Amino Acids Disposal of Nitrogen Overview Amino acids


























- Slides: 45

Amino Acids: Disposal of Nitrogen

Overview Amino acids are not stored in the body So, Amino Acids must be obtained from : 1 -Diet 2 -Synthesized de novo 3 -produced from normal protein degradation (turnover) Amino Acids in excess of biosynthetic needs of the cell Not stored But: rapidly degraded first step: removal of the a-amino group a-ketoacid (of a. a. ) + ammonia exc. in urine UREA

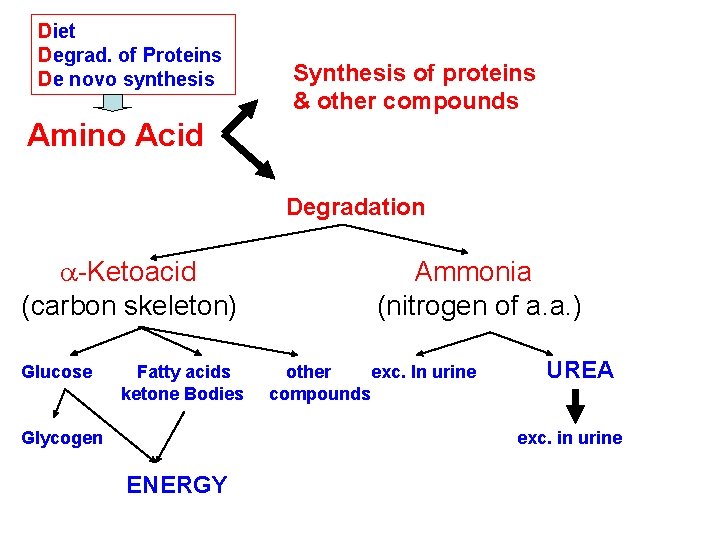
Diet Degrad. of Proteins De novo synthesis Synthesis of proteins & other compounds Amino Acid Degradation a-Ketoacid (carbon skeleton) Glucose Fatty acids ketone Bodies Glycogen Ammonia (nitrogen of a. a. ) other exc. In urine compounds UREA exc. in urine ENERGY


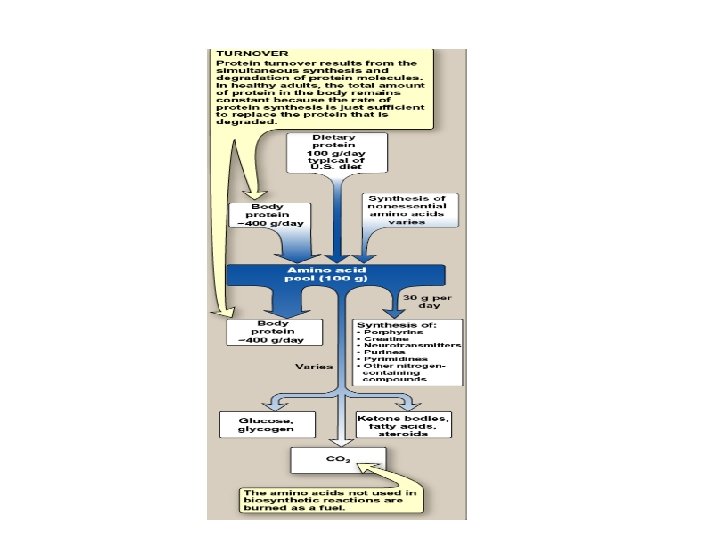
Amino Acid Pool 100 grams of a. a. Collected from: Dietary Proteins (by hydrolysis) Degradation of Tissue Proteins De novo synthesis of a. a. Fate of amino acids obtained by tissue protein hydrolysis 75% of amino acids 25% of amino acids used to synthesize new proteins metabolized precursor for other compounds compensated by dietary proteins ---- amino acids

Protein Turnover • Protein turnover results from the simultaneous synthesis & degradation of tissue proteins • The total amounts of protein in the body is constant because the rate of protein synthesis is just sufficient to replace the degraded protein • Protein turnover leads to hydrolysis & synthesis of 300400 grams of body protein each day

Rate of Protein Turnover • Short-lived proteins: minutes – hours half-life (as many regulatory proteins & misfolded proteins) • Long-lived proteins: days – weeks half-life (majority of proteins in the cell) • Structural proteins: months – years half-live (as collagen)

Protein Degradation By Two Major Enzyme Systems 1 - Ubiquitin-proteasome mechanism • energy-dependent • mainly for endogenous proteins (proteins synthesized within the cell) 2 - Lysosomes • non-energy-dependen • primarily for extracellular proteins as: - plasma proteins that are taken into cells by endocytosis - cell surface membrane proteins: for receptor-mediated endocytosis

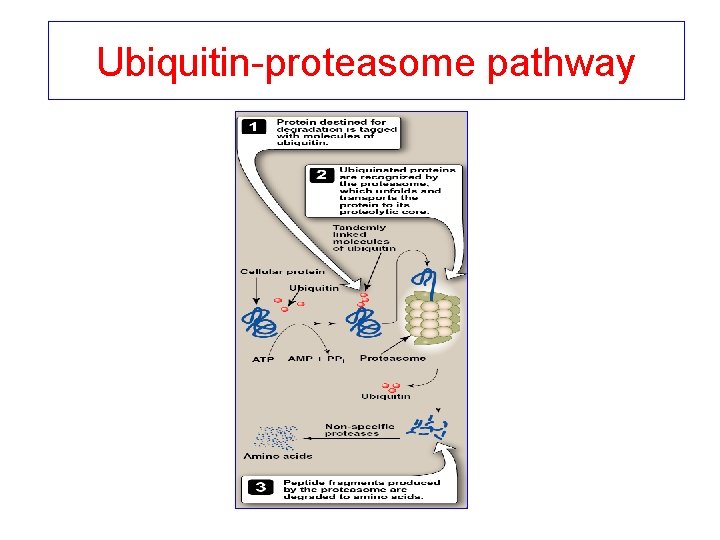
Ubiquitin-proteasome pathway

Mechanism of action of ubiquitin-proteasome system • Protein is covalently attached to ubiquitin (small globular protein) • More ubiquitin is added to form polyubiquitin chain (protein is tagged with ubiquitin) • Ubiquitin-tagged protein is recognized by the proteasome (proteolytic molecule) • The proteasome cuts the target protein into fragments (requires ATP) • Fragments are cut by non-specific proteases to amino acids

Chemical Signals for Protein Degradation • Protein degradation is influenced by some structural aspect of the protein Examples: • The half-life of a protein is influenced by the nature of the N-terminal residue: with serine: long-lived proteins (half-life is more than 20 hours) with aspartate: short-lived (half-life is about 3 minutes) • Proteins rich sequence containing PEST: rapidly degraded (i. e. with short half-life) PEST sequence: proline, glutamate, serine & threonine

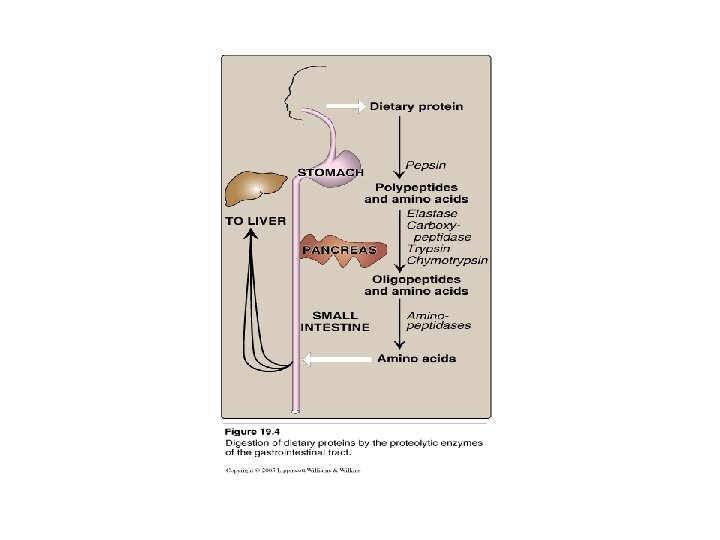
Digestion of Dietary Proteins • Most of the nitrogen in diet is consumed in the form of protein • Dietary protein/day: 70 – 100 grams • Dietary proteins must be hydrolyzed to amino acids by proteolytic enzymes • Proteolytic enzymes are produced by three different organs stomach pancreas small intestine


1 - Digestion of Proteins By Gastric Secretion • Gastric secretions contains: 1 - hydrochloric Acid: - p. H 2 – 3 (for activation pepsinogen) - kills some bacteria - denature proteins 2 - Pepsin: secreted as peopsinogen (inactive zymogen) activated to pepsin by Hcl or autocatalytically by pepsin product of hydrolysis of proteins: peptides + few free amino acids (oligo- + poly-)

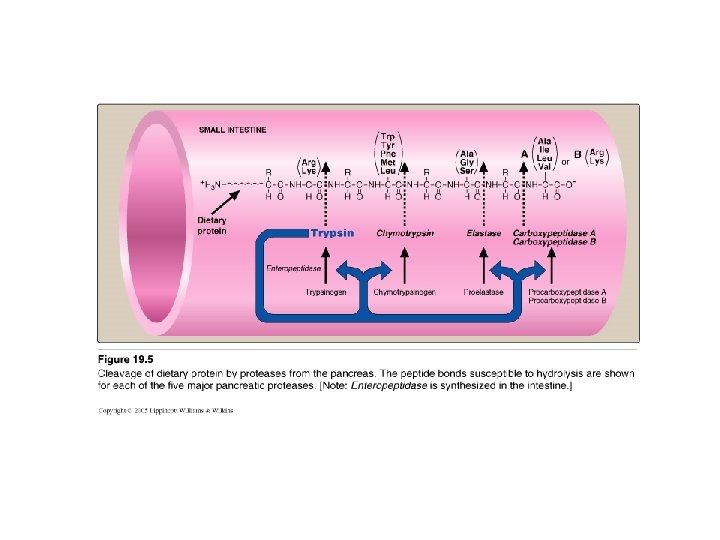
2 - Digestion of Proteins by Pancreatic Enzymes • • On entering the small intestine: Pancreatic Proteases: Large polypeptides are further cleaved to oligopeptides & free amino acids PROTEASES: • Secreted as inactive zymogen from pancreatic cells • Secretion of zymogens is mediated by the secretion of cholecystokinin & secretin (polypeptide hormones of GIT) • Activation of zymogen; by enteropeptidase (enterokinase): enzyme present on the luminal surface of intestinal mucosal cells converts trypsinogen to trypsin (by removal of hexapeptide from trypsinogen) Trypsin converts other trypsinogen molecules to trypsin Trypsin activates all other pancreatic zymogens (chymotrypsinogen, proelastase & procarboxypeptidases) • Specificity: example Trypsin: cleaves when the carbonyl group of the peptide bond is contributed by argenine or lysine


Abnormalities in Protein Digestion • Deficiency of pancreatic secretion occurs due to chronic pancreatitis, cystic fibrosis or surgical removal of the pancreas • Digestion & absorption of fat & protein is incomplete • Abnormal appearance of lipids (steatorrohea) & undigested protein in feces

3 - Digestion of oligopeptides by enzymes of the Small Intestine • Aminopeptidases - on the luminal surface of the intestine - is an exopeptidase - cleaves the N-terminal residue from oligopeptides to produce free amino acids & smaller peptides

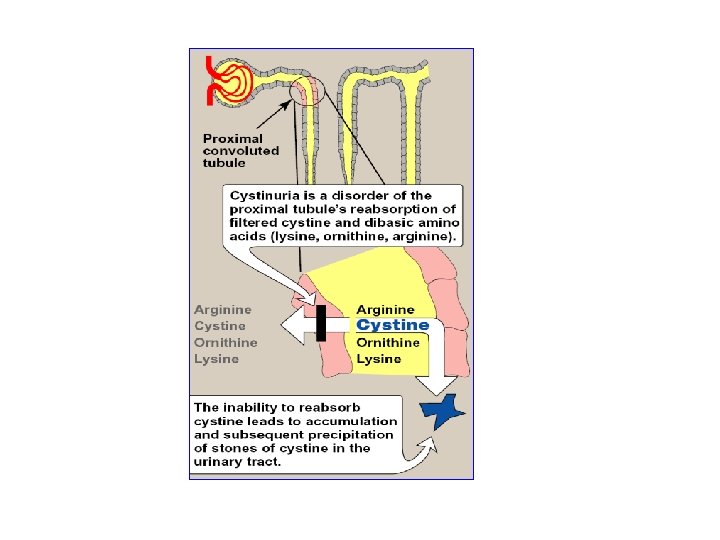
Transport of Amino acids into cells • Movement of a. a. to cells is performed by active transport (requires ATP) • Seven different transport systems: with overlapping specificity for different amino acids for example: cystine, ornithine, argenine & lysine are transported in kidney tubules by one transporter • In cystinuria: Inherited disease , one of the most common inherited dis. 1: 7000 individuals • defective carrier system for these 4 amino acids appearance of all 4 amino acids in the urine precipitation of cystine to form kidney stones (may block urinary tract)


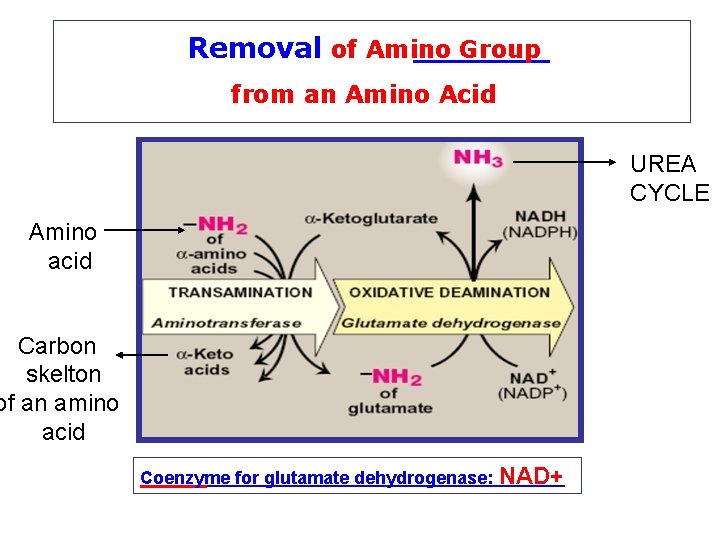
Removal of Nitrogen from Amino Acids Removing the a-amino group • Essential for producing energy from any amino acid • An obligatory step for the catabolism of all amino acids

Amino Acids

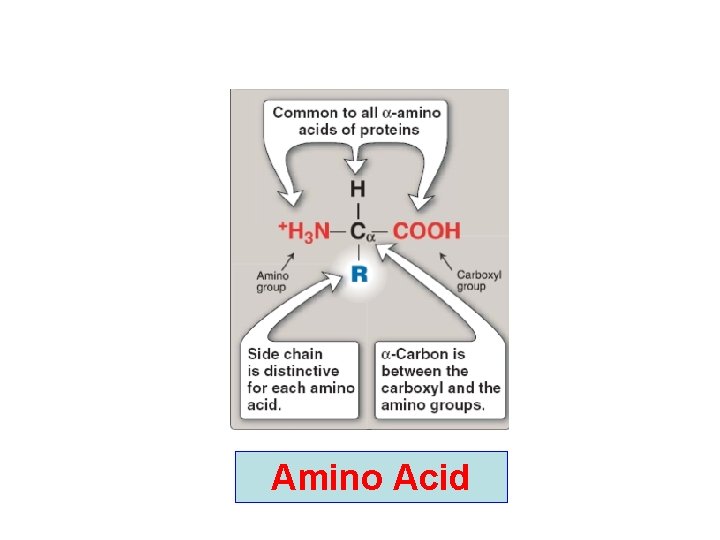
Amino Acid

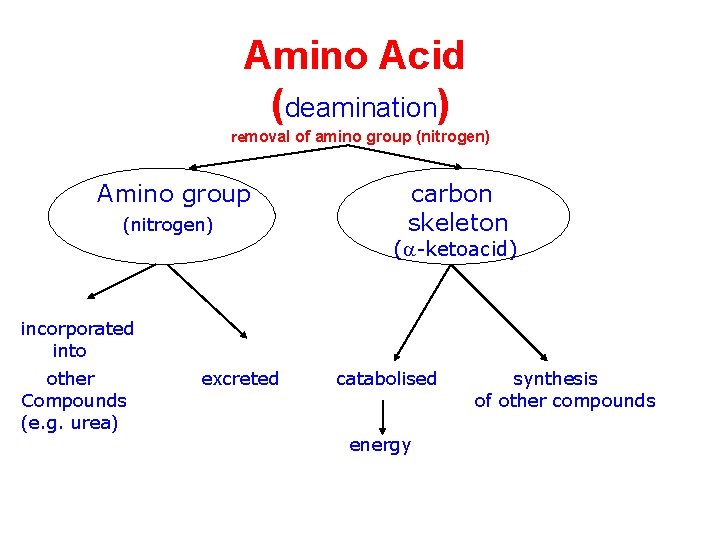
Amino Acid (deamination) removal of amino group (nitrogen) Amino group (nitrogen) carbon skeleton (a-ketoacid) incorporated into other Compounds (e. g. urea) excreted catabolised energy synthesis of other compounds

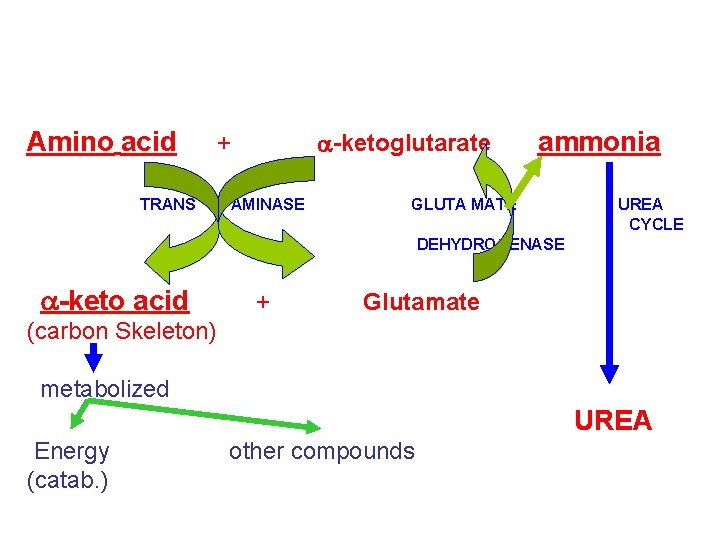
Deamination Pathways Amino group (nitrogen) is removed from an amino acid by either 1 - Transamination 2 - Oxidative (BY: TRANSAMINASES) Deamination (BY: GLUTAMATE DEHYDROGENASE)

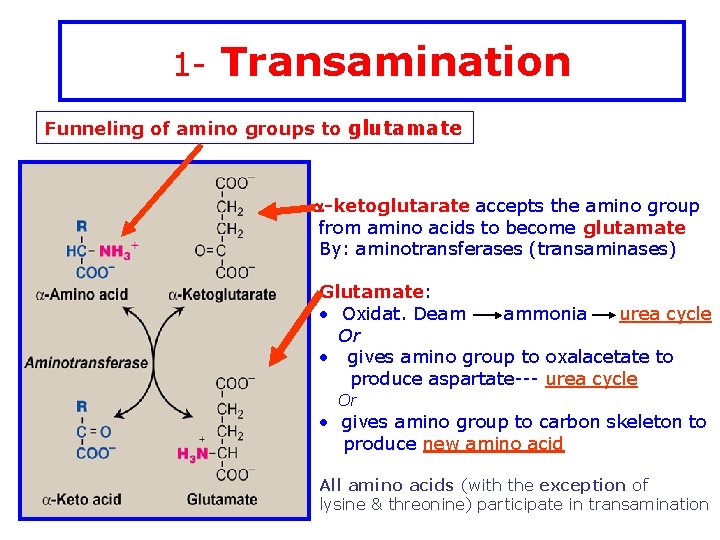
1 - Transamination Funneling of amino groups to glutamate a-ketoglutarate accepts the amino group from amino acids to become glutamate By: aminotransferases (transaminases) Glutamate: • Oxidat. Deam ammonia urea cycle Or • gives amino group to oxalacetate to produce aspartate--- urea cycle Or • gives amino group to carbon skeleton to produce new amino acid All amino acids (with the exception of lysine & threonine) participate in transamination

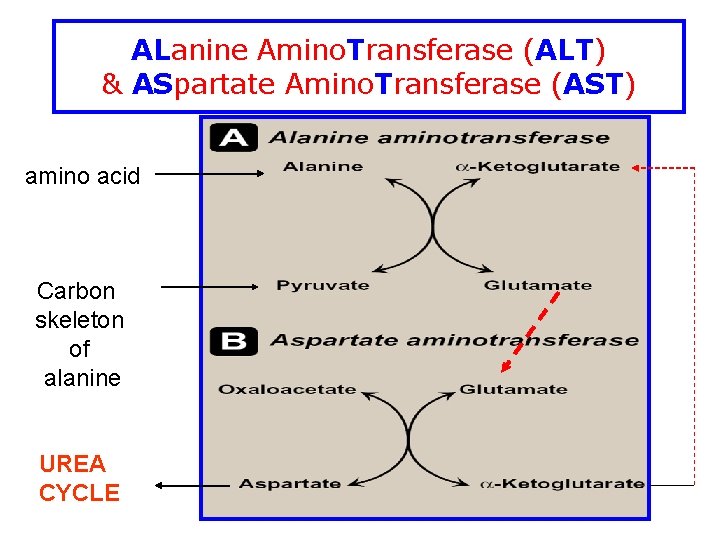
Substrate Specificity of Aminotransferases • Each aminotransferase is specific for one or few group of donors • Aminotransferase is named after the specific amino group donor (amino acid that donates its amino group) • The 2 most important aminotransferases 1 - alanine aminotransferase (ALT) 2 - aspartate aminotranspeptidase (AST)

ALanine Amino. Transferase (ALT) & ASpartate Amino. Transferase (AST) amino acid Carbon skeleton of alanine UREA CYCLE

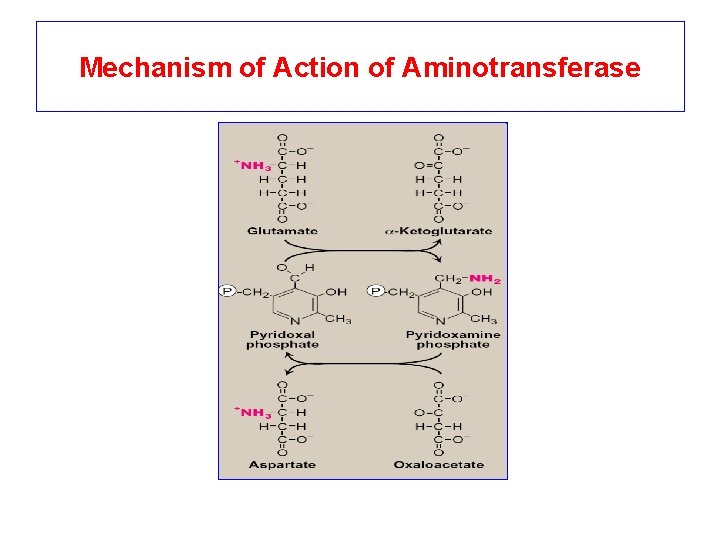
Mechanism of Action of Aminotransferase

Equlibrium of Transamination Reactions Equilibrium constant : ~ 1 Allowing the reaction to function in both directions after protein-rich meal amino acid degradation (removal of amino group from amino acid a-keto acid) supply of amino acids from diet is not adequate amino acid biosynthesis (addition of amino group to a-keto acid amino acid)

Diagnostic Value of Plasma Aminotransferases • Aminotransferases are normally intracellular enzymes • Plasma contains low levels of aminotransferases representing release of cellular contents during normal cell turnover • Elevated plasma levels of aminotransferases indicate damage to cells rich in these enzymes (as physical trauma or disease to tissue) • Plasma AST & ALT are of particular diagnostic value

Diagnostic Value of Plasma Aminotransferases 1 - liver disease: Plasma ALT & AST are elevated in nearly all liver diseases but, particularly high in conditions that cause cell necrosis as: viral hepatitis toxic injury prolonged circulatory collapse ALT is more specific for liver disease than AST is more sensitive (as liver contains a large amount of AST) 2 - Nonhepatic disease: as: myocardial infarction muscle disorders These disorders can be distinguished clinically from liver disease

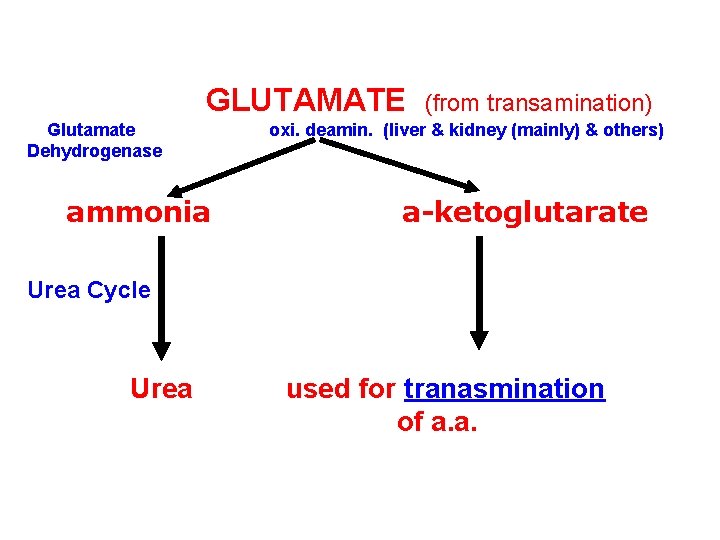
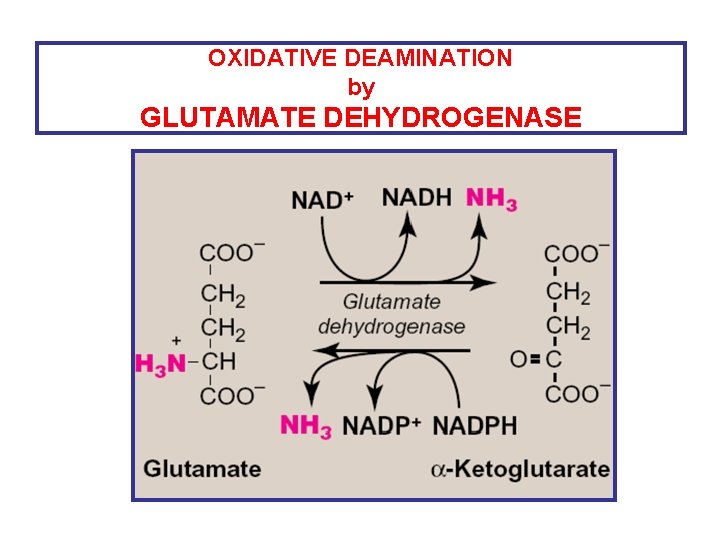
2 - Oxidative deamination of amino acids By: Glutamate Dehydrogenase • Oxidative Deamination of glutamate by glutamate dehydrogenase results in liberation of the amino group as free ammonia (i. e. no transfer of amino group) • primarily in the liver & kidney (but can occur in other cells of the body) • Oxi Deamin. Of Glutamate provides: 1 - a-ketoglutarate (can be reused for transamination of a. a. ) 2 - free ammonia urea cycle urea

GLUTAMATE Glutamate Dehydrogenase ammonia (from transamination) oxi. deamin. (liver & kidney (mainly) & others) a-ketoglutarate Urea Cycle Urea used for tranasmination of a. a.

OXIDATIVE DEAMINATION by GLUTAMATE DEHYDROGENASE

Glutamate Dehydrogenase • Amino group of most amino acids are funneled to GLUTAMATE (by transamination with a-ketoglutarate) • GLUTAMATE is the only amino acid that undergoes oxidative deamination (by glutamate dehydrogenase) to give a-ketoglutarate & ammonia • Amino Acids donate their amino group to a-ketoglutarate to produce glutamate (Transamination) • Glutamate is oxi deamin. to a-ketoglutarate & ammonia (by Glutamate dehydrogenase)

Amino acid TRANS a-ketoglutarate + AMINASE ammonia GLUTA MATE UREA CYCLE DEHYDROGENASE a-keto acid + Glutamate (carbon Skeleton) metabolized UREA Energy (catab. ) other compounds

Removal of Amino Group from an Amino Acid UREA CYCLE Amino acid Carbon skelton of an amino acid Coenzyme for glutamate dehydrogenase: NAD+

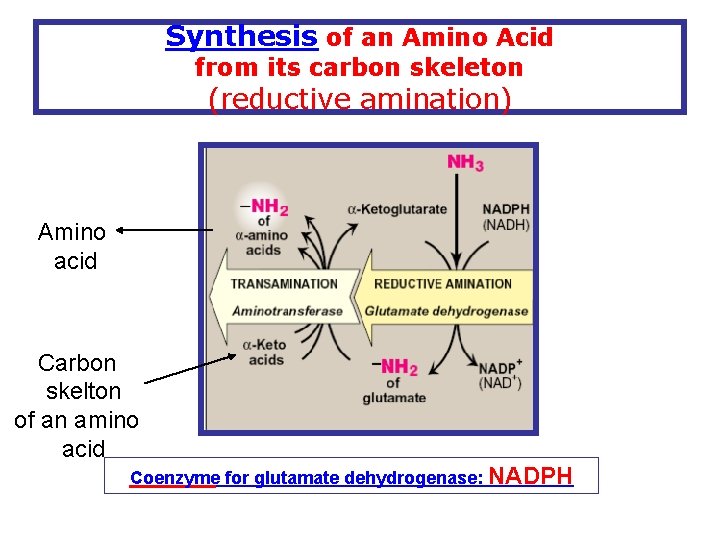
Synthesis of an Amino Acid from its carbon skeleton (reductive amination) Amino acid Carbon skelton of an amino acid Coenzyme for glutamate dehydrogenase: NADPH

Combined Actions of Transaminases & Glutamate Dehydrogense reactions

Direction of Reactions • After protein ingestion Glutamate level in liver is elevated Reactions proceeds in direction of amino acid degradation & formation of ammonia

Allosteric Regulation of Glutamate Dehydrogenase • ATP & GTP are allosteric inhibitors of glutamate dehydrogenase • ADP & GDP are allosteric activators of glutamate dehydrogenase Energy in cells glutamate degradation by glutamate dehydrogenase Energy production from the carbon skeleton of amino acids

D-amino acid oxidase • D-amino acids are found in plants & cell walls of microorganisms • Not used in synthesis of mammalian proteins • D-amino acids is available in diet (from plants) • Metabolized by D-amino acid oxidase (in liver) FAD-dependent Oxidative Deamination of D-amino acids a-keto acids Energy Reaminated ammonia L-amino acids UREA

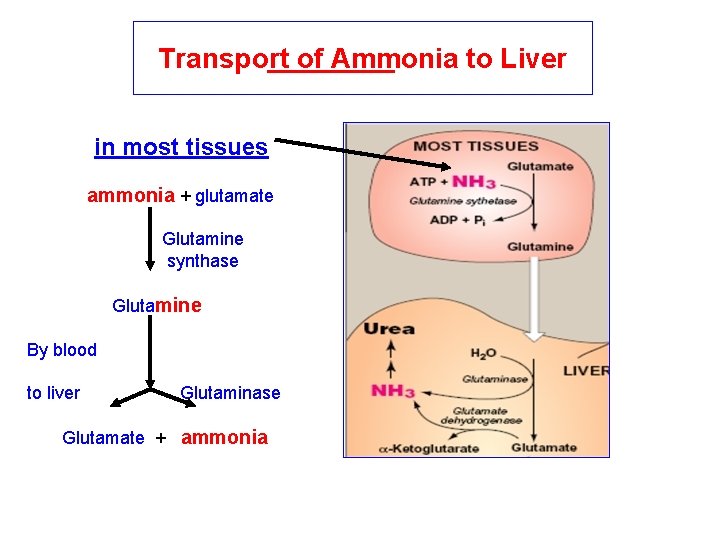
Transport of Ammonia to Liver in most tissues ammonia + glutamate Glutamine synthase Glutamine By blood to liver Glutaminase Glutamate + ammonia

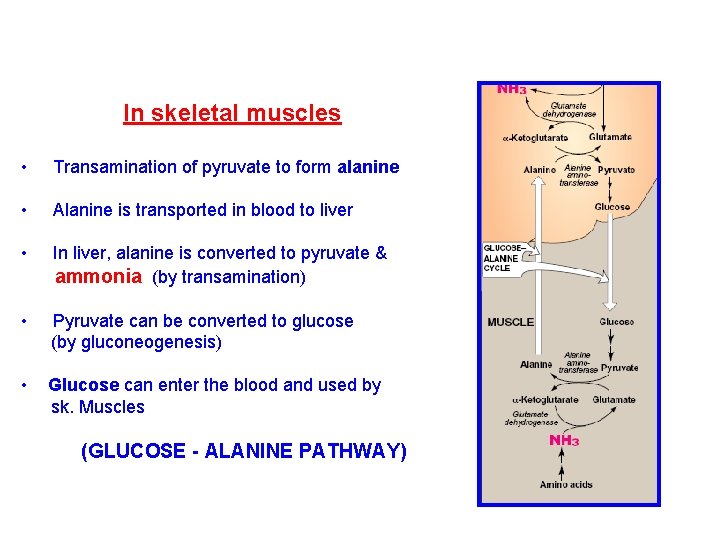
In skeletal muscles • Transamination of pyruvate to form alanine • Alanine is transported in blood to liver • In liver, alanine is converted to pyruvate & ammonia (by transamination) • Pyruvate can be converted to glucose (by gluconeogenesis) • Glucose can enter the blood and used by sk. Muscles (GLUCOSE - ALANINE PATHWAY)