6 Free Electron Fermi Gas Energy Levels in





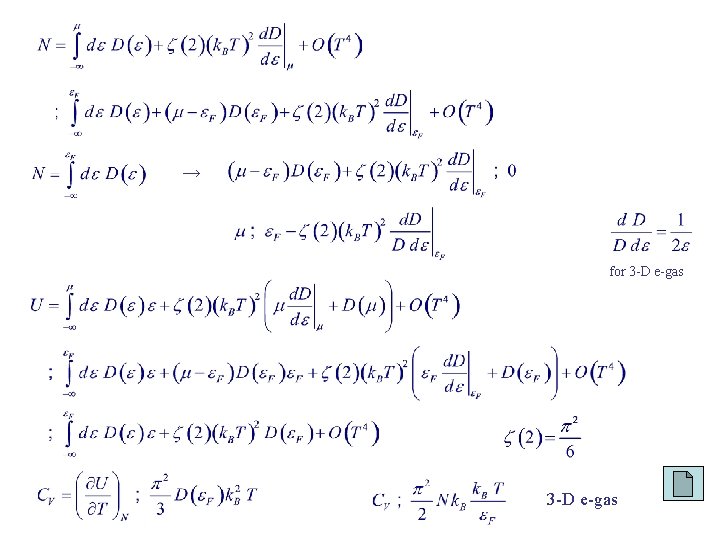





- Slides: 24

6. Free Electron Fermi Gas • • Energy Levels in One Dimension Effect of Temperature on the Fermi-Dirac Distribution Free Electron Gas in Three Dimensions Heat Capacity of the Electron Gas Electrical Conductivity and Ohm’s Law Motion in Magnetic Fields Thermal Conductivity of Metals Nanostructures

Introduction Free electron model: Works best for alkali metals (Group I: Li, Na, K, Cs, Rb) Na: ionic radius ~. 98 A, n. n. dist ~ 1. 83 A. Successes of classical model: Ohm’s law. σ/κ Failures of classical model: Heat capacity. Magnetic susceptibility. Mean free path. Quantum model ~ Drude model

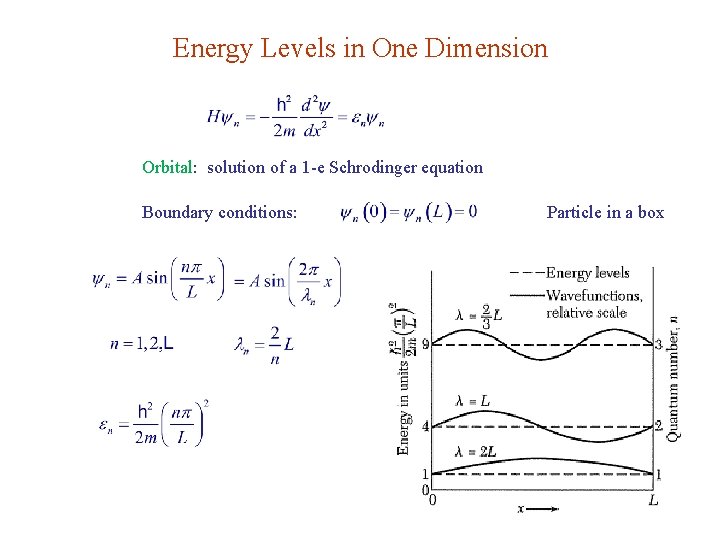
Energy Levels in One Dimension Orbital: solution of a 1 -e Schrodinger equation Boundary conditions: Particle in a box

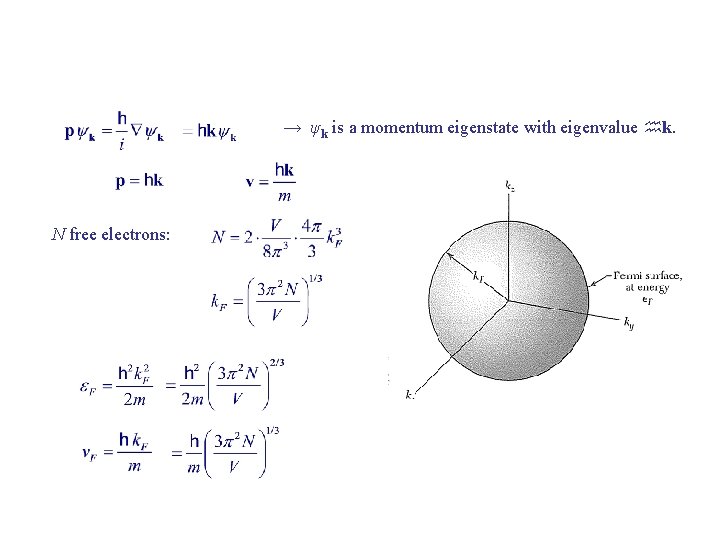
Pauli-exclusion principle: No two electrons can occupy the same quantum state. Quantum numbers for free electrons: (n, ms ) Degeneracy: number of orbitals having the same energy. Fermi energy εF = energy of topmost filled orbital when system is in ground state. N free electrons:

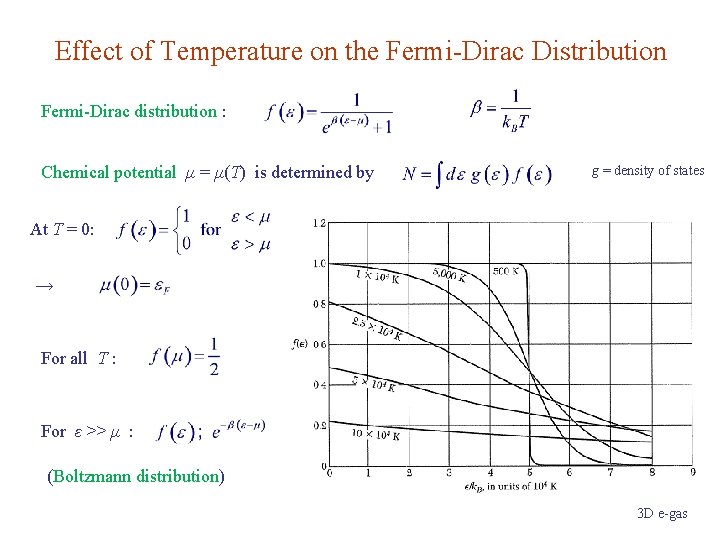
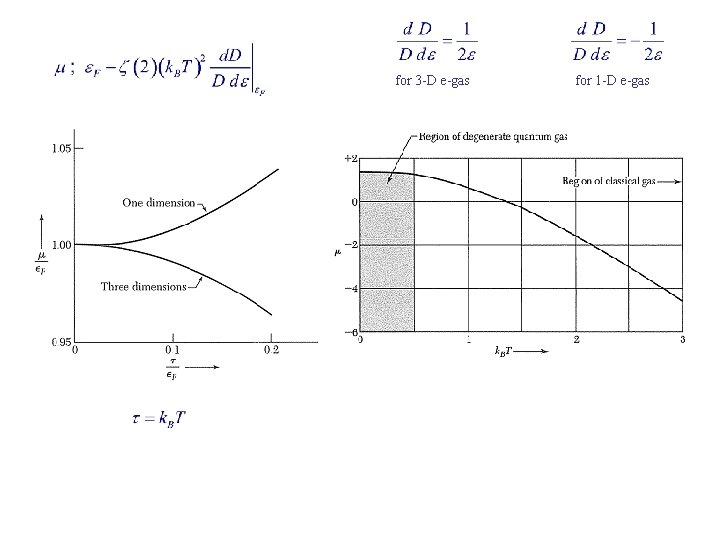
Effect of Temperature on the Fermi-Dirac Distribution Fermi-Dirac distribution : Chemical potential μ = μ(T) is determined by g = density of states At T = 0: → For all T : For ε >> μ : (Boltzmann distribution) 3 D e-gas

Free Electron Gas in Three Dimensions Particle in a box (fixed) boundary conditions: → Standing waves Periodic boundary conditions: → Traveling waves

→ ψk is a momentum eigenstate with eigenvalue k. N free electrons:


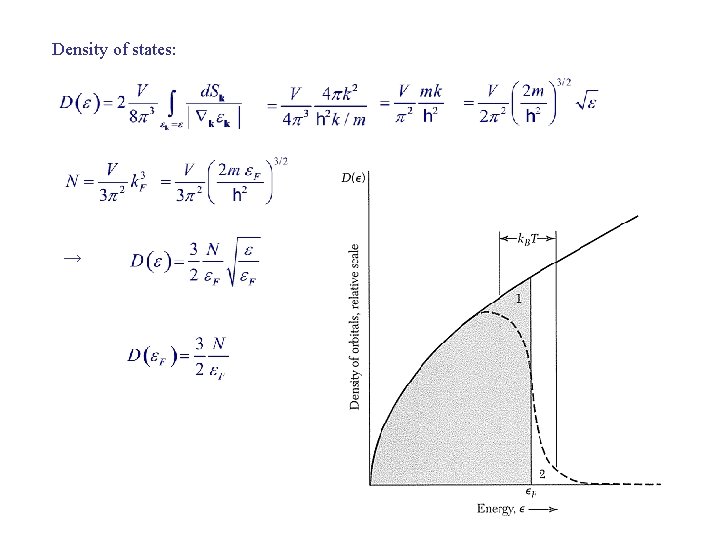
Density of states: →

Heat Capacity of the Electron Gas (Classical) partition theorem: kinetic energy per particle = (3/2) k. BT. N free electrons: ( 2 orders of magnitude too large at room temp) Pauli exclusion principle → Using the Sommerfeld expansion formula TF ~ 104 K for metal free electrons
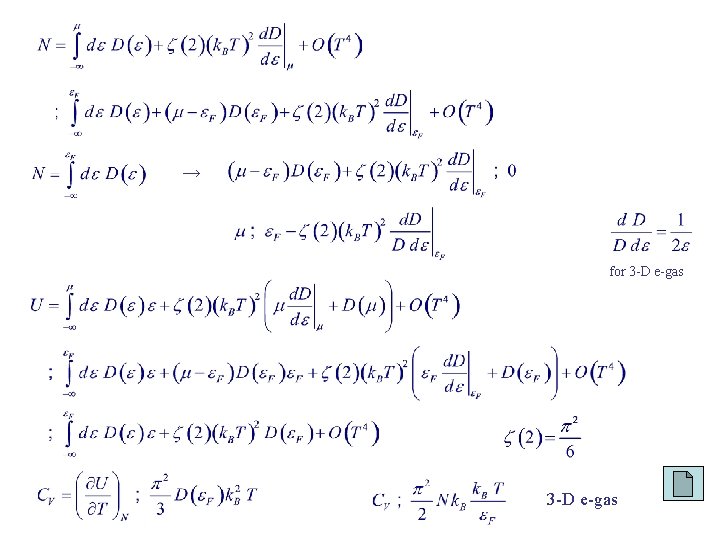
→ for 3 -D e-gas

for 3 -D e-gas for 1 -D e-gas

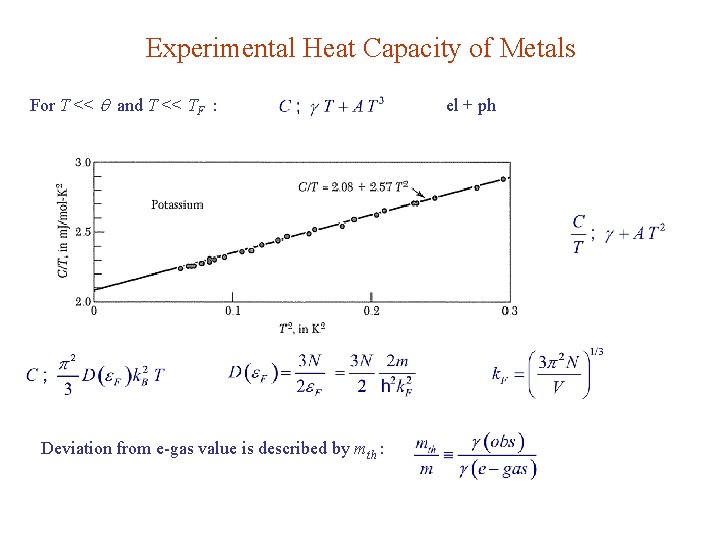
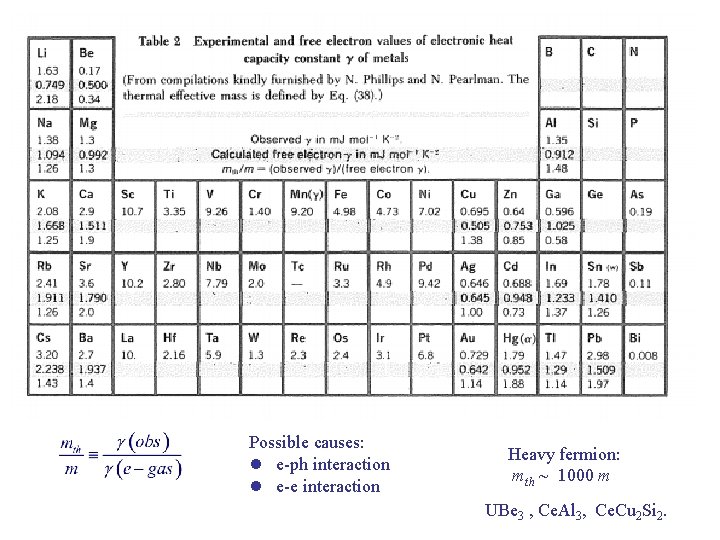
Experimental Heat Capacity of Metals For T << and T << TF : Deviation from e-gas value is described by mth : el + ph

Possible causes: l e-ph interaction l e-e interaction Heavy fermion: mth ~ 1000 m UBe 3 , Ce. Al 3, Ce. Cu 2 Si 2.

Electrical Conductivity and Ohm’s Law Free particle in constant E field Heisenberg picture: Lorentz force on free electron: No collision: Collision time : Ohm’s law

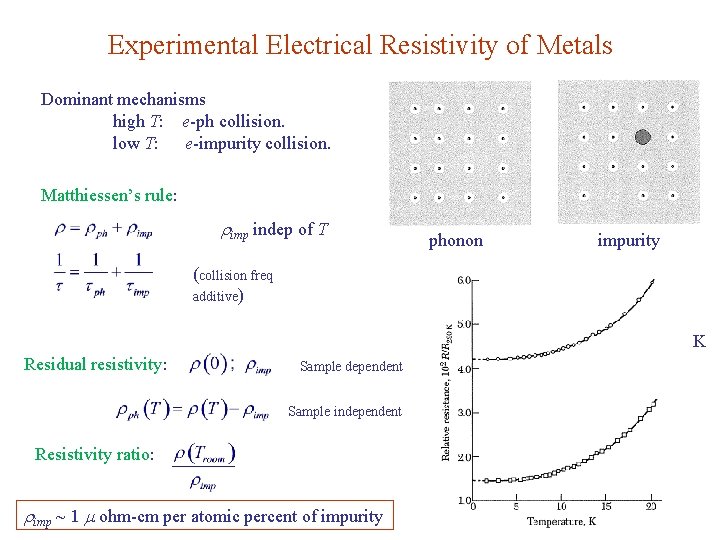
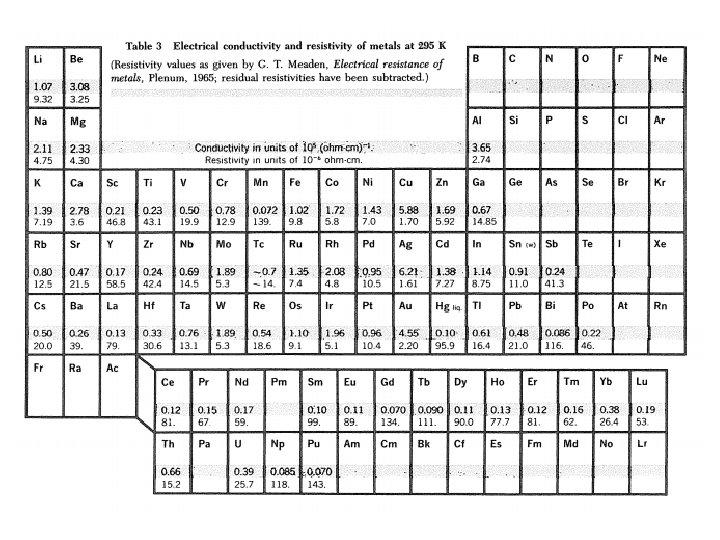
Experimental Electrical Resistivity of Metals Dominant mechanisms high T: e-ph collision. low T: e-impurity collision. Matthiessen’s rule: imp indep of T phonon impurity (collision freq additive) K Residual resistivity: Sample dependent Sample independent Resistivity ratio: imp ~ 1 ohm-cm per atomic percent of impurity

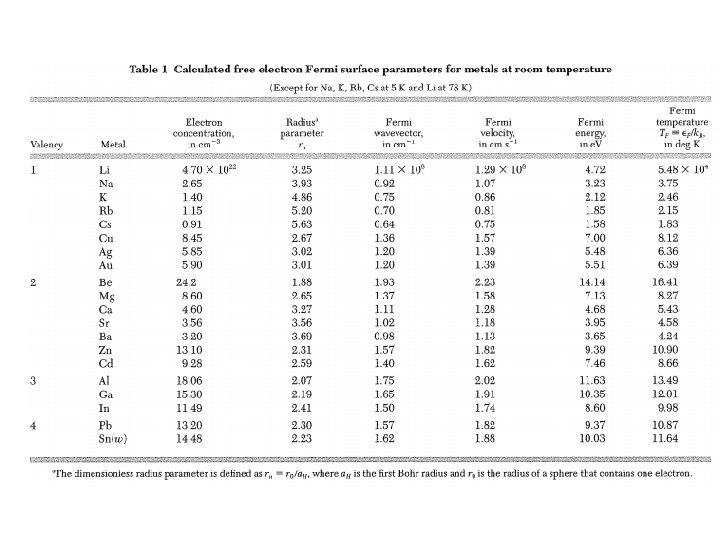
Consider Cu with resistivity ratio of 1000: From Table 3, we have imp ~ 1 ohm-cm per atomic percent of impurity Impurity concentration: = 17 ppm Very pure Cu sample: For T > : See App. J


Umklapp Scattering Normal: Umklapp : Large scattering angle ( ~ ) possible For Fermi sphere completely inside BZ, U-processes are possible only for q > q 0 = 0. 267 k. F for 1 e /atom Fermi sphere inside a bcc BZ. Number of phonon available for U-process exp( U /T ) For K, U = 23 K, = 91 K U-process negligible for T < 2 K

Motion in Magnetic Fields Equation of motion with relaxation time : q = –e for electrons Let be a right-handed orthogonal basis Steady state: = cyclotron frequency

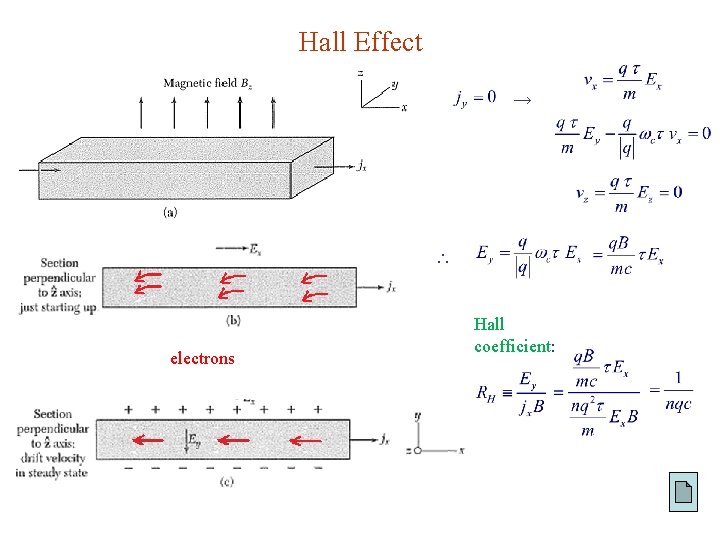
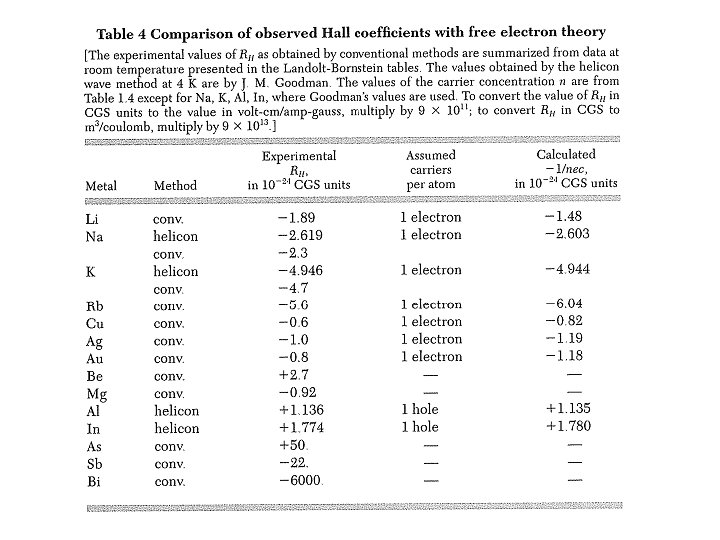
Hall Effect → electrons Hall coefficient:


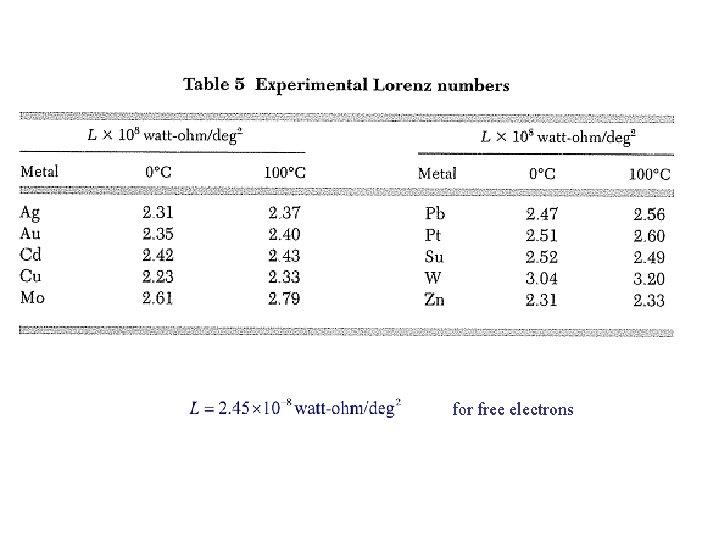
Thermal Conductivity of Metals From Chap 5: Fermi gas: In pure metal, Kel >> Kph for all T. Wiedemann-Franz Law: Lorenz number: for free electrons

for free electrons