Alkaline Earth Metals Beryllium Magnesium Strontium Radium Barium

Alkaline Earth Metals Beryllium Magnesium Strontium Radium Barium Calcium

Alkaline Earth Metals • Why were these elements called “alkaline earth metals”? • What do you think about the uses of these elements commonly?
![Properties Beryllium Magnesium Calcium 1. 9 x 10 -4 2. 9 5 [He]2 s Properties Beryllium Magnesium Calcium 1. 9 x 10 -4 2. 9 5 [He]2 s](http://slidetodoc.com/presentation_image_h2/3dab97404429ea4d97db5f3f1935ee37/image-4.jpg)
Properties Beryllium Magnesium Calcium 1. 9 x 10 -4 2. 9 5 [He]2 s 2 [Ne]3 s 2 [Ar]4 s 2 Melting point (o. C) 1287 650 842 Boiling point (o. C) Density Common oxidation num. Physical State Color 2469 1. 85 +2 solid grey 1090 1. 74 +2 solid white 1484 1. 55 +2 solid White Earth`s crust abundance Electron configuration

• The word “earth” was used for the oxides of group 2 A by the ancient scientists. • Alkaline earth metals, after the alkali metals, are secondary metals with strong metallic properties. • The group 2 A elements are less active than those of 1 A, but more active than those of group 3 A. • Except Be, all form ionic compounds. • Be forms mostly covalent compounds.

• The atomic radius increases from top to bottom. • The melting point, boiling point and the hardness of the elements decrease from top to bottom. • Beryllium is the hardest alkaline earth metal • Barium is the softest alkaline earth metal. • Malleable and ductile. • Excess Mg+2 and Ca+2 in water means that it is hard.

Characteristic Flame Colors of Alkaline Earth Metals Calcium Strontium Barium Magnesium
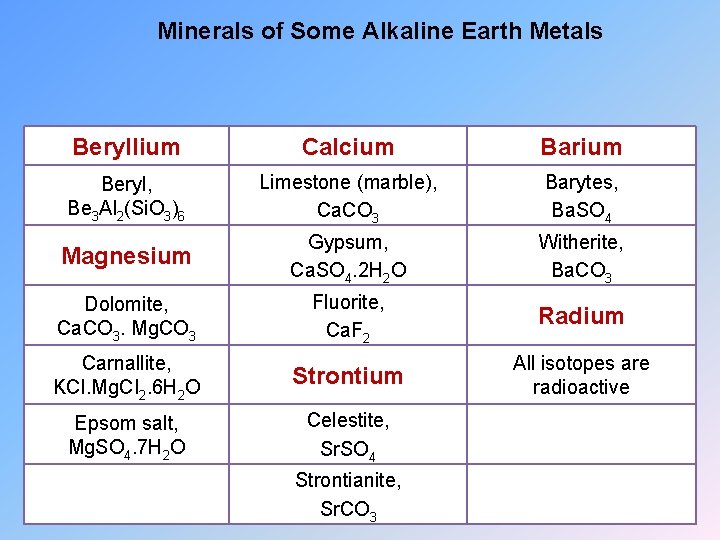
Minerals of Some Alkaline Earth Metals Beryllium Calcium Barium Beryl, Be 3 Al 2(Si. O 3)6 Limestone (marble), Ca. CO 3 Barytes, Ba. SO 4 Magnesium Gypsum, Ca. SO 4. 2 H 2 O Witherite, Ba. CO 3 Dolomite, Ca. CO 3. Mg. CO 3 Fluorite, Ca. F 2 Radium Carnallite, KCI. Mg. CI 2. 6 H 2 O Strontium All isotopes are radioactive Epsom salt, Mg. SO 4. 7 H 2 O Celestite, Sr. SO 4 Strontianite, Sr. CO 3

Chemical Properties • They are Covered by their oxides when they are exposed to air. • Burn in air with bright flame. • Activity of the metals increases from Be to Ba. • Reactions of Ca, Sr and Ba are easy with water. • Reactions of Be and Mg with water are difficult.

Chemical Properties • Ca (s) + 2 H 2 O(l) → Ca+2(aq) + 2 OH-(aq) + H 2(g) • Mg (s) + 2 H 2 O(l) → Mg(OH)2(s) + H 2(g) • 2 Mg (s) + O 2(g) → 2 Mg. O(s) • Mg (s) + H 2(g) → Mg. H 2(s) • Ca (s) + 2 HCI(aq) → Ca. Cl 2 + H 2(g) • Be (s) + 2 HCI(aq) → Be. CI 2 + H 2(g) • All oxides and hydroxides of alkaline earth metals show basic properties. • Oxides and its hydroxides of beryllium show amphoteric properties.

Compounds • Ionic • Have high melting and boiling points. • Thermally stable. • Less soluble in water than those of 1 A metals. • Solubility of salts increase from Be to Ba.

Calcium Oxide, Ca. O • Obtained by heating calcium carbonate (limestone) at 900°C. • Ca. CO 3(s) → Ca. O(s) + CO 2(g) Calcium Carbonate, Ca. CO 3 • It forms by evolving CO 2 gas from Ca(OH)2 solution.

Calcium Carbide, Ca. C 2 • Small gray solid pieces • Used widely in industry • Prepared by the reaction of calcium oxide (lime) and coke. Ca. O(s) + 3 C(s) ↔ Ca. C 2(s) + CO (g) Ca. C 2(s) + 2 H 2 O(l) → C 2 H 2(g) + Ca(OH)2(s)

Calcium Sulfate, Ca. SO 4. 2 H 2 O : Gypsum Ca. SO 4. 1/2 H 2 O : Plaster of Paris • Gypsum is a white substance and used for fabrication into wallboard.

Beryllium Uses • Very light element • To prepare some special alloys. • 2% Be in Cu alloy in springs. • Making glass for X-ray tubes. • In the aerospace industry.

Uses Magnesium • Light metal • Production of planes, missiles • Some light household items. • Flash in photography

Magnesium • To increase the hardness, durability and resistance to corrosion of alloys. • Additive in rocket fuels and signal rockets. • Mg(OH)2 is used as a stomach antacid.

Calcium Compounds • Construction materials, bleaching agents. • Ca. O in the production of some chemicals, steel, glass, paper and sugar. • Ca. C 2 is used to produce acetylene gas for welding. • In fertilizers and toothpaste

Barium and Its Compounds • Ba is used as a gas absorbent in vacuum tubes. • The alloys of Ba with Ni donate electrons so used in vacuum tubes and in ignitors and spark plugs. • Barium nitrate and barium chlorate are used to obtain green light in fireworks.

Barium and Its Compounds • Ba. SO 4 is used to take the photos of stomach and intestines. • Ba does not allow X-rays to pass like all elements with large atomic number. • The Ba+2 ion is toxic, but since the solubility of Ba. SO 4 in water is very low, its poisonous effect disappears.

Strontium • The salts of strontium in signal rockets and fireworks to produce red light. • Nuclear batteries.
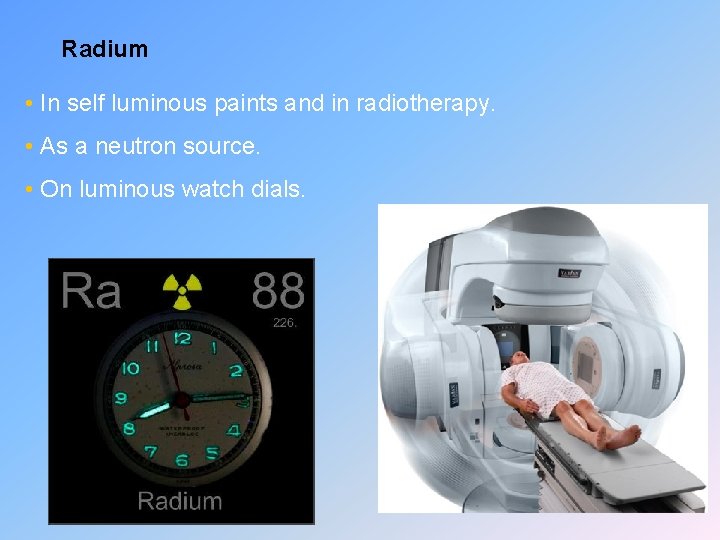
Radium • In self luminous paints and in radiotherapy. • As a neutron source. • On luminous watch dials.
- Slides: 22