FREE ELECTRON THEORY ARC TOPICS TO BE COVERED

FREE ELECTRON THEORY ARC

TOPICS TO BE COVERED Classical free electron theory and its limitations Deduction of Ohm’s law, Statement of Weidemann-Franz law Quantum theory of free electrons, Fermi level Density of states, Fermi-Dirac distribution Thermionic emission, Richardson equation

Chief Characteristics of Metals • Metal possesses high electrical and thermal conductivity • Metals obey Ohm’s law • Conductivity of metals decreases with rise of temperature • Metals obey Wiedemann-Franz law • Near absolute zero, certain metals exhibit superconductivity

Classical Free Electron Theory • Initially stated by Drude in 1900 v He was working prior to the development of quantum mechanics, so he began with a classical model. (1863 -1906) CONCEPT • In Drude model, the valence electrons from each atom become detached and wander freely through the metal, while the metallic ions remain intact and play the role of the immobile positive particles. • Each electron behaves as a perfect gas molecule • Each electron is free to move through the entire volume of the metal • System of free electrons in a metal = free electron gas
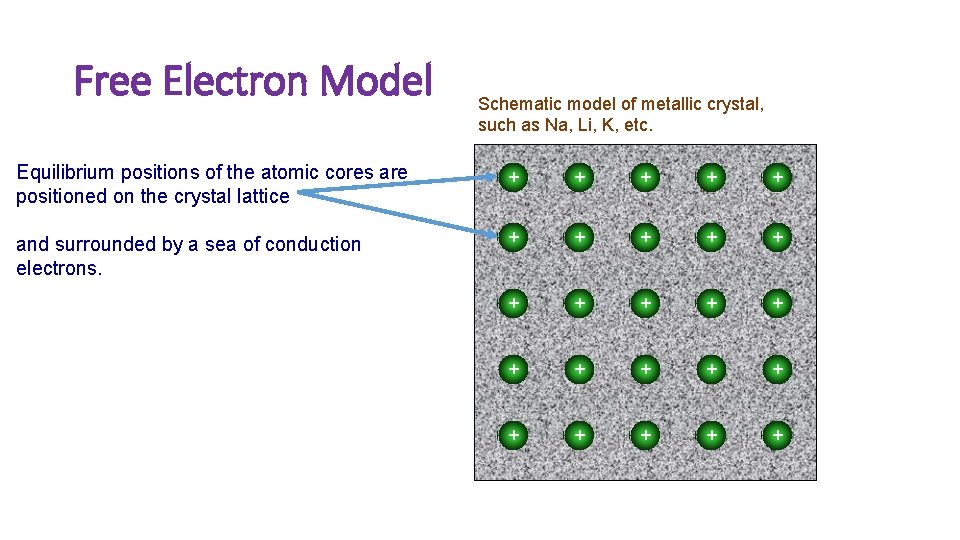
Free Electron Model Schematic model of metallic crystal, such as Na, Li, K, etc. Equilibrium positions of the atomic cores are positioned on the crystal lattice + + + and surrounded by a sea of conduction electrons. + + + + +

Drude’s Assumptions 1. Matter consists of light negatively charged electrons which are mobile & heavy, positively charged ions. 2. The only interactions are electron-ion collisions, which take place in a very short time t. • The neglect of the electron-electron interactions is THE INDEPENDENT ELECTRON APPROXIMATION. • The neglect of the electron-ion interactions is THE FREE ELECTRON APPROXIMATION 3. Electron-ion collisions are assumed to dominate. These will abruptly alter the electron velocity & maintain thermal equilibrium. 4. The mean time between collisions is �� 5. Electrons emerge from each collision with their velocity changed. • Till the application of an external electric field, the electrons move about in a random manner
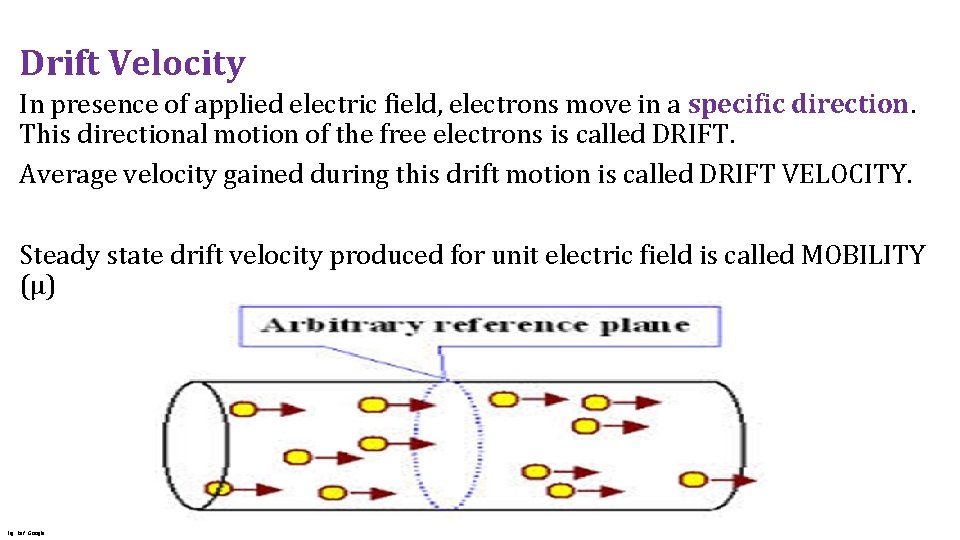
Drift Velocity In presence of applied electric field, electrons move in a specific direction. This directional motion of the free electrons is called DRIFT. Average velocity gained during this drift motion is called DRIFT VELOCITY. Steady state drift velocity produced for unit electric field is called MOBILITY (μ) Fig. Ref. Google
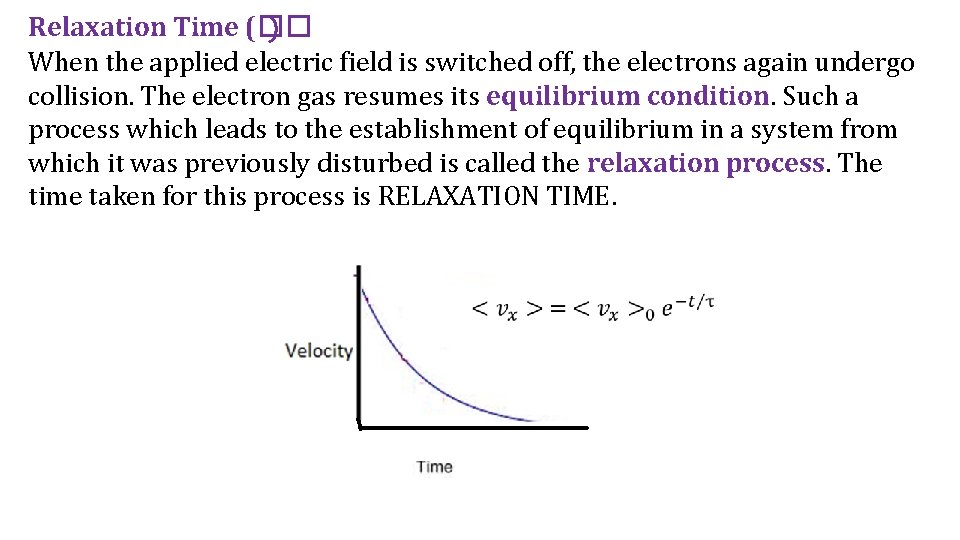
Relaxation Time (�� ) When the applied electric field is switched off, the electrons again undergo collision. The electron gas resumes its equilibrium condition. Such a process which leads to the establishment of equilibrium in a system from which it was previously disturbed is called the relaxation process. The time taken for this process is RELAXATION TIME.


Drift Velocity Expression Integrating, we get If is the average time between collisions then the average drift velocity is 10

Ohm’s Law • Basic law concerning the flow of electricity. • Ohm's law states that the current through a conductor between two points is directly proportional to the potential difference across the two points. • Constant of proportionality, resistance, is introduced • In mathematical terms, V = I x R where V is voltage, I is current, and R is resistance • When an electric field, E is applied to a conductor, an electric current begins to flow, and the current density by Ohm’s law is • Materials that obey Ohm’s law are said to be ohmic
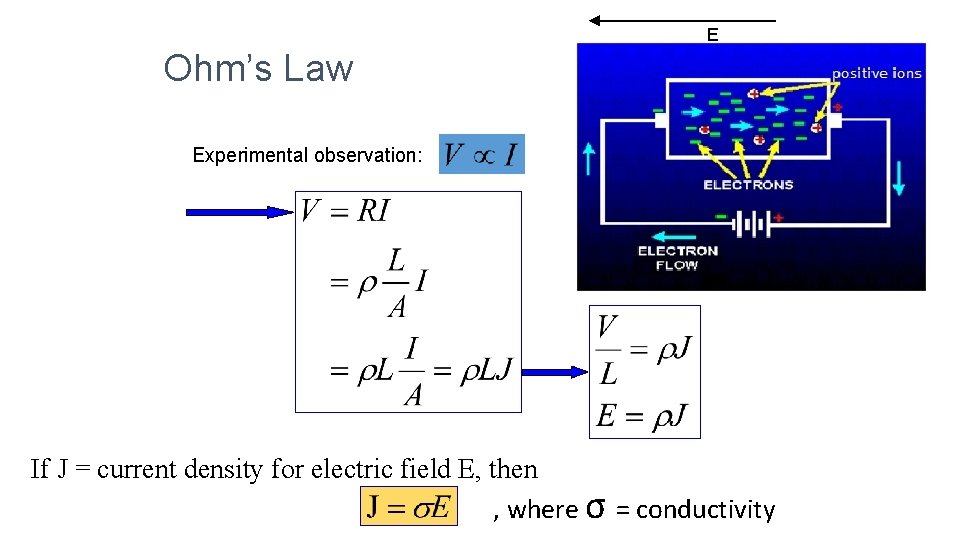
E Ohm’s Law Experimental observation: If J = current density for electric field E, then , where σ = conductivity
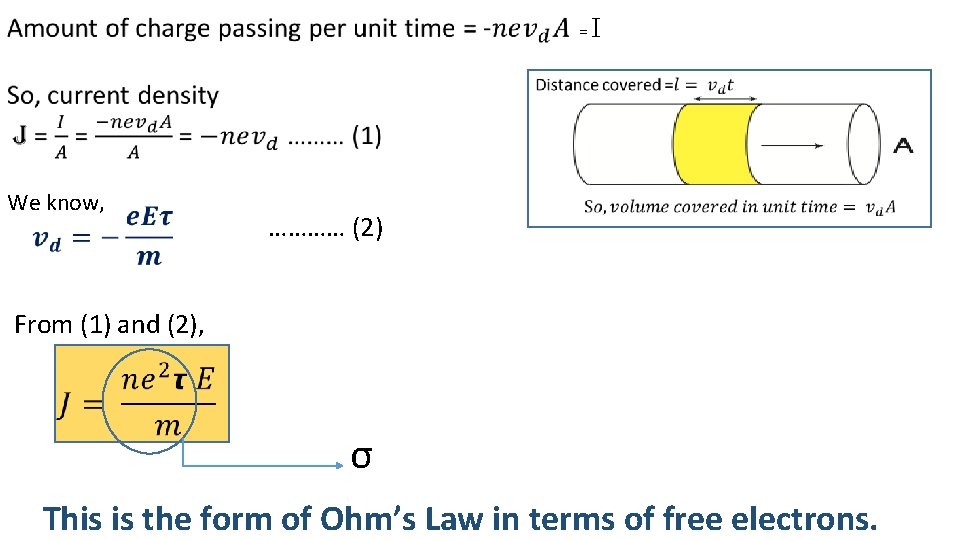
= I We know, ………… (2) From (1) and (2), σ This is the form of Ohm’s Law in terms of free electrons.

Wiedemann-Franz Law Wiedemann and Franz law states that the ratio of thermal and electrical conductivity of all metals is constant at a given temperature (for room temperature and above). Thermal conductivity Electrical Conductivity Later it was found by L. Lorenz that this constant is proportional to the absolute temperature L = Lorentz Number

Drawbacks of Classical Free Electron Theory

Drawbacks (continued. . ) • Wiedemann- Franz law: At low temperatures, K/σT is not a constant. But in classical free electron theory, it is a constant at all temperatures. • Paramagnetism of Metals: Theoretical value of paramagnetic susceptibility is greater than the experimental value. Experimental fact that paramagnetism of metals is nearly independent of temperature could not be explained

Salient features of Quantum Free Electron Theory Proposed by Sommerfeld in 1928 • Electrons obey the laws of quantum mechanics • Energy levels of electrons are quantized • Electrons occupy energy orbitals according to Pauli’s exclusion principle • Distribution of electrons in different energy levels are according to Fermi. Dirac statistics • Retained concept of free electrons moving in a uniform potential but prevented them from escaping the crystal by very high potential barriers at the surfaces

FERMI LEVEL : Highest energy level occupied by electrons at Absolute zero. All the energy states upto Fermi level are OCCUPIED and all energy levels above Fermi level are VACANT. FERMI ENERGY: Energy corresponding to Fermi Level. Constant for a particular system Probability of an electron occupying a particular energy level ‘E’ is given by Fermi-Dirac Distribution At T = 0 K and for E < E , f(E) = 1 F for E > E , f(E) = 0 F On increasing the temperature, electrons get excited to higher energy level. Distribution of electrons in different energy levels gets determined by Fermi-Dirac Distribution function. For lower energies, f tends to 1. For higher energies, f tends to 0.
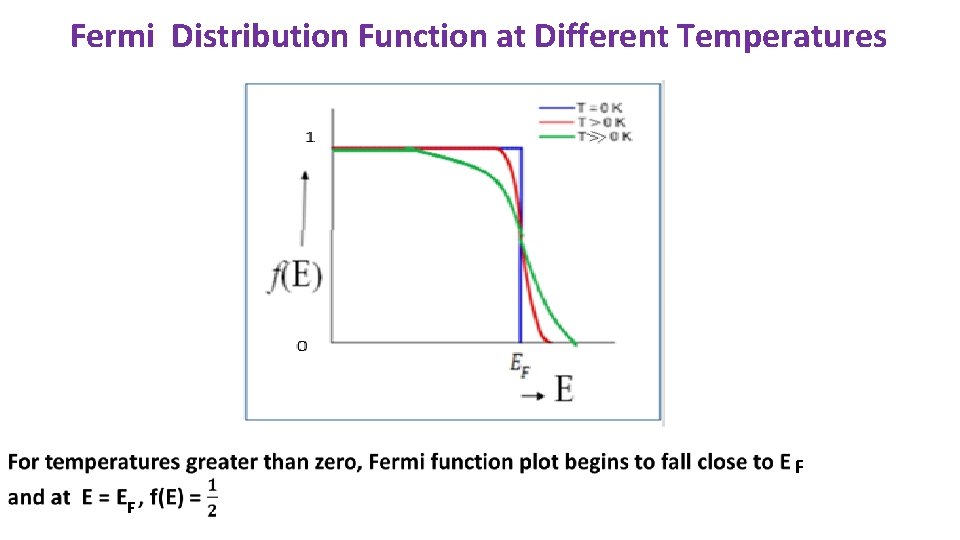
Fermi Distribution Function at Different Temperatures F F

FERMI VELOCITY = velocity associated with Fermi Energy i. e. velocity of electrons occupying Fermi Level FOR SODIUM, FERMI VELOCITY FERMI TEMPERATURE = Temperature associated with Fermi energy Ref: Eisberg, R. and Resnick, R. Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles, 2 nd ed. New York: Wiley, 1985. FERMI TEMPERATURE

DENSITY OF ENERGY STATES CONCEPT OF DENSITY OF ENERGY STATES simplifies OUR CALCULATION. It helps in finding the number of energy states with a specific energy. Density of states (DOS) of a system describes the number of available states in a unit volume per unit energy range. In a system, if N(E) = number of electrons with energy E, g(E) = number of energy states with energy E, f(E) = probability of an electron to occupy energy state E, then N(E) d. E = g(E)d. E f(E)

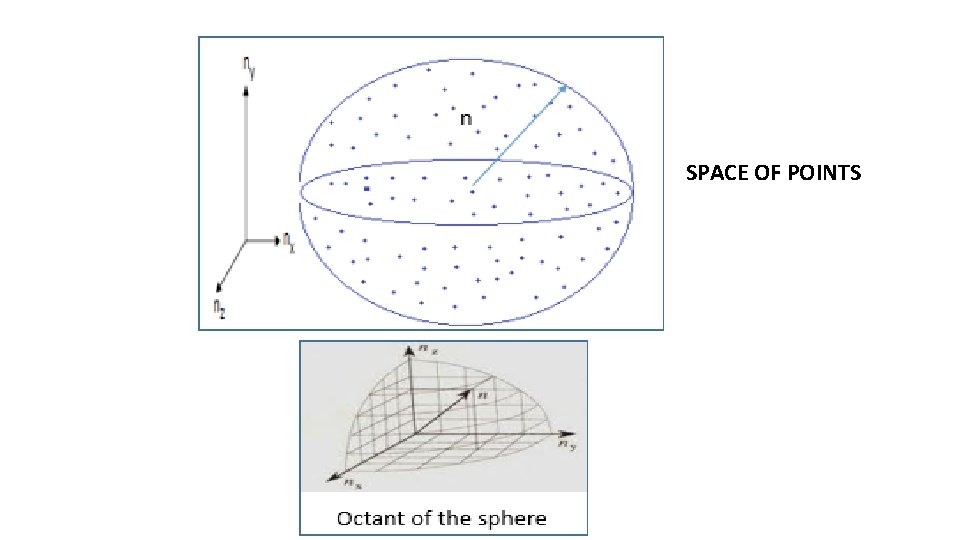
SPACE OF POINTS

If n = radius of sphere whose octant encloses all the points upto an energy ‘E’, then Number of allowed energy values upto an energy E = number of points in the octant of sphere of radius ‘n’ We consider another sphere of radius n+dn whose octant encloses all points upto an energy ‘E+d. E’, then Number of allowed energy values upto an energy E + d. E = number of points in the octant of sphere of radius ‘ n + dn’ So, number of allowed energy states in energy range d. E = number of points in the space between two octant shells of radii n and n+dn = (volume of space between two octant shells of radii n and n+dn) X (number of points / unit volume) -------- (3)
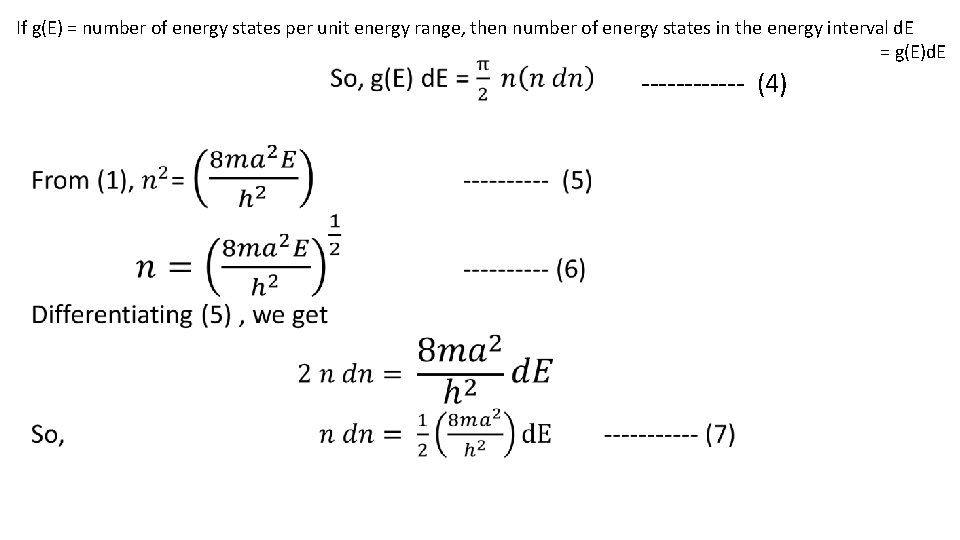
If g(E) = number of energy states per unit energy range, then number of energy states in the energy interval d. E = g(E)d. E ------ (4)

Using (4), (6) and (7), we get ----- (8) Hence, the number of energy states present in unit volume having energy values lying between E and E + d. E (DOS) is given by

Density of energy states for a free electron gas TASK: Find out the number of electrons present per unit volume of a cubical metal block at absolute zero temperature

Thermionic Emission The emission of electrons from a metal under the effect of thermal energy is called THERMIONIC EMISSION. Emitted electrons are called THERMIONS. Electrons are free to move inside the metal Electrons cannot come out of the metal surface on its own as high potential barrier is present at the surface but when the temperature of the metal is sufficiently high, electrons gain sufficient energy to overcome the barrier and ESCAPE from the metal surface Free electron theory assumes that the potential within the metal is constant. The minimum energy to be supplied to the electron for its emission from the metal is termed as WORK FUNCTION (Ф) of the metal

Richardson’s Equation If W = minimum energy of the electron for its emission from the surface, E = Fermi energy of the metal, F then, Ф = W – E = work function of the metal. F No. of energy states / unit volume in energy range E to E + d. E, So, density of energy states per unit volume in momentum range p to p+dp, Volume element in momentum space,

From (1) and (2), the density of states in momentum space,

Standard Integral Form So, current density,
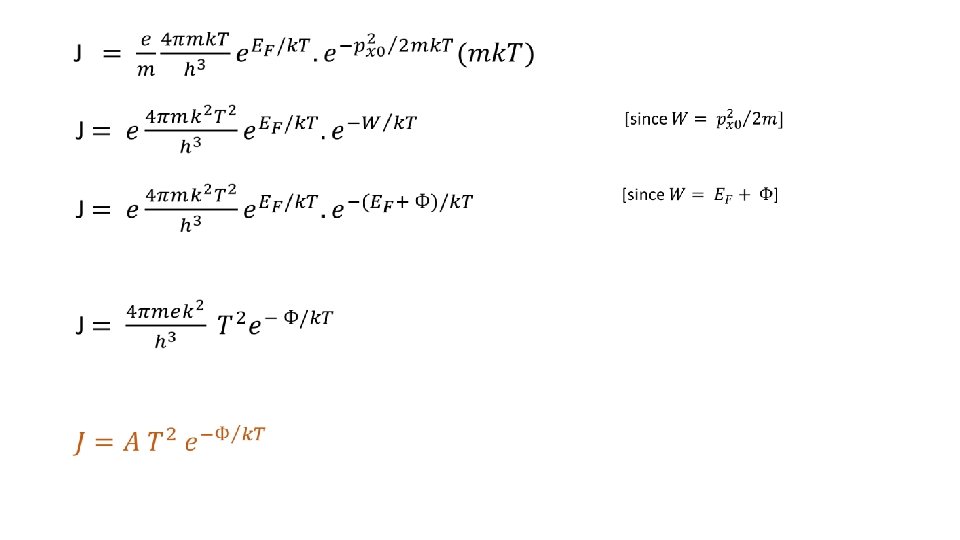
- Slides: 32