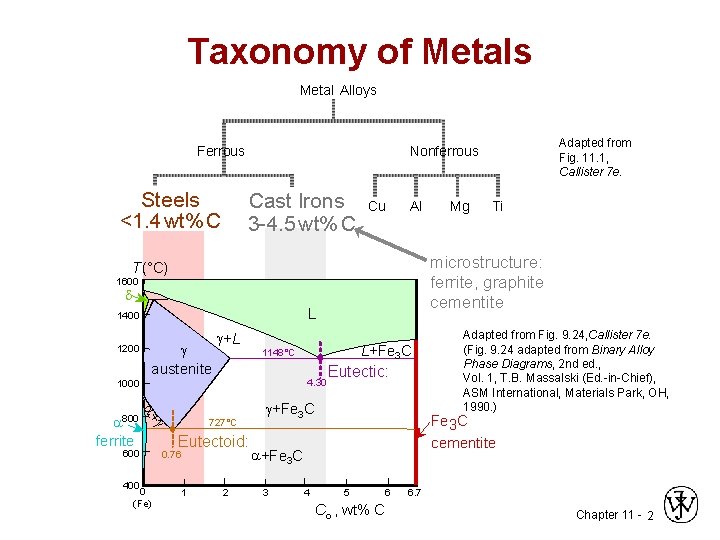
Taxonomy of Metals Metal Alloys Ferrous Steels <1. 4 wt%C <1. 4 wt% C Cast Irons 3 -4. 5 wt%C 3 -4. 5 wt% C Cu Al 1600 d L 1400 austenite +L 4. 30 + 800 ferrite 600 400 L+Fe 3 C 1148°C 1000 0 (Fe) 727°C Eutectoid: 0. 76 1 2 Eutectic: +Fe 3 C 4 Ti Adapted from Fig. 9. 24, Callister 7 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in-Chief), ASM International, Materials Park, OH, 1990. ) Fe 3 C cementite +Fe 3 C 3 Mg microstructure: ferrite, graphite cementite T(°C) 1200 Adapted from Fig. 11. 1, Callister 7 e. Nonferrous 5 6 Co , wt% C 6. 7 Chapter 11 - 2

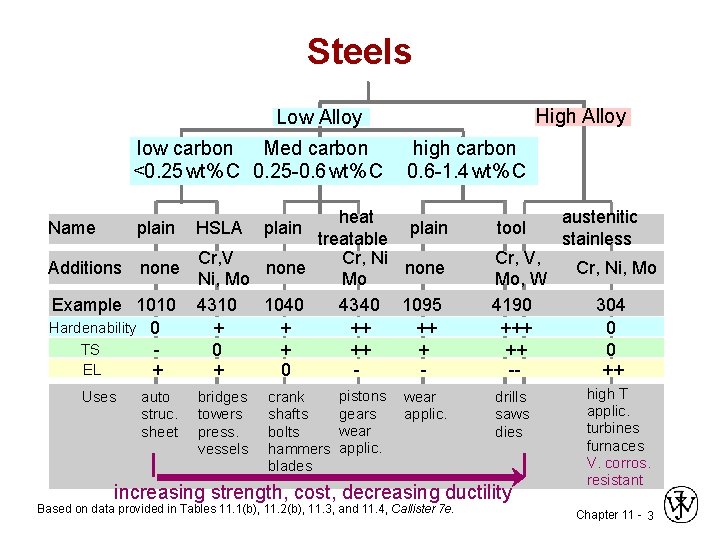
Steels High Alloy Low Alloy low carbon Med carbon <0. 25 wt% C 0. 25 -0. 6 wt% C high carbon 0. 6 -1. 4 wt% C heat plain treatable Cr, V Cr, Ni Additions none Ni, Mo Mo Example 1010 4310 1040 4340 1095 Hardenability 0 + + ++ ++ TS 0 + ++ + EL + + 0 - Name plain Uses auto struc. sheet HSLA bridges towers press. vessels plain crank shafts bolts hammers blades pistons gears wear applic. tool Cr, V, Mo, W 4190 +++ ++ -drills saws dies increasing strength, cost, decreasing ductility Based on data provided in Tables 11. 1(b), 11. 2(b), 11. 3, and 11. 4, Callister 7 e. austenitic stainless Cr, Ni, Mo 304 0 0 ++ high T applic. turbines furnaces V. corros. resistant Chapter 11 - 3

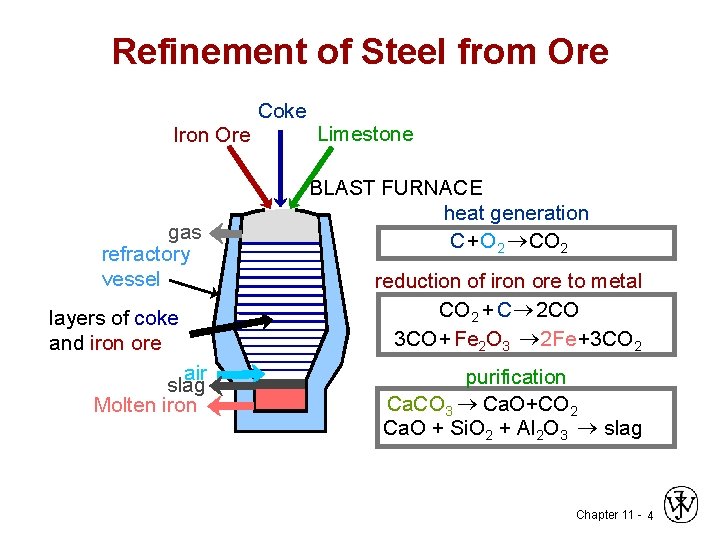
Refinement of Steel from Ore Coke Iron Ore gas refractory vessel layers of coke and iron ore air slag Molten iron Limestone BLAST FURNACE heat generation C+O 2 ®CO 2 reduction of iron ore to metal CO 2 + C ® 2 CO 3 CO + Fe 2 O 3 ® 2 Fe+3 CO 2 purification Ca. CO 3 ® Ca. O+CO 2 Ca. O + Si. O 2 + Al 2 O 3 ® slag Chapter 11 - 4

Ferrous Alloys Iron containing – Steels - cast irons Nomenclature AISI & SAE 10 xx Plain Carbon Steels 11 xx Plain Carbon Steels (resulfurized for machinability) 15 xx Mn (10 ~ 20%) 40 xx Mo (0. 20 ~ 0. 30%) 43 xx Ni (1. 65 - 2. 00%), Cr (0. 4 - 0. 90%), Mo (0. 2 - 0. 3%) 44 xx Mo (0. 5%) where xx is wt% C x 100 example: 1060 steel – plain carbon steel with 0. 60 wt% C Stainless Steel -- >11% Cr Chapter 11 - 5

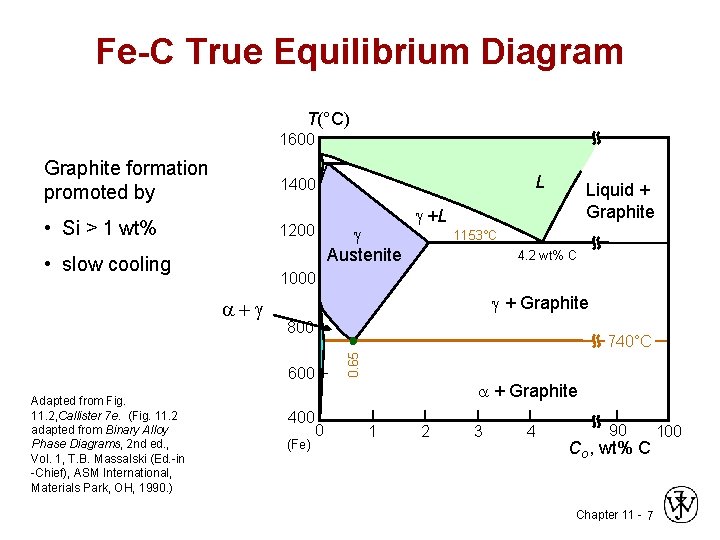
Cast Iron • Ferrous alloys with > 2. 1 wt% C – more commonly 3 - 4. 5 wt%C • low melting (also brittle) so easiest to cast • Cementite decomposes to ferrite + graphite Fe 3 C 3 Fe ( ) + C (graphite) – generally a slow process Chapter 11 - 6

Fe-C True Equilibrium Diagram T(°C) 1600 Graphite formation promoted by 1400 • Si > 1 wt% 1200 • slow cooling Austenite Liquid + Graphite +L 1153°C 4. 2 wt% C 1000 + Graphite 800 740°C 0. 65 + 600 Adapted from Fig. 11. 2, Callister 7 e. (Fig. 11. 2 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in -Chief), ASM International, Materials Park, OH, 1990. ) L 400 (Fe) + Graphite 0 1 2 3 4 90 Co , wt% C Chapter 11 - 7 100

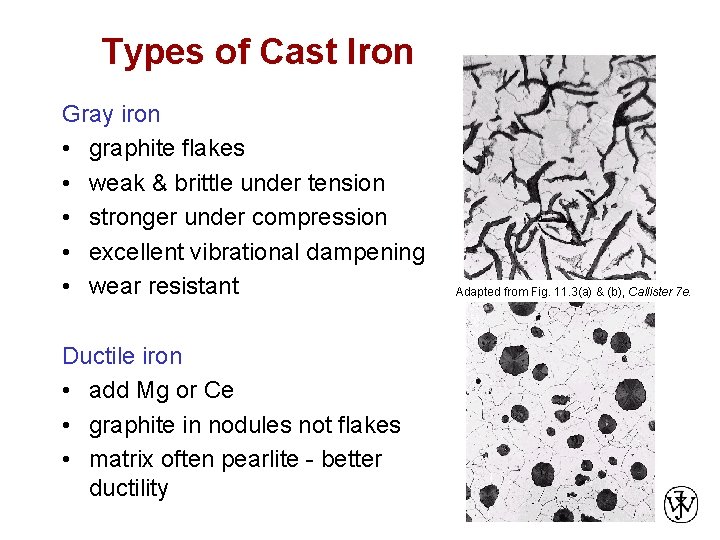
Types of Cast Iron Gray iron • graphite flakes • weak & brittle under tension • stronger under compression • excellent vibrational dampening • wear resistant Adapted from Fig. 11. 3(a) & (b), Callister 7 e. Ductile iron • add Mg or Ce • graphite in nodules not flakes • matrix often pearlite - better ductility Chapter 11 - 8

Types of Cast Iron White iron • <1 wt% Si so harder but brittle • more cementite Adapted from Fig. 11. 3(c) & (d), Callister 7 e. Malleable iron • heat treat at 800 -900ºC • graphite in rosettes • more ductile Chapter 11 - 9

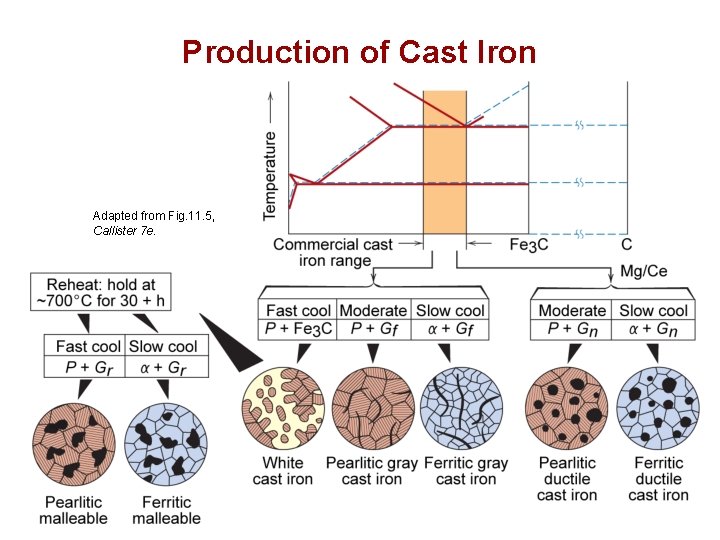
Production of Cast Iron Adapted from Fig. 11. 5, Callister 7 e. Chapter 11 - 10

Limitations of Ferrous Alloys 1) Relatively high density 2) Relatively low conductivity 3) Poor corrosion resistance Chapter 11 - 11

Nonferrous Alloys • Cu Alloys • Al Alloys -lower r: 2. 7 g/cm 3 Brass: Zn is subst. impurity (costume jewelry, coins, -Cu, Mg, Si, Mn, Zn additions corrosion resistant) -solid sol. or precip. Bronze : Sn, Al, Si, Ni are strengthened (struct. subst. impurity aircraft parts (bushings, landing & packaging) gear) • Mg Alloys Non. Ferrous Cu-Be: -very low r: 1. 7 g/cm 3 Alloys precip. hardened -ignites easily for strength -aircraft, missiles • Ti Alloys • Refractory metals -lower r: 4. 5 g/cm 3 -high melting T vs 7. 9 for steel • Noble metals -Nb, Mo, W, Ta -reactive at high T -Ag, Au, Pt -oxid. /corr. resistant -space applic. Based on discussion and data provided in Section 11. 3, Callister 7 e. Chapter 11 - 12

Metal Fabrication • How do we fabricate metals? – Blacksmith - hammer (forged) – Molding - cast • Forming Operations – Rough stock formed to final shape Hot working • T high enough for recrystallization • Larger deformations vs. Cold working • well below Tm • work hardening • smaller deformations Chapter 11 - 13

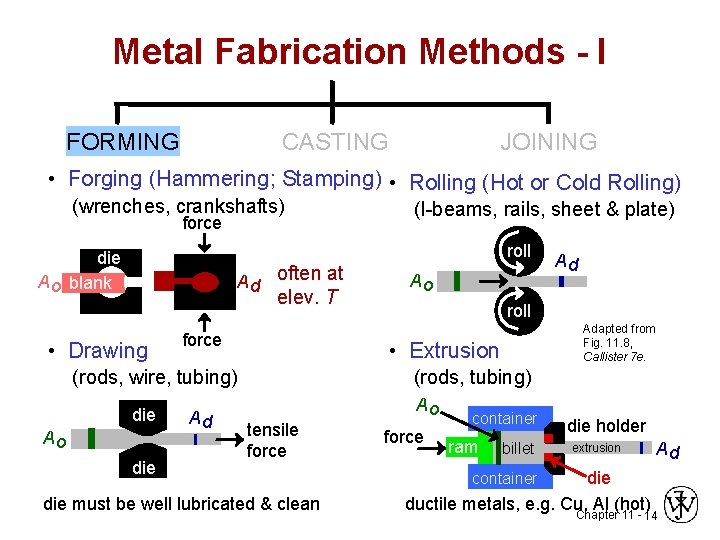
Metal Fabrication Methods - I FORMING CASTING JOINING • Forging (Hammering; Stamping) • Rolling (Hot or Cold Rolling) (wrenches, crankshafts) force (I-beams, rails, sheet & plate) roll die A o blank A d often at elev. T • Drawing force Ao die Ad roll Adapted from Fig. 11. 8, Callister 7 e. • Extrusion (rods, wire, tubing) die Ao Ad (rods, tubing) Ao tensile force die must be well lubricated & clean force container ram billet die holder Ad extrusion die ductile metals, e. g. Cu, Al (hot) Chapter 11 container 14

Metal Fabrication Methods - II FORMING CASTING JOINING • Casting- mold is filled with metal – metal melted in furnace, perhaps alloying elements added. Then cast in a mold – most common, cheapest method – gives good production of shapes – weaker products, internal defects – good option for brittle materials Chapter 11 - 15

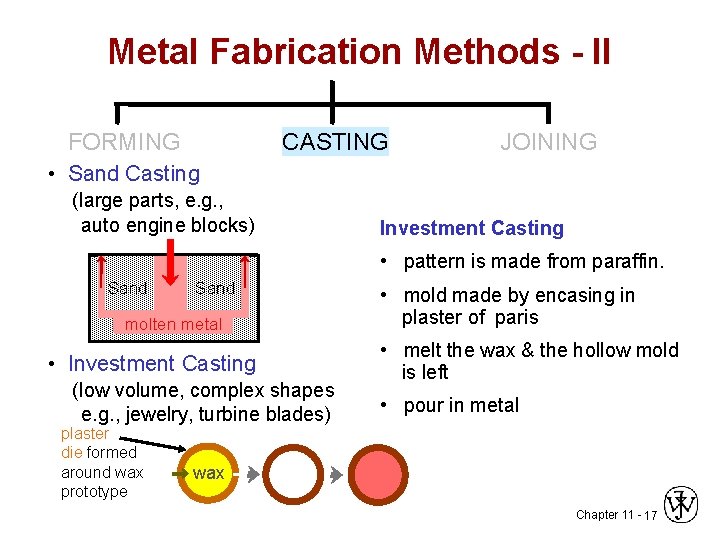
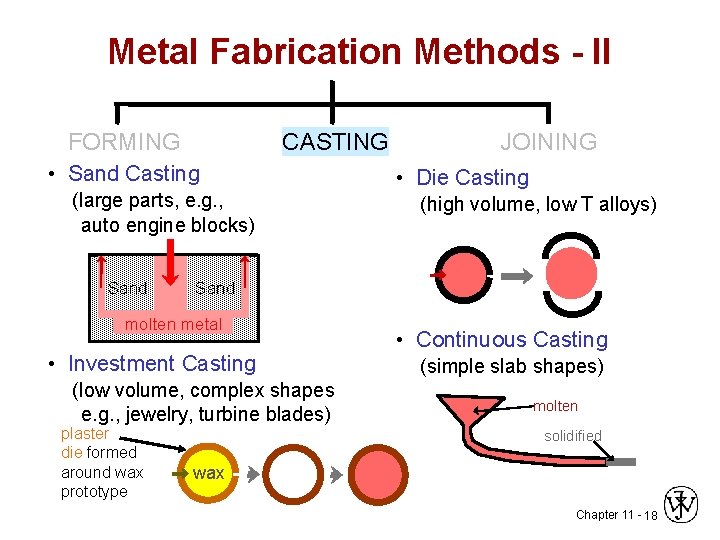
Metal Fabrication Methods - II FORMING CASTING JOINING • Sand Casting (large parts, e. g. , auto engine blocks) • trying to hold something that is hot • what will withstand >1600ºC? Sand molten metal • cheap - easy to mold => sand!!! • pack sand around form (pattern) of desired shape Chapter 11 - 16

Metal Fabrication Methods - II FORMING CASTING JOINING • Sand Casting (large parts, e. g. , auto engine blocks) Investment Casting • pattern is made from paraffin. Sand molten metal • Investment Casting (low volume, complex shapes e. g. , jewelry, turbine blades) plaster die formed around wax prototype • mold made by encasing in plaster of paris • melt the wax & the hollow mold is left • pour in metal wax Chapter 11 - 17

Metal Fabrication Methods - II FORMING CASTING • Sand Casting (large parts, e. g. , auto engine blocks) Sand • Die Casting (high volume, low T alloys) Sand molten metal • Investment Casting (low volume, complex shapes e. g. , jewelry, turbine blades) plaster die formed around wax prototype JOINING • Continuous Casting (simple slab shapes) molten solidified wax Chapter 11 - 18

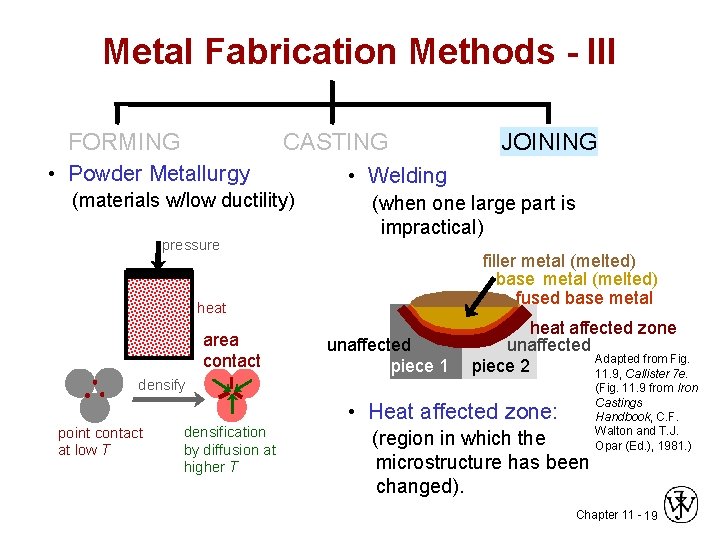
Metal Fabrication Methods - III FORMING CASTING • Powder Metallurgy (materials w/low ductility) pressure • Welding (when one large part is impractical) filler metal (melted) base metal (melted) fused base metal heat area contact densify JOINING unaffected piece 1 heat affected zone unaffected Adapted from Fig. piece 2 11. 9, Callister 7 e. • Heat affected zone: point contact at low T densification by diffusion at higher T (region in which the microstructure has been changed). (Fig. 11. 9 from Iron Castings Handbook, C. F. Walton and T. J. Opar (Ed. ), 1981. ) Chapter 11 - 19

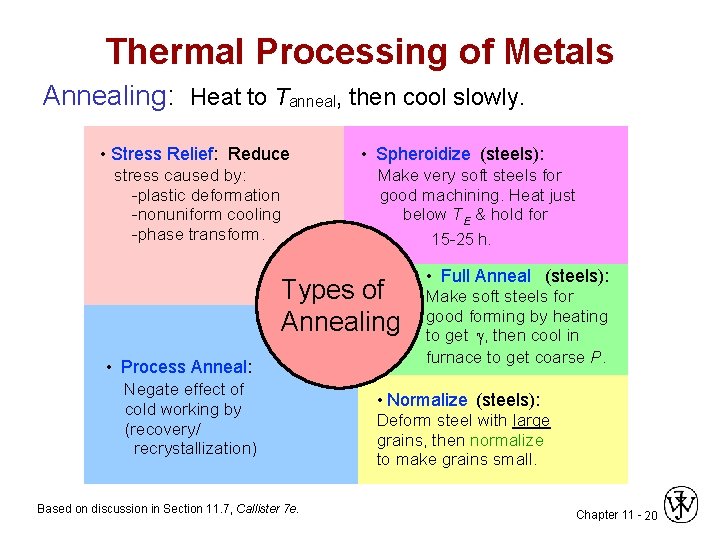
Thermal Processing of Metals Annealing: Heat to Tanneal, then cool slowly. • Stress Relief: Reduce stress caused by: -plastic deformation -nonuniform cooling -phase transform. • Spheroidize (steels): Make very soft steels for good machining. Heat just below TE & hold for 15 -25 h. Types of Annealing • Process Anneal: Negate effect of cold working by (recovery/ recrystallization) Based on discussion in Section 11. 7, Callister 7 e. • Full Anneal (steels): Make soft steels for good forming by heating to get , then cool in furnace to get coarse P. • Normalize (steels): Deform steel with large grains, then normalize to make grains small. Chapter 11 - 20

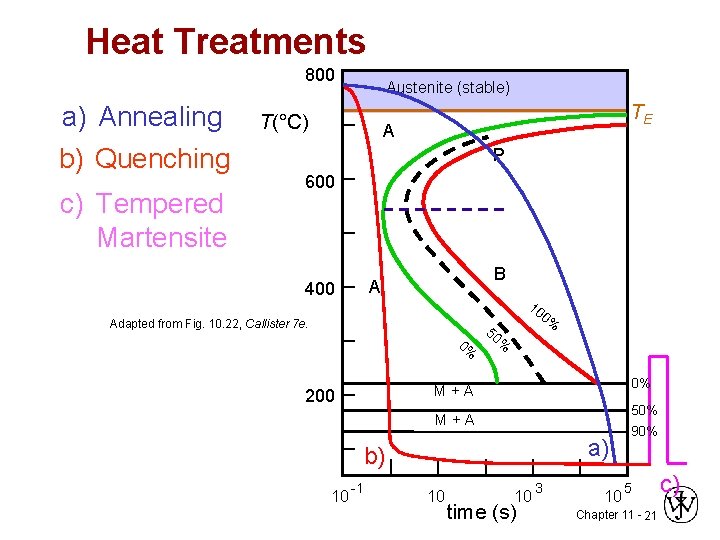
Heat Treatments 800 a) Annealing b) Quenching c) Tempered Martensite Austenite (stable) T(°C) TE A P 600 B A 400 10 Adapted from Fig. 10. 22, Callister 7 e. 0% 0% 50 % 0% M+A 200 50% M+A a) b) 10 -1 10 10 time (s) 3 10 90% 5 Chapter 11 - 21 c)

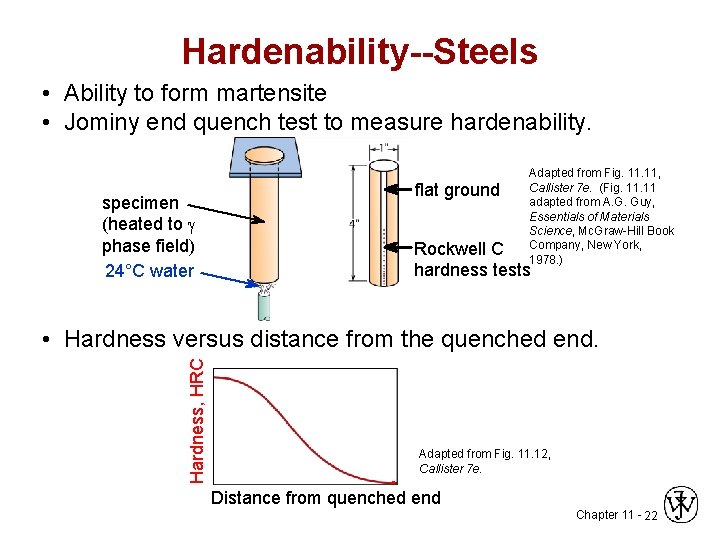
Hardenability--Steels • Ability to form martensite • Jominy end quench test to measure hardenability. specimen (heated to phase field) 24°C water flat ground Adapted from Fig. 11, Callister 7 e. (Fig. 11 adapted from A. G. Guy, Essentials of Materials Science, Mc. Graw-Hill Book Company, New York, 1978. ) Rockwell C hardness tests Hardness, HRC • Hardness versus distance from the quenched end. Adapted from Fig. 11. 12, Callister 7 e. Distance from quenched end Chapter 11 - 22

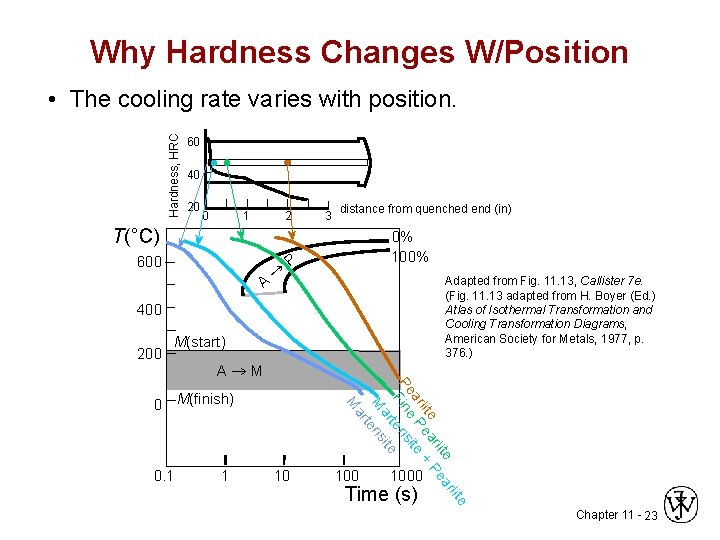
Why Hardness Changes W/Position Hardness, HRC • The cooling rate varies with position. 60 40 20 0 1 2 3 distance from quenched end (in) T(°C) 600 A ® 0% 100% P Adapted from Fig. 11. 13, Callister 7 e. (Fig. 11. 13 adapted from H. Boyer (Ed. ) Atlas of Isothermal Transformation and Cooling Transformation Diagrams, American Society for Metals, 1977, p. 376. ) 400 200 M(start) A®M 1 li te ar rli Pe lite ea + ar P ite Pe ine ens F t ar ite M ens t ar 0. 1 M 0 M(finish) 10 1000 te Time (s) Chapter 11 - 23

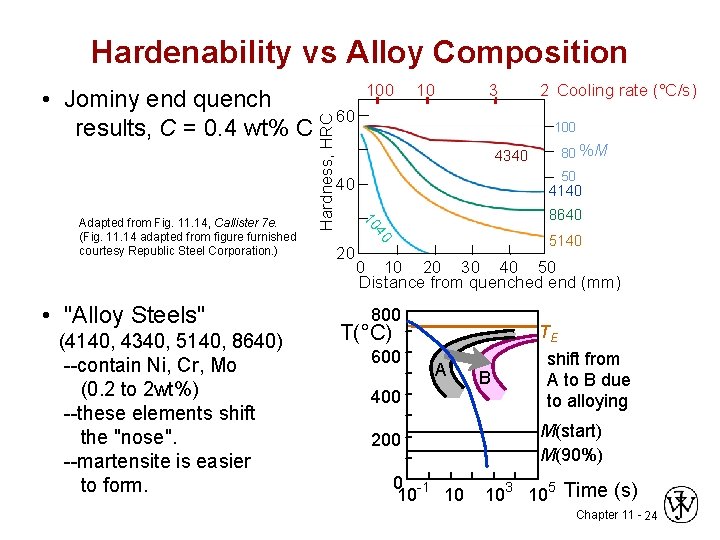
Hardenability vs Alloy Composition (4140, 4340, 5140, 8640) --contain Ni, Cr, Mo (0. 2 to 2 wt%) --these elements shift the "nose". --martensite is easier to form. 3 60 4340 80 %M 50 40 20 2 Cooling rate (°C/s) 100 4140 8640 40 • "Alloy Steels" 10 10 Adapted from Fig. 11. 14, Callister 7 e. (Fig. 11. 14 adapted from figure furnished courtesy Republic Steel Corporation. ) 100 Hardness, HRC • Jominy end quench results, C = 0. 4 wt% C 5140 0 10 20 30 40 50 Distance from quenched end (mm) 800 T(°C) 600 A 400 200 0 -1 10 10 B TE shift from A to B due to alloying M(start) M(90%) 103 105 Time (s) Chapter 11 - 24

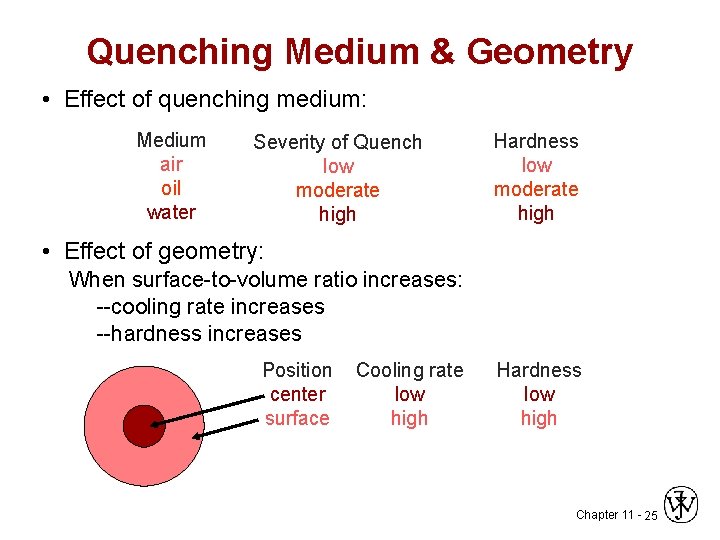
Quenching Medium & Geometry • Effect of quenching medium: Medium air oil water Severity of Quench low moderate high Hardness low moderate high • Effect of geometry: When surface-to-volume ratio increases: --cooling rate increases --hardness increases Position center surface Cooling rate low high Hardness low high Chapter 11 - 25

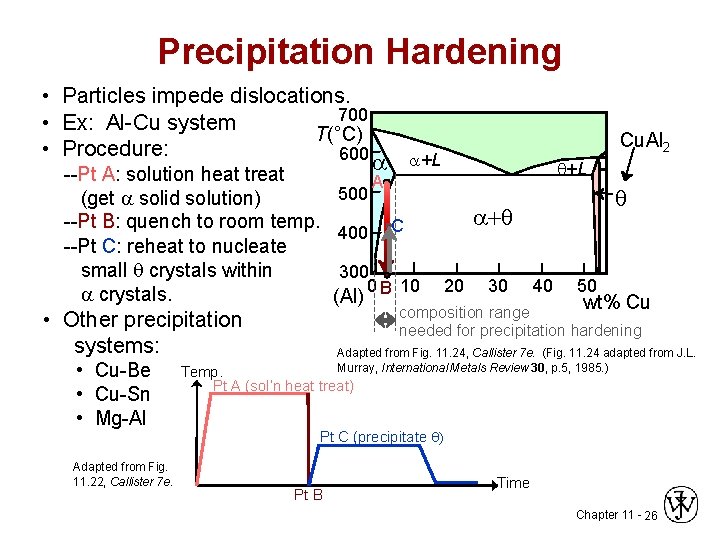
Precipitation Hardening • Particles impede dislocations. 700 • Ex: Al-Cu system T(°C) • Procedure: 600 --Pt A: solution heat treat (get solid solution) --Pt B: quench to room temp. --Pt C: reheat to nucleate small crystals within crystals. • Other precipitation systems: • Cu-Be • Cu-Sn • Mg-Al Adapted from Fig. 11. 22, Callister 7 e. 500 400 +L A + C 300 0 B 10 (Al) Cu. Al 2 L 20 30 40 50 wt% Cu composition range needed for precipitation hardening Adapted from Fig. 11. 24, Callister 7 e. (Fig. 11. 24 adapted from J. L. Murray, International Metals Review 30, p. 5, 1985. ) Temp. Pt A (sol’n heat treat) Pt C (precipitate ) Pt B Time Chapter 11 - 26

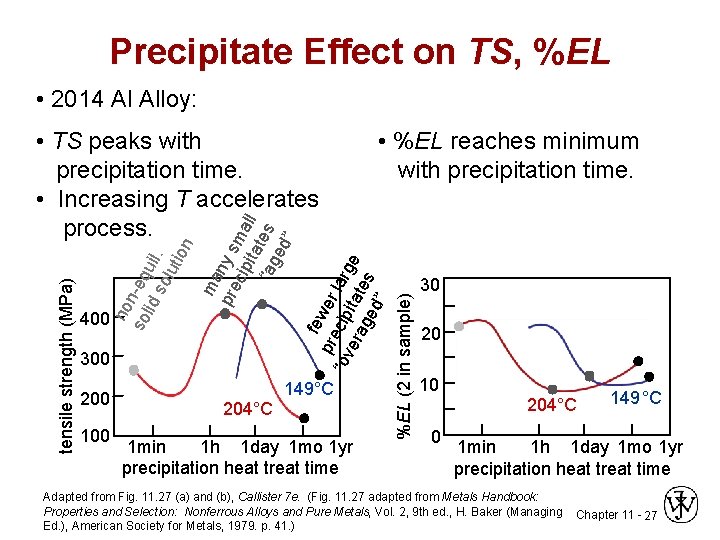
Precipitate Effect on TS, %EL • 2014 Al Alloy: 300 200 100 204°C 149°C 1 min 1 h 1 day 1 mo 1 yr precipitation heat treat time %EL (2 in sample) 400 • %EL reaches minimum with precipitation time. fe pre wer “ov cip larg era itat e ge es d” ma pre ny s cip ma ita ll “ag tes ed ” no so n-eq lid uil so. lut ion tensile strength (MPa) • TS peaks with precipitation time. • Increasing T accelerates process. 30 20 10 0 204°C 149 °C 1 min 1 h 1 day 1 mo 1 yr precipitation heat treat time Adapted from Fig. 11. 27 (a) and (b), Callister 7 e. (Fig. 11. 27 adapted from Metals Handbook: Properties and Selection: Nonferrous Alloys and Pure Metals, Vol. 2, 9 th ed. , H. Baker (Managing Ed. ), American Society for Metals, 1979. p. 41. ) Chapter 11 - 27

Metal Alloy Crystal Stucture Alloys • substitutional alloys – can be ordered or disordered – disordered solid solution – ordered - periodic substitution example: Cu. Au FCC Cu Au Chapter 11 - 28

Metal Alloy Crystal Stucture • Interstitial alloys (compounds) – one metal much larger than the other – smaller metal goes in ordered way into interstitial “holes” in the structure of larger metal – Ex: Cementite – Fe 3 C Chapter 11 - 29

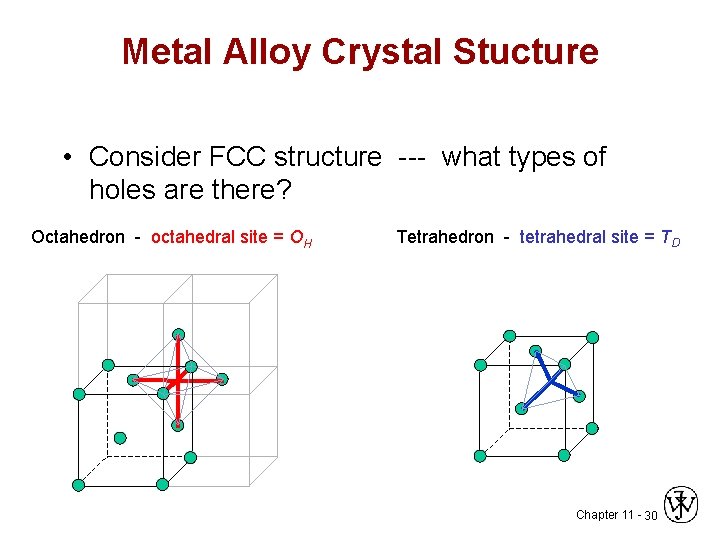
Metal Alloy Crystal Stucture • Consider FCC structure --- what types of holes are there? Octahedron - octahedral site = OH Tetrahedron - tetrahedral site = TD Chapter 11 - 30

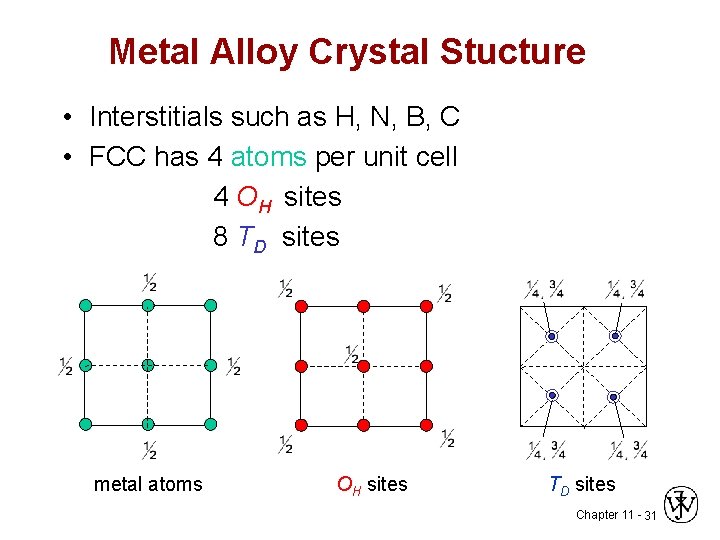
Metal Alloy Crystal Stucture • Interstitials such as H, N, B, C • FCC has 4 atoms per unit cell 4 OH sites 8 TD sites metal atoms OH sites TD sites Chapter 11 - 31

Summary • Steels: increase TS, Hardness (and cost) by adding --C (low alloy steels) --Cr, V, Ni, Mo, W (high alloy steels) --ductility usually decreases w/additions. • Non-ferrous: --Cu, Al, Ti, Mg, Refractory, and noble metals. • Fabrication techniques: --forming, casting, joining. • Hardenability --increases with alloy content. • Precipitation hardening --effective means to increase strength in Al, Cu, and Mg alloys. Chapter 11 - 32

ANNOUNCEMENTS Reading: Core Problems: Self-help Problems: Chapter 11 - 33