When atoms combine metal metal alloy metal nonmetal

When atoms combine • metal + metal = alloy • metal + nonmetal = ionic compound • nonmetal + nonmetal = covalent compound or ionic compound

Ionic vs. Covalent Compounds

Covalent Compounds A tour of the molecules.

Molecular vs. Extended(Network) Compounds

Molecular vs. Empirical Formulas

Interactive Table 2. 3. 1 1. The first word in the compound name is the name of the first element in the compound formula. If the compound contains more than one atom of the first element, use a prefix (Table 2. 3. 2) to indicate the number of atoms in the formula. CS 2 First word in compound name: carbon N 2 O 4 First word in compound name: dinitrogen 2. The second word in the compound name is the name of the second element in the formula that has been changed to end with -ide. In all cases, use a prefix (Table 2. 3. 2) to indicate the number of atoms in the formula. CS 2 Second word in compound name: disulfide N 2 O 4 Second word in compound name: tetraoxide 3. The compound is named by combining the first and second words of the compound name. CS 2 carbon disulfide N 2 O 4 dinitrogen tetraoxide
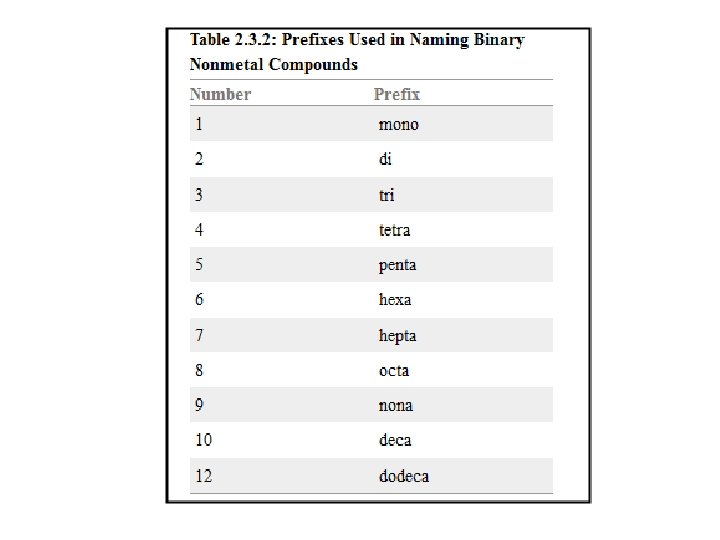

What is the name of N 2 O 5? What is the formula of disulfur decafluoride?


Which compounds exist, and which do not? wikipedia time

Which of the following does not exist? 1. sulfur dioxide 2. sulfur trioxide 3. sulfur tetraoxide

Which of the following does not exist? 1. chlorine monoxide 2. chlorine pentaoxide 3. dichlorine oxide

Poke around with elements: N, O, F, P, S, Cl See if you can find trends.

Monatomic Ions Trends? #p vs. #e-?
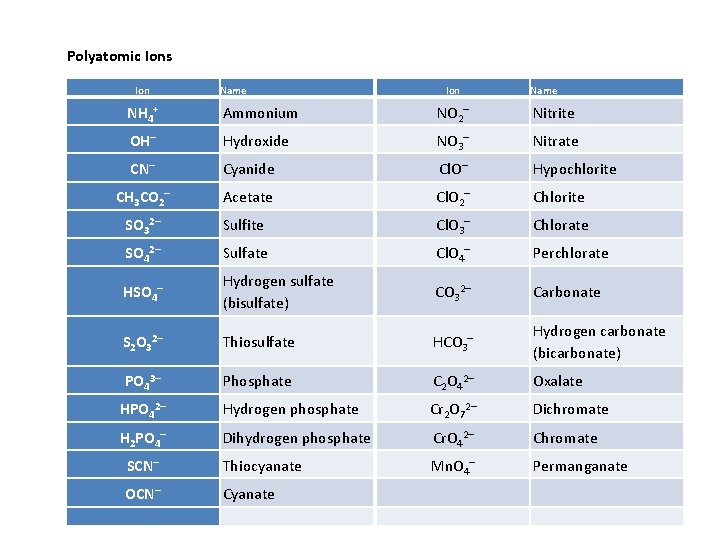
Polyatomic Ions Ion Name NH 4+ Ammonium NO 2– Nitrite OH– Hydroxide NO 3– Nitrate CN– Cyanide Cl. O– Hypochlorite CH 3 CO 2– Acetate Cl. O 2– Chlorite SO 32– Sulfite Cl. O 3– Chlorate SO 42– Sulfate Cl. O 4– Perchlorate HSO 4– Hydrogen sulfate (bisulfate) CO 32– Carbonate S 2 O 32– Thiosulfate HCO 3– Hydrogen carbonate (bicarbonate) PO 43– Phosphate C 2 O 42– Oxalate HPO 42– Hydrogen phosphate Cr 2 O 72– Dichromate H 2 PO 4– Dihydrogen phosphate Cr. O 42– Chromate SCN– Thiocyanate Mn. O 4– Permanganate OCN– Cyanate

Which Ionic Compounds Exist use sim

Naming Ionic Compounds use sim

Naming Ionic Compounds use sim

Naming Ionic Compounds use sim

Is a compound covalent, or ionic?
- Slides: 20