Amino Acids Side Groups Polar Charged ACIDIC negatively










- Slides: 24

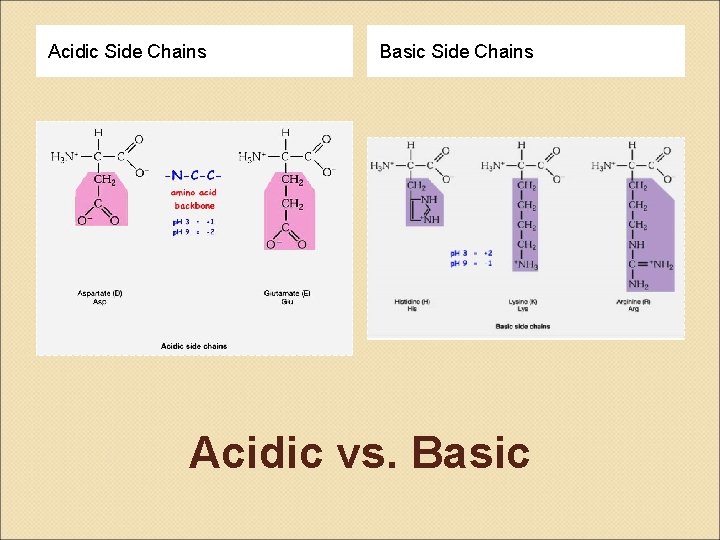
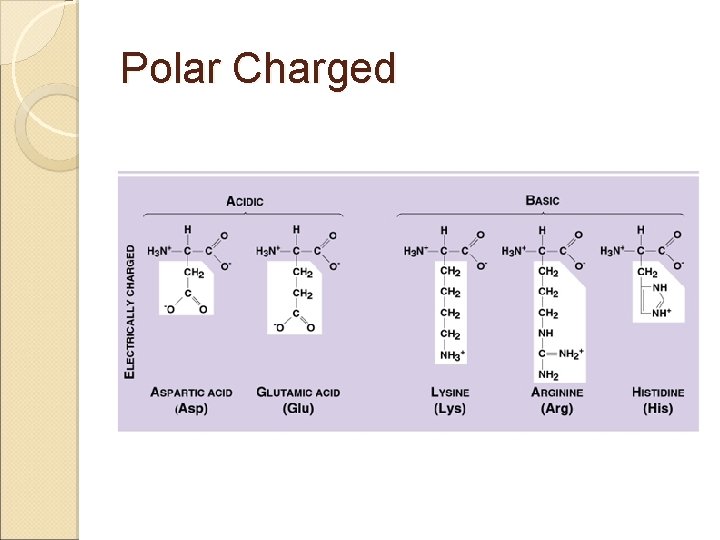
Amino Acids & Side Groups Polar Charged ◦ ACIDIC negatively charged amino acids ASP & GLU R group with a 2 nd COOH that ionizes* above p. H 7. 0 Polar Charged ◦ BASIC positively charged amino acids LYS, ARG, HIS R group with a 2 nd amide* that protonates below p. H 7. 0

Acidic Side Chains Basic Side Chains Acidic vs. Basic

Polar Charged

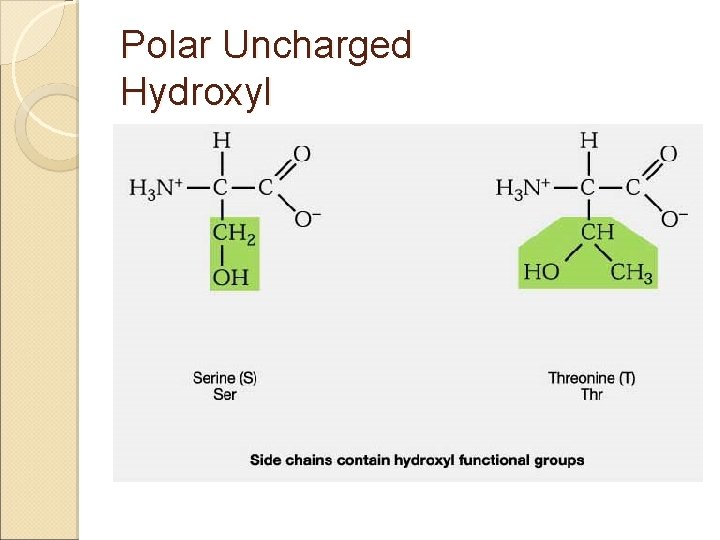
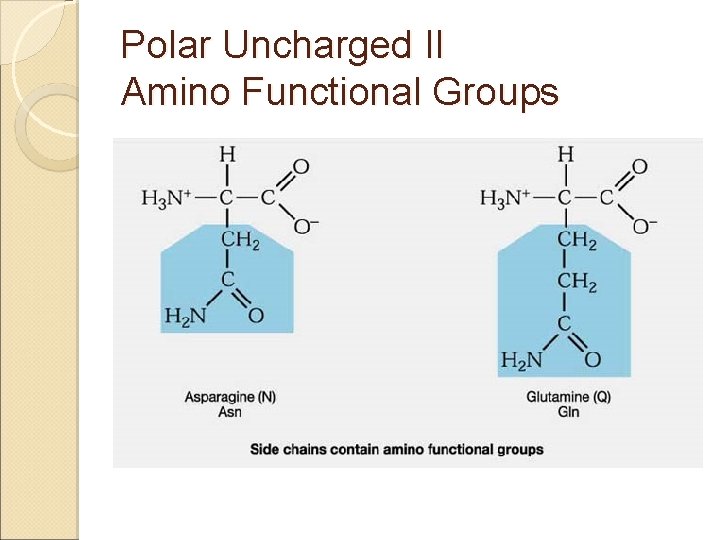
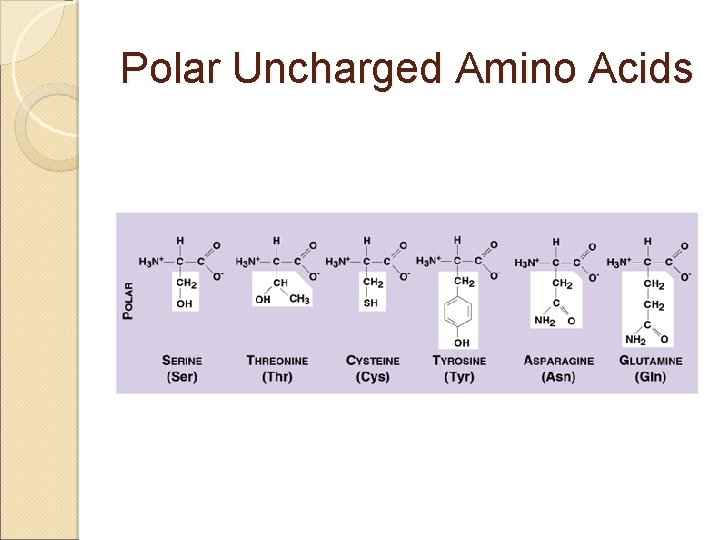
Amino Acids & Side Groups Polar Uncharged ◦ THR, TYR, ASN, GLN (cys) are soluble in water, i. e. , hydrophilic (attract H -bonds) Contain hydroxyl or amino functional groups

Polar Uncharged Hydroxyl

Polar Uncharged II Amino Functional Groups

Polar Uncharged Amino Acids

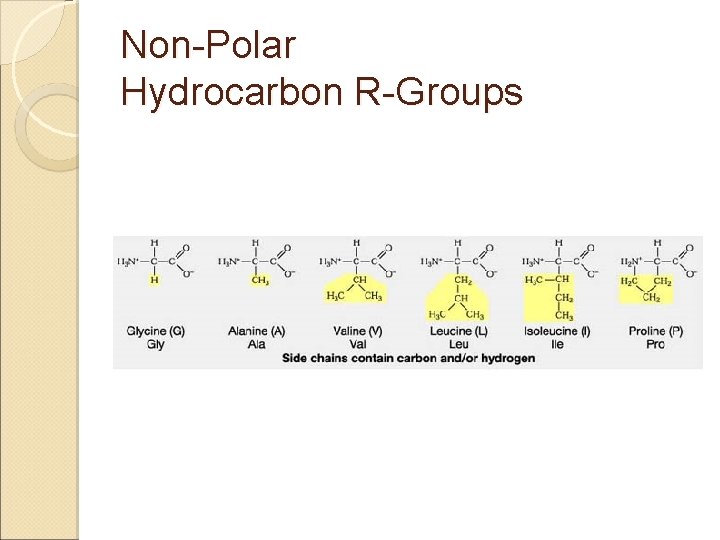
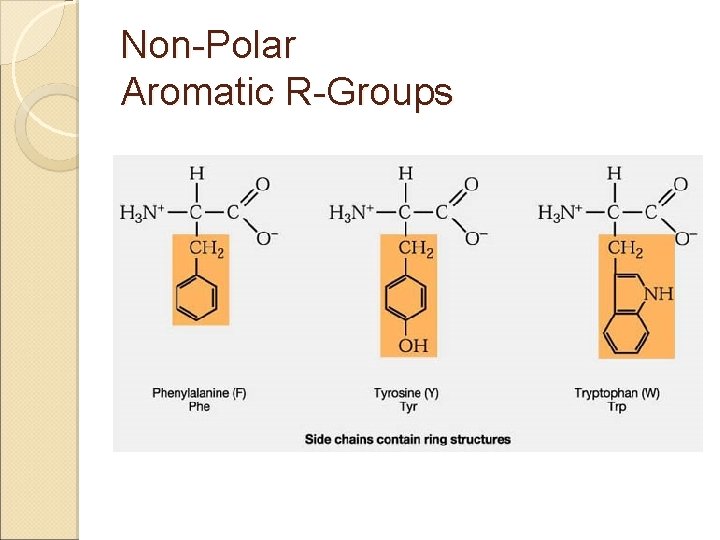
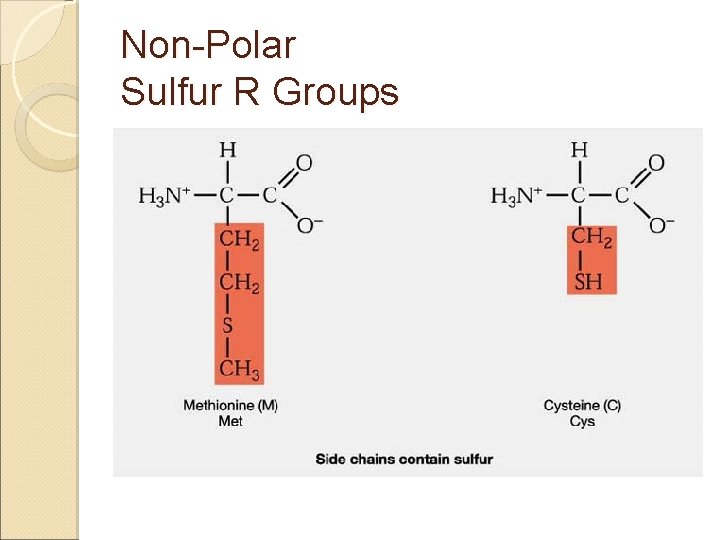
Amino Acids & Side Groups NON-POLAR (aliphatic) ◦ Includes GLY, ALA, VAL, LEU, ILE, PRO all contain only hydrocarbons groups = hydrophobicity AROMATIC (hydrophobic non-polars) ◦ PHE & TRP (TYR) all contain R groups with ring structures* or Sulfur* R groups with sulfur ◦ MET, CYS

Non-Polar Hydrocarbon R-Groups

Non-Polar Aromatic R-Groups

Non-Polar Sulfur R Groups

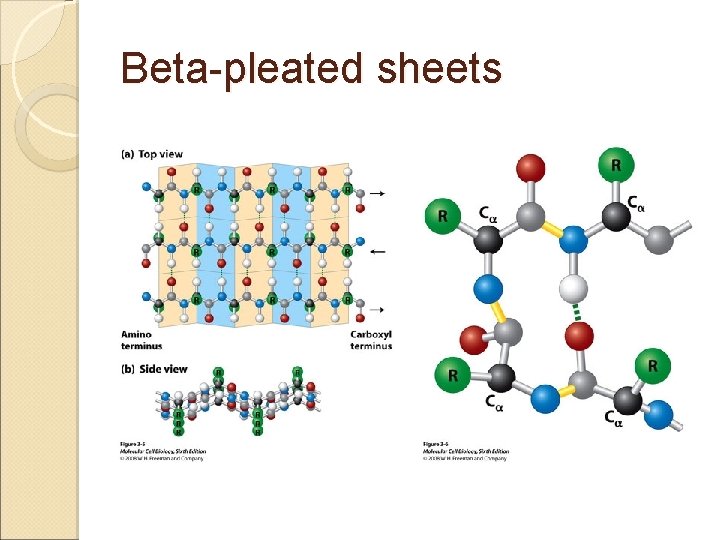
Alpha Helix The most common polypeptide helix Stabilized by extensive hydrogen bonding Hydrogen bonds extend up from the oxygen from the carbonyl group to the NH group of a peptide linkage ◦ This was shown in class via the visuals There approximately 4 peptide bond links up stream between the atoms involved in the hydrogen bonds Each turn of an alpha helix contains 3. 6 amino acids. Beta-pleated sheets Unlike the alpha helix, composed of two or more peptide chains • Polypeptide chains are joined by hydrogen bonds • When the hydrogen bonds are formed between the polypeptide chains they are termed interchains. • The polypeptide chains can run parallel to each other or anti-parallel • – Recall the “ends” of a polypeptide chain • C-terminus • N-terminus/Amino-terminus Secondary Protein Structures

Alpha Helix

Beta-pleated sheets

Beta-pleated Sheets and Alzheimer’s Disease The amyloid protein, a class of fibrous proteins, is deposited in the brain. • Individuals, that have Alzheimer’s Disease, have the amyloid protein composed of twisted Beta-pleated sheet fibrils whose three-dimentional structure is virtually identical to that of silk fibrils • – Silk • Contain Beta-pleated sheet protein structures

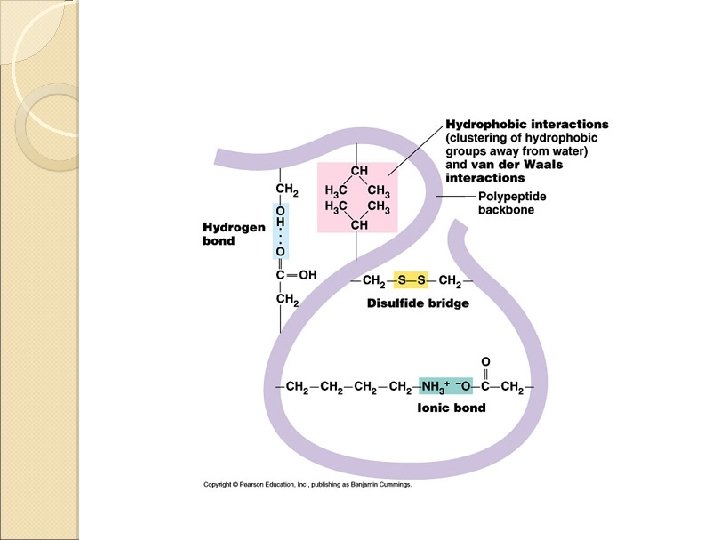
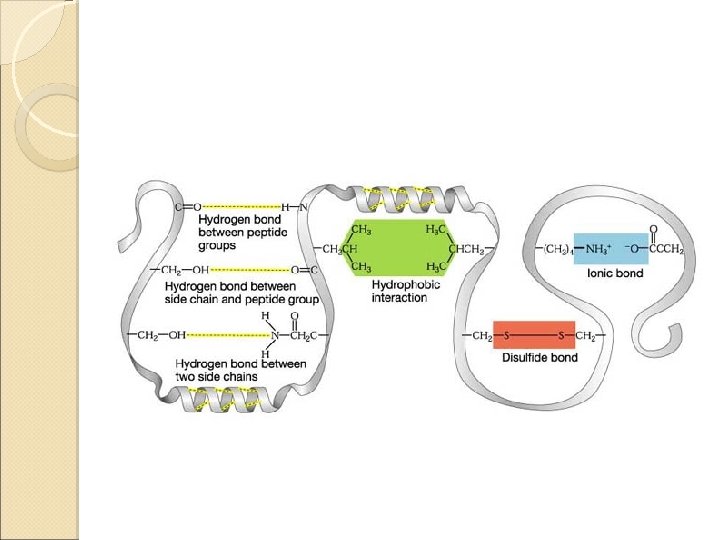
Tertiary Structure Interactions stabilizing Tertiary Structures ◦ Four were mentioned in class Disulfide Bonds Hydrophobic interactions Hydrogen bonds Ionic interactions

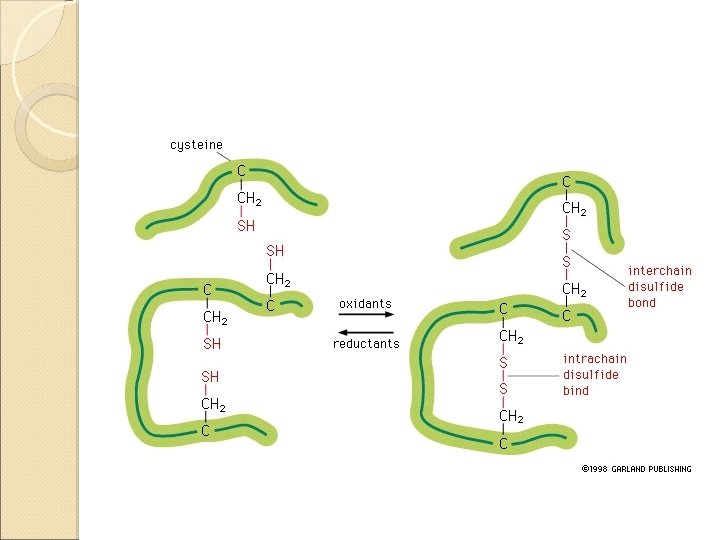
Disulfide Bonds A disulfide bond is a covalent linkage formed by the sulfhydryl group (-SH) of two cysteine residues to form cystine • The folding of the polypeptide chain brings the cysteine residues near each other • Disulfide linkage contributes to the stability of the three-dimensional shape of the protein molecule • Disulfide bonds are found in proteins that are secreted by cells • – Thought that these strong covalent bonds help stabilize the structure of proteins and help prevent them from becoming denatured in the extra-cellular environment


Hydrophobic Interactions • Recall that amino acids with non-polar side chains tend to be located in the interior of the polypeptide – Here, they associate with other hydrophobic amino acids • Special Note – Proteins located in non-polar (lipid) environments such as the phopholipid bilayer, tend to be in an opposite form • Hydrophobic amino acids are located on the surface • Hydrophilic amino acids are located on the interior

Ionic Interactions Negatively charged groups interact with positively charged groups ◦ Negatively charged groups (-COO-) in the side chain of aspartate or glutamate ◦ Positively charged groups (-NH 3+) in the side chain of lysine



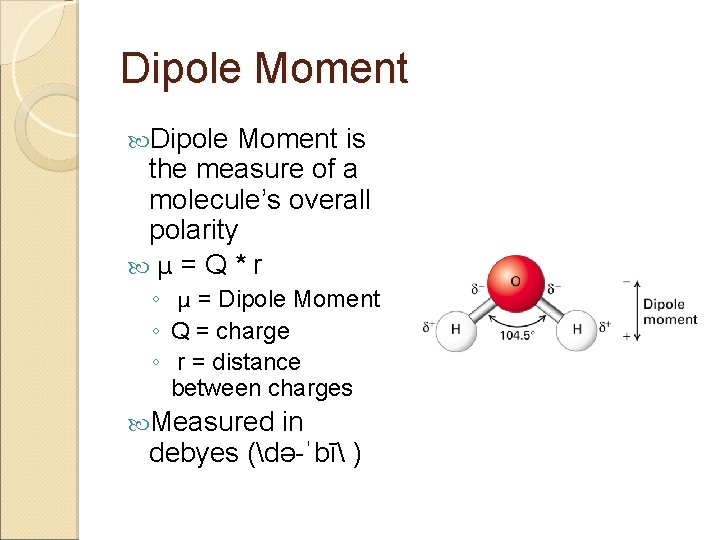
Dipole Moment is the measure of a molecule’s overall polarity μ = Q * r ◦ μ = Dipole Moment ◦ Q = charge ◦ r = distance between charges Measured in debyes (də-ˈbī )

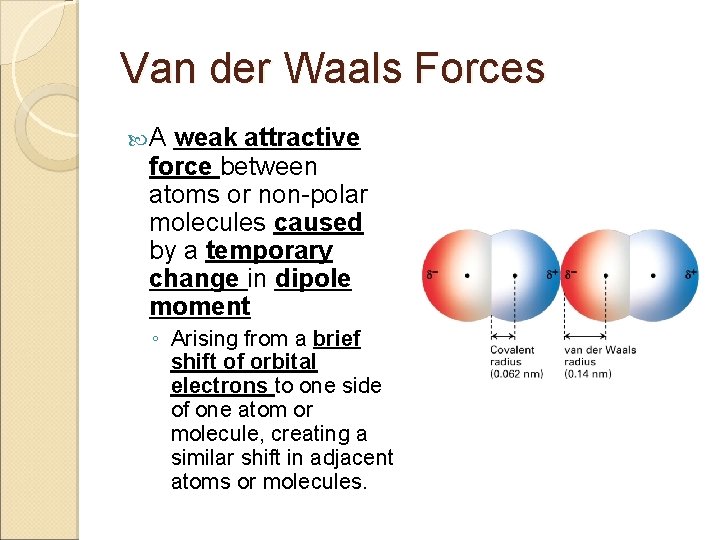
Van der Waals Forces A weak attractive force between atoms or non-polar molecules caused by a temporary change in dipole moment ◦ Arising from a brief shift of orbital electrons to one side of one atom or molecule, creating a similar shift in adjacent atoms or molecules.