Ch 30 Compounding of NonSterile Products O Pharmacy
















































































- Slides: 82

Ch 30: Compounding of Non-Sterile Products O Pharmacy personnel engage in compounding when a practitioner prescribes a strength or type of medication that is not commercially available O It should never be done to create a product that is already available in the marketplace © 2013 -2015

Compounding of Non-Sterile Products O Compounding can be divided between “sterile” and “non-sterile” depending on the intended use of the product O Examples of non-sterile products may include tablets, capsules, and suppositories (all enteral routes of administration) O This session covers non-sterile products O We will cover sterile products in the future © 2013 -2015

Compounding of Non-Sterile Products O United States Pharmacopeia O Non-profit organization that sets standards O Helps ensure quality and safety O Medications sold and produced must meet these standards O Published in the USP-NF O Cited as Book – Chapter USP ### or USP <###> O Chapter that covers non-sterile compounding is: USP 795 © 2013 -2015

Compounding of Non-Sterile Products O USP Chapter 795 - “Pharmaceutical Compounding Non-Sterile Preparations” O Sets forth the rules for extemporaneous compounding O Defines standard of care O Provides an enforceable set of standards O Outlines responsibility of the compounder O Mandates a compounding record © 2013 -2015

Responsibilities of the Preparer Under USP 795 O USP Chapter puts responsibilities on the preparer of the product: O Have properly trained and capable associates O Appropriate and clean compounding area O Use ingredients that are; O Appropriate identity, quality, and purity O From a reputable source O Appropriate, clean, and well functioning equipment O Only authorized personnel are in the work vicinity O Compounding is reproducible according to the directions recorded – allowing for correction or errors or problems © 2013 -2015

The Compounding Record O The compounding record must contain: O A unique ID number for each product or batch O The date of preparation O The name of the preparer O The name of each ingredient O The lot number & beyond use date of each ingredient O The amount used of each ingredient O The patient’s name O The directions used to produce the compound O MUST be reproducible © 2013 -2015

Assigning a Beyond-Use Date O A Beyond-Use date must be assigned to the compounded product O The rule to remember is set forth by USP 795: “for non-sterile solid and liquid dosage forms that are repackaged in single-unit and unit-dose containers, the beyond-use date shall be one year from the date packaged or the beyond-use date on the manufacturer’s container, whichever is earlier” © 2013 -2015

Compounding Calculations O Percentage Calculations O How much of a final product is the actual drug O Broken up by the type of contents % weight in weight (w/w) % weight in liquid volume (w/v) % liquid volume in liquid volume (v/v) © 2013 -2015

Percent weight / weight O Used when both portions are in units of weight O 1% w/w = 1 g of active drug per 100 g of total product (1 g/100 g) © 2013 -2015

Percent weight / volume O Used when the solute is in units of weight and the solvent is in units of volume O 1% w/v = 1 g of active drug per 100 ml of total product (1 g/100 ml) © 2013 -2015

Percent volume / volume O Used when both portions are in the same units of volume O 1% v/v = 1 ml of active drug per 100 ml of total product (1 ml/100 ml) © 2013 -2015

Compounding Calculations O Always be sure you use the finished weight or volume in your calculation O Formula active ingredient <g or ml> / total amount <g or ml> x 100 = % Concentration Total Preparation <g or ml> Active ingredient <g or ml> © 2013 -2015

Example 1. 4 g of phenol is mixed with 9. 0 g of glycerin. What is the resulting percentage concentration of phenol? What do we know? O there is 1. 4 g of drug in the product O the final weight of the product will be 10. 4 g (1. 4 g + 9 g = 10. 4 g) © 2013 -2015

Example O Plug in the values and calculate the answer 1. 4 g 10. 4 g x 100 = % Concentration 0. 1346 x 100 = 13. 5% w/w © 2013 -2015

Another Example 4 g of KCl is dissolved in enough sweetened vehicle to make a total of 25 ml. What is the resulting percentage concentration? © 2013 -2015

Another Example 4 g of KCl is dissolved in enough sweetened vehicle to make a total of 25 ml. What is the resulting percentage concentration? 4 g 25 ml x 100 = % Concentration 0. 16 x 100 = 16% w/v © 2013 -2015

A Twist We can also used percentage concentration expressions as part of proportion calculations ie, How much KCl would be needed to make 240 ml of a 10% solution? © 2013 -2015

The Answer O Remember our definitions of percentage calculation? 1 g /100 ml = 1 % Therefore 10% would be 10 g / 100 ml and we can set up a proportion…. . 10 g = 100 ml x 240 ml 2, 400 = 24 g of KCl 100 © 2013 -2015

How about another…. How much propranolol must be used to prepare 240 ml of a 3% suspension? © 2013 -2015

How about another…. O How much propranolol must be used to prepare 240 ml of a 3% suspension? 3 g = 100 ml x 240 ml 720 = 7. 2 g 100 © 2013 -2015

Alligation O Alligation is useful whenever we are using two different concentrations of a drug product to arrive at a final concentration between the two components O For instance preparing a 10% product from 20% and 5% components © 2013 -2015

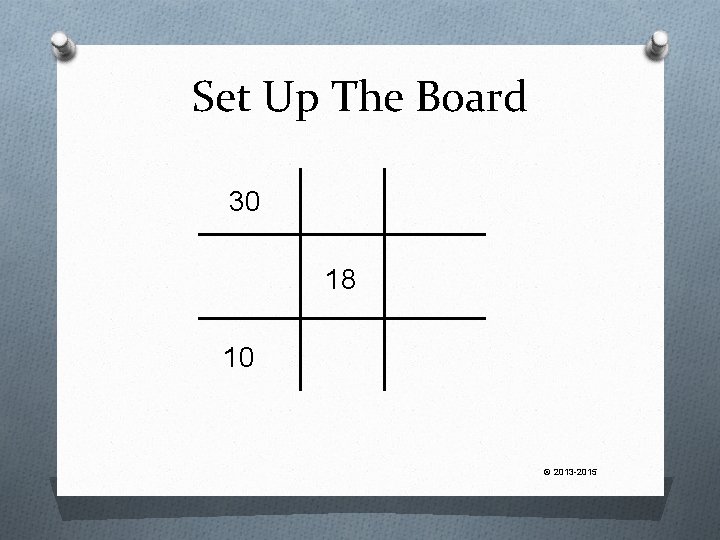
The Game Board O Alligations start with the traditional tic-tac-toe board © 2013 -2015

The Set Up Set up the problem like this: O the highest concentration goes in the upper left O the lowest concentration goes in the lower left O the final concentration goes in the center The highest Final Conc. The lowest © 2013 -2015

Let’s Try One How much of 20% Drug “A” cream is needed to mix with 5% Drug “A” cream to obtain 240 g of a final 10% concentration? © 2013 -2015

Set up the Board Put your values in the appropriate places The highest 20 10 The lowest The Final Concentration 5 © 2013 -2015

Now We…. O Go diagonally from each corner, through the middle and find the DIFFERENCE between the two numbers O Do NOT worry about negative numbers, you only want the DIFFERENCE 20 5 10 © 2013 -2015

The Answers Tell 20 10 5 The resulting 5 parts of the 20% differences tell us how much of each concentration we must use Therefore, out of 10 parts of the 5 % every 15 parts, 5 parts must be the 20% concentration. 15 total parts Or, another words, 5/15 of the product must be 20% cream © 2013 -2015

Finally O Since we know 5/15 of the product must be the 20% strength, all we do is multiply 5/15 times the amount of product we are making. 5 15 x 20% cream 240 g = 80 g of © 2013 -2015

Not So Bad, But…. . O Be SURE you are solving for the correct concentration O Label all calculations as to which strength you are solving for © 2013 -2015

Let’s Try Another We need to compound 300 g of an 18% concentration of Drug “B”. However, Drug “B” is available only in 30% and 10% concentrations. How much of the 10% cream will be needed? © 2013 -2015

Set Up The Board 30 18 10 © 2013 -2015

Calculate the Differences 8 parts of the 30 30% 18 10 12 parts of the 10 % 20 total parts © 2013 -2015

Solve We know that 12/20 of the final product must be the 10% 12 20 x 300 g = 180 g of 10% cream © 2013 -2015

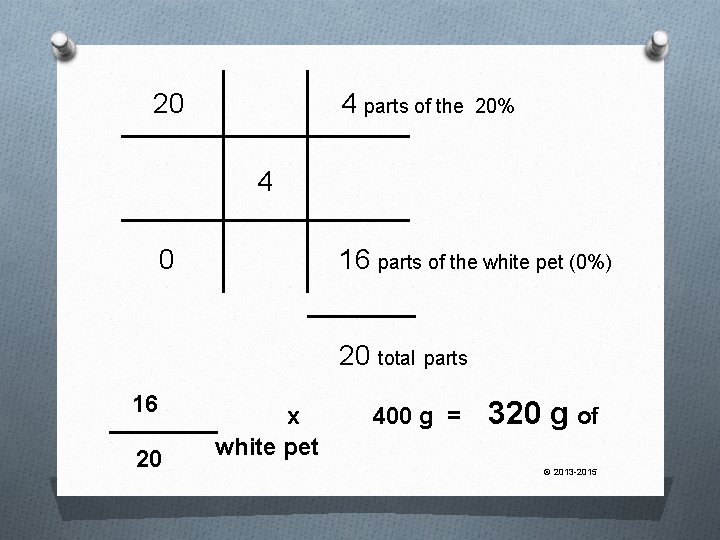
One More, Anyone? How much white petrolatum must be mixed with 20% ichthamol ointment to make 400 g of 4% product? © 2013 -2015

20 4 parts of the 20% 4 0 16 parts of the white pet (0%) 20 total parts 16 20 x white pet 400 g = 320 g of © 2013 -2015

Ratio Concentration O Once again we have standards to memorize 1: 1, 000 = 1 mg / 1 ml 1: 100 = 10 mg / 1 ml 1: 10 = 100 mg / 1 ml Hint – the ratios all start with 1, and the number of zeros always stay the same. They just get “squished” from one side of the equal sign to the other. © 2013 -2015

Example An order states that we need to give 3 mg of epinephrine. Our stock bottle is a 1: 100 concentration. How much should we give? © 2013 -2015

Example O An order states that we need to give 3 mg of epinephrine. Our stock bottle is a 1: 100 concentration. How much should we give? We know that 1: 100 means there is 10 mg in each milliliter. Therefore, we can set up a proportion 1 ml = 10 mg x 3 3 mg 10 = 0. 3 ml © 2013 -2015

Tonicity O Tonicity is the relative strength of a salt solution vs. the concentration normally found in the body Isotonic – Same as the body = 0. 9% Na. Cl Hypertonic – Higher than the body > 0. 9% Na. Cl Hypotonic – Lower than the body < 0. 9% Na. Cl O Know the values for Na. CL O 0. 9% is isotonic (Normal Saline) © 2013 -2015

Ch 31: Payment for Drug Products O Self Payment O The individual pays for his own medication in the entirety O Third Party Payment O Any party other than the patient who pays all or part of the prescription price © 2013 -2015

Copay & Deductible O Copayment O amount of money the patient is required to pay towards their drug expense O can be a fixed dollar amount or a percentage O Deductible O the amount of money a patient must pay towards their expenses before their insurance company begins to pay © 2013 -2015

Methods of Calculating the Selling Price O Based on the acquisition cost of the product, or O Based on the Average Wholesale Price (AWP) of the product © 2013 -2015

Based on Cost O Formula cost + markup + dispensing fee = selling price O Markup O the amount of profit you expect to make on the item (markup = cost x gross profit expected) O Dispensing Fee O a calculated amount to cover the fixed costs of filling an individual prescription © 2013 -2015

Example 60 tabs of medicine cost us $23. 99. We have a dispensing fee of $4. 00 and a markup of 25%. What is the selling price for the prescription? We know our cost for the amount dispensed = $23. 99 So, using cost + markup + dispensing fee = selling $$ $23. 99 + ($23. 99 x 0. 25) + $4. 00 = $23. 99 + $6. 00 + $4. 00 = $33. 99 © 2013 -2015

Another Example Our pharmacy uses a 30% markup. Our dispensing fee is $3. 25. The drug costs us $45. 99. What is the selling price? © 2013 -2015

The Answer Cost + markup + dispensing fee = selling price $45. 99 + ($45. 99 x 0. 30) + $3. 25 = $45. 99 + $13. 80 + $3. 25 = $63. 04 © 2013 -2015

A Twist Our pharmacy has a dispensing fee of $4. 99 Our markup is 20% The drug costs us $239. 99 for a bottle of 500 tabs The prescription calls for #60 tabs What would our selling price be? © 2013 -2015

The Answer O First you must use a proportion calculation $239. 99 x = 500 tabs 60 tabs $14399. 40 = $28. 80 500 tabs O Next you use the selling price calculation $28. 80 + ($28. 80 x 0. 20) + $4. 99 = $28. 80 + $5. 76 + $4. 99 = $39. 55 © 2013 -2015

AWP Method O Average Wholesale Price O much like a car sticker price O nobody actually pays this price O that’s why there is normally a discount off of AWP involved in this calculation O The formula AWP ± percentage + dispensing fee = selling price © 2013 -2015

AWP Example The AWP for the medication dispensed is $35. 77. The insurance company allows reimbursement at AWP less 15% plus $2. 00 dispensing fee. What is the selling price? We know our AWP for the amount dispensed = $35. 77 So, using AWP ± Percentage + dispensing fee = selling $$ $35. 77 - ($35. 77 x 0. 15) + $2. 00 = $35. 77 - $5. 37 + $2. 00 = $32. 40 © 2013 -2015

Another Example Our prescription calls for 45 capsules. The AWP for a bottle of 100 caps is $43. 99. The Insurance reimbursement is AWP less 14% plus a $3. 50 dispensing fee. The patient also must pay a $5. 00 copayment. How much should we bill the insurance company? …… Wow! What a Mess! © 2013 -2015

Calculate the Answer O First calculate the AWP for the amount dispensed using a proportion $43. 99 = 100 caps 45 caps x $1979. 55 = $19. 80 100 caps …. . then © 2013 -2015

Calculate the Answer O Use the AWP formula AWP ± Percentage + dispensing fee = selling price $19. 80 – ($19. 80 x 0. 14) + $3. 50 = $19. 80 - $2. 77 + $3. 50 = $20. 53 O BUT DON’T FORGET THE COPAYMENT! $20. 53 - $5. 00 = $15. 53 © 2013 -2015

Ch 32: Third Party Billing Process O Two main types of third party payers: O Private Insurance O Either employer paid or privately purchased O Government O Includes federal, state, or local programs O Biggest are Medicare and Medicaid © 2013 -2015

“Parts” of Medicare O Part A O Covers institutional care O ie, hospital charges O Part B O Physician & outpatient services O Durable medical equipment O Part C O Medicare advantage plans O Part D O Prescription drugs © 2013 -2015

Medicaid O Covers indigent patients O By law, must cover hospital, medical, and long term care charges O Prescription drug coverage is actually optional, although most states provide some type of coverage O Amount and type of coverage will vary between states O Coverage is not usually transferrable between states © 2013 -2015

Managed Care O System that integrates both financial and delivery of health care O Managed Care Organizations (MCO) form networks of providers with whom they contract O Some contracts pay providers a set dollar amount per enrolled patient O Means the MCO assumes the financial risk O The MCO then “manages” the care through utilization and cost controls © 2013 -2015

The Pharmacy Benefit Manager (PBM) O PBMs are responsible for the processing and payment of prescription drug claims for the insurance companies O When we submit a claim for the customer, we are sending it to the PBM, not to the insurance company O PBMs follow the directions from the insurance company’s contract with the insured, but they also are charged with reducing health costs when possible © 2013 -2015

Methods to Reduce Third Party Prescription Drug Costs O Preferred provider networks O Negotiated price discounts from pharmacies O Negotiated rebates from manufacturers O Mandatory Mail Order O Deductibles and Copayments O Formularies O Tier structures O Prior Authorizations O Medication Therapy Management O Mandatory Education © 2013 -2015

Multi-Tiered Structures O Drugs are ranked by the MCO according to certain predefined factors, such as: O Cost O Generic availability O Plan formulary O With each successive increase in tier ranking, the amount of the patient’s copay increases © 2013

Information on the Insurance Card O Cardholder’s Name O NOT always the patient’s name © 2013 -2015

Information on the Insurance Card O Cardholder’s ID Number O Used for all covered individuals on the card O Also requires a dependent number that is rarely on the card © 2013 -2015

Information on the Insurance Card O Dependent Numbers 00 = cardholder 01 = spouse 02 = oldest dependent 03 = next oldest dependent 04, 05, 06, etc = with each © 2013 -2015 dependent

Information on the Insurance Card O BIN Number O Tells us who to bill the claim to © 2013 -2015

Information on the Insurance Card O Group Number O Identifies which plan the cardholder belongs to © 2013 -2015

Required Information When Billing Third Party Claims O In addition to all of the required information for a prescription to be valid, the following items must also be present on the prescription: O Dispense as Written Codes (DAW) O Day Supply O Actual Package Size Used O Diagnosis Codes © 2013 -2015

Dispense as Written Codes O Determined by prescriber’s notation on the prescription O Common DAW Codes: 0 = no product selection indicated (may use generic) 1 = Prescriber wants the brand name product 2 = Patient wants the brand name product 3 = Pharmacist wants the brand name product 4 = generic not in stock 5 = brand drug dispensed as generic 6 = special override 7 = brand mandated by law 8 = generic not available in marketplace © 2013 -2015

Day Supply O The day supply is calculated using the amount of medication dispensed divided by the daily expected usage of the medication O Monitoring the day supply can give several pieces of information to the PBM and the pharmacy: O Is the drug being prescribed correctly? O Is the patient taking too much or too little of the medication based on expected use? (called “adherence”) O The PBM can then approve or deny the © 2013 -2015 claim based on use

Actual Package Size Used O Integrity of the billing system is dependent on accurate information being submitted O The NDC number of the product used is submitted with the claim O NDC indicates package size used O Acquisition cost varies by the product size O Payment is based on the acquisition cost O You must use the correct NDC for the item used O It can be considered insurance fraud to use an incorrect NDC © 2013 -2015

Diagnosis Codes O ICD-10 codes O Identifies main disease state, plus: O Disease variants, unusual characteristics, and cause of the condition O Codes can contain letters in addition to numbers O Diagnosis codes are required on all Medicare Part B prescriptions © 2013 -2015

Third Party Claim Rejections O When a claim is submitted, the PBM will conduct a drug utilization review (DUR) O Common reasons for rejections: O Refill too soon O Invalid day supply O Prior authorization needed O Rejections require investigation and resolution (when possible) by pharmacy staff O Online documentation through DUR override codes © 2013 -2015

Reconciliation and Audits O Reconciliation is the act of comparing submitted insurance claims with amounts actually paid O May require claim correction and resubmission O Audits are conducted by the PBMs O Do the prescriptions actually exist? O Did the patient pick up the prescription? O Does the pharmacy stock the package size they billed for? O Audit exceptions can trigger forfeited money to PBM © 2013 -2015

Ch 33: Return of Pharmaceuticals O Reasons for returns O returns from patients O over stock O ordered by mistake O expired drugs O recalls O What we can do with the product depends on many factors © 2013 -2015

Returns from Outpatients O We have lost control over the product O where was it stored? O was it contaminated? O is it even the same product that you dispensed? O Never place the drug back in stock O do not risk other patients health O Controlled substances O recommendation is not to take controlled substances back. O let the patient destroy the drugs © 2013 -2015 O give them a refund if desired

Returns from Inpatient Areas O We had control over the medication while it was gone O We can return unit dosed medications back into stock O Exemption exists for bulk drugs O Sent home with patient or destroyed © 2013 -2015

Manufacturer Recalls O FDA or manufacturer can issue recalls O FDA has the power to seize affected medicine O Rated into Classes by the severity of damage that could be caused with use © 2013 -2015

FDA Recall Classes O Class I O serious health problems or death O Class II O temporary health problems or slight risk of serious health problem O Class III O unlikely to cause serious health problem but still violates FDA rules © 2013 -2015

Returns to Wholesalers for Credit O Each will have their own rules for returns O Credit invoices must be filed with your other invoices from the wholesaler O Returns of schedule 2 drugs require the use of a 222 form from the wholesaler © 2013 -2015

Return of Drugs for Destruction O Most returns will be of drugs that should no longer be used O Beyond-use date O Recalls O Returns from customers O Returned to “reverse distributors” O Company contracted who handles all returns at a central location O Advantages: O Simplifies O Makes obtaining and credits easy O Helps stop diversion of any beyond-use drugs © 2013 -2015

Returns of Controlled Substances for Destruction O Many return centers exist O Schedule 2 drugs require a 222 form from the return center O Sent through reverse distributor or in rare cases to the DEA O Maintain return paperwork with your invoices © 2013 -2015

Hazardous Substance Returns O Must be returned to a licensed Hazardous Waste Disposal Company O Process O Receipt for waste when picked up (“cradle”) O Confirmation sent to pharmacy when the substance is destroyed (“grave”) O BOTH copies must be kept on file O Gives “cradle to grave” accountability © 2013 -2015

Questions? © 2013 -2015