SIGNIFICANCE OF QUANTUM MECHANICS MOST IMPORTANT SIGNIFICANCE Quantum



















- Slides: 24

SIGNIFICANCE OF QUANTUM MECHANICS

MOST IMPORTANT SIGNIFICANCE • Quantum Mechanics was able to describe the wave particle duality. • 1. Blackbody Radiation • 2. Photoelectric Effect • 3. Compton Effect • 4. de Broglie Equation • It also revolutionized the way we understand Chemistry.

HEISENBERG UNCERTAINTY PRINCIPLE IN CHEMISTRY • One question to examine is: “Was Neil’s Bohr right about his model, does it actually work? ” • Let’s examine this idea using the hydrogen atom: • r = 5. 3 x 10 -11 m or d = 1. 06 x 10 -10 • v (of electron) = 2. 2 x 106 m/s • m (of electron) = 9. 11 x 10 -31 kg • We know that the uncertainty of the velocity of the electron is about 10% (i. e. we are almost certain of the velocity of the electron) • Uncertainty of velocity =. 1

HEISENBERG UNCERTAINTY PRINCIPLE IN CHEMISTRY •

HEISENBERG UNCERTAINTY PRINCIPLE IN CHEMISTRY •

HEISENBERG UNCERTAINTY PRINCIPLE IN CHEMISTRY • WOWZA, Bohr’s model has some errors. The distance that an electron travels around the nucleus is larger than 1. 6 x 10 -10 m, it is actually 2. 631 x 10 -10 m. • So what does this mean? • Well thanks to Heisenberg we know that the electron is located somewhere around the atom and has a diameter of 2. 631 x 10 -10 m.

COMBINE BOHR AND HEISENBERG • This is what the 1 s orbital looks like in an atom. • The dots represent all the possible places where the electron can be. • Note that this shell can only hold two electrons.

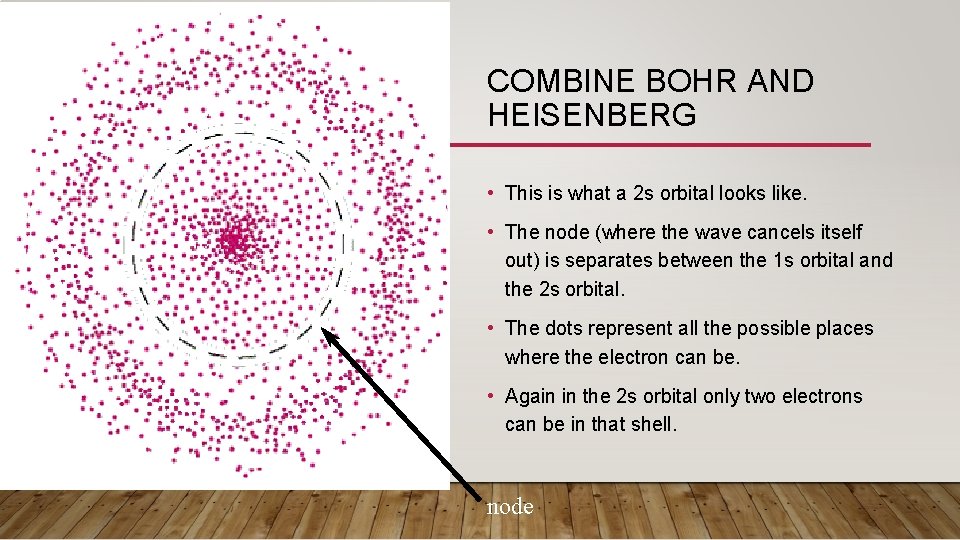
COMBINE BOHR AND HEISENBERG • This is what a 2 s orbital looks like. • The node (where the wave cancels itself out) is separates between the 1 s orbital and the 2 s orbital. • The dots represent all the possible places where the electron can be. • Again in the 2 s orbital only two electrons can be in that shell. node

QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • Through a series of complex calculations, physicists and chemists where able to create four specific numbers of importance that help us understand how the electrons move in an atom. • 1. Principle Quantum Number (n) – size and energy level of the orbit. • 1, 2, 3, 4, 5, 6, 7 • Each number identifies the row on the periodic table.

QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • 2. Angular Momentum Number (l) – the shape of the orbit. • The numerical numbers in the equations are 0, 1, 2, and 3. Each number represents the s, p, d, and f orbital shells. • It is the sublevel of the orbits. • subshell no. • shell notation s 0 1 2 p d f 3

QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • 3. Magnetic Quantum Number (ml) – defines the three dimensional shape of the orbits. • If l = 0 (s shell), then ml = 0 each ml value has room for two electrons • If l = 1 (p shell), the ml = -1, 0, 1 each ml value has room for two electrons • If l = 2 (d shell), then ml = -2, -1, 0, 1, 2 each ml value has room for two electrons • This is rather remarkable, how many electrons can fit in the s shell? Two, and there is one spot for two electrons. • How many electrons can fit in the p shell? Six, and there are three spots for six electrons.

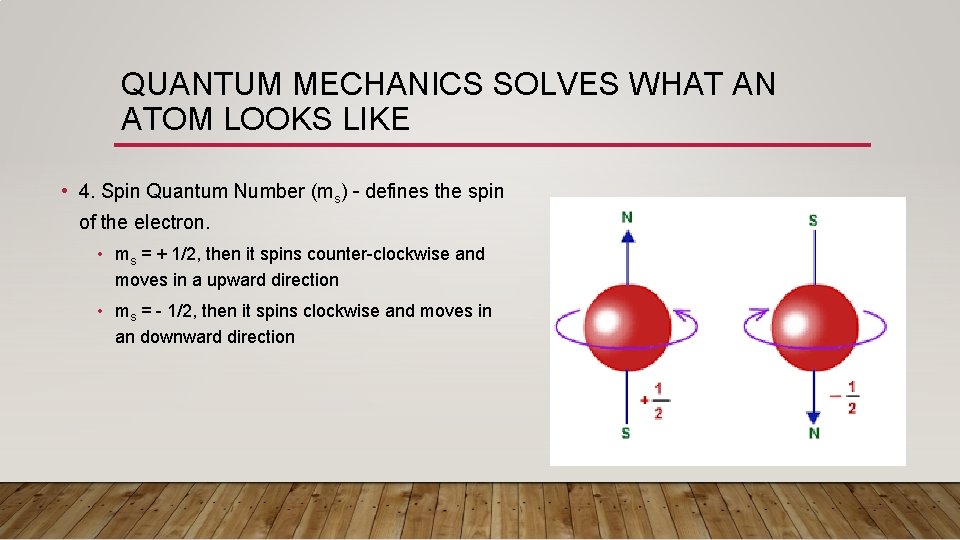
QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • 4. Spin Quantum Number (ms) – defines the spin of the electron. • ms = + 1/2, then it spins counter-clockwise and moves in a upward direction • ms = - 1/2, then it spins clockwise and moves in an downward direction

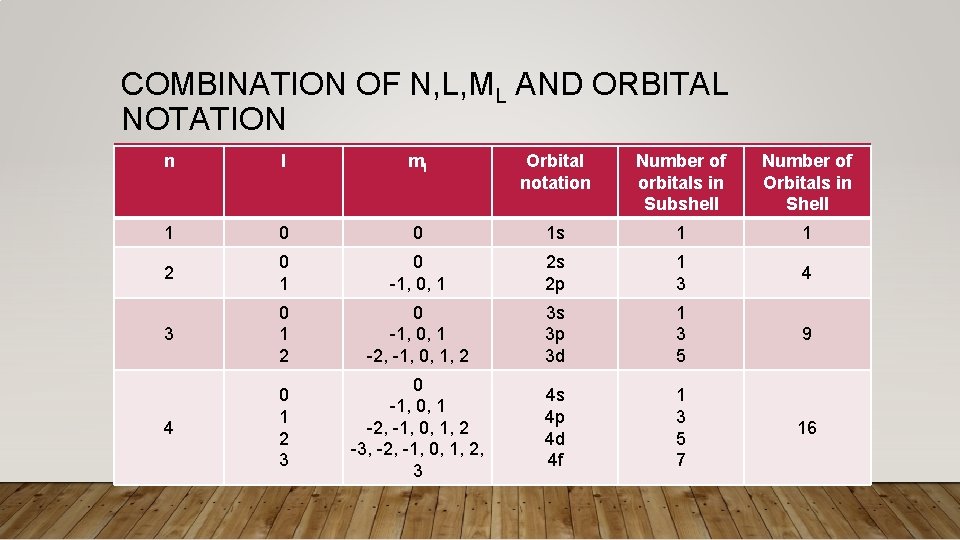
COMBINATION OF N, L, ML AND ORBITAL NOTATION n l ml Orbital notation Number of orbitals in Subshell Number of Orbitals in Shell 1 0 0 1 s 1 1 2 0 1 0 -1, 0, 1 2 s 2 p 1 3 4 3 0 1 2 0 -1, 0, 1 -2, -1, 0, 1, 2 3 s 3 p 3 d 1 3 5 9 4 0 1 2 3 0 -1, 0, 1 -2, -1, 0, 1, 2 -3, -2, -1, 0, 1, 2, 3 4 s 4 p 4 d 4 f 1 3 5 7 16

HOW TO FILL THE SHELLS • Note that 3 d starts with Scantium, 4 d starts with Yitrium, 5 d starts with Lanthanium, and 6 d starts with Actinium. • Note that 4 f starts with Cerium and 5 f starts with Thorium.

FINDING THE ELEMENT USING QUANTUM NUMBERS • First find the row number you are in - 1, 2, 3, 4…. • Secondly find the subshell you are in - 0(s), 1(p), 2(d), 3(f) • Thirdly find the subshell location you are in by drawing out how many subshells there are in the shell orbit you are in. Example for subshell p draw three lines and number them -1, 0, 1. For subshell d number them -2, -1, 0, 1, 2. • Finally, fill the electron subshells with electron until you get to the proper +1/2 or 1/2 spin.

FEW EXAMPLES • If n = 2, l = 0, ml = 0 and ms = -1/2 what element is it? • Berylium • If n = 3, l = 2, ml = -1 and ms = -1/2 what element is it? • Cobalt • If n = 2, l = 1, ml = 1 and ms = -1/2 what element is it? • Neon

MORE EXAMPLES • If n = 4, l = 2, ml = 2 and ms = 1/2 what element is it? • Technellium • If n = 4, l = 1, ml = -1 and ms = 1/2 what element is it? • Gallium

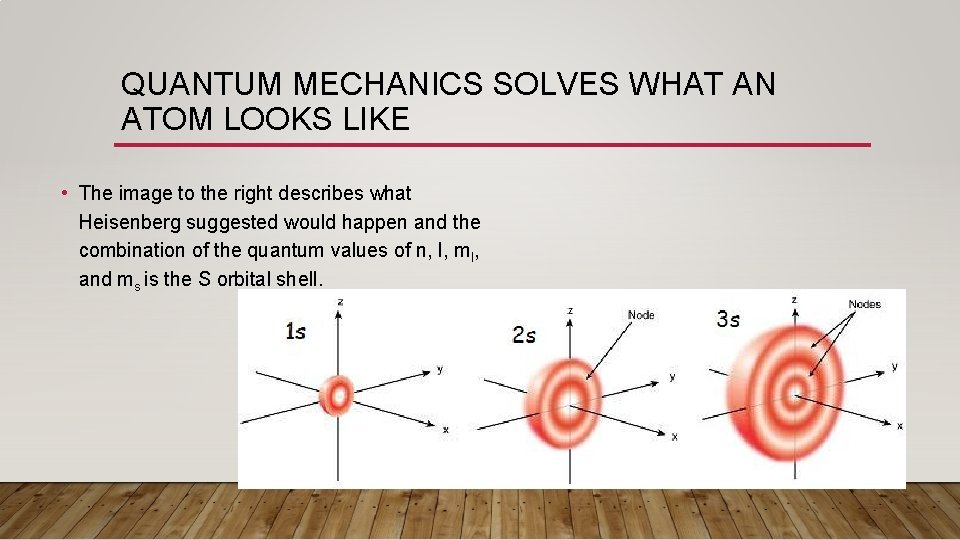
QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • The image to the right describes what Heisenberg suggested would happen and the combination of the quantum values of n, l, ml, and ms is the S orbital shell.

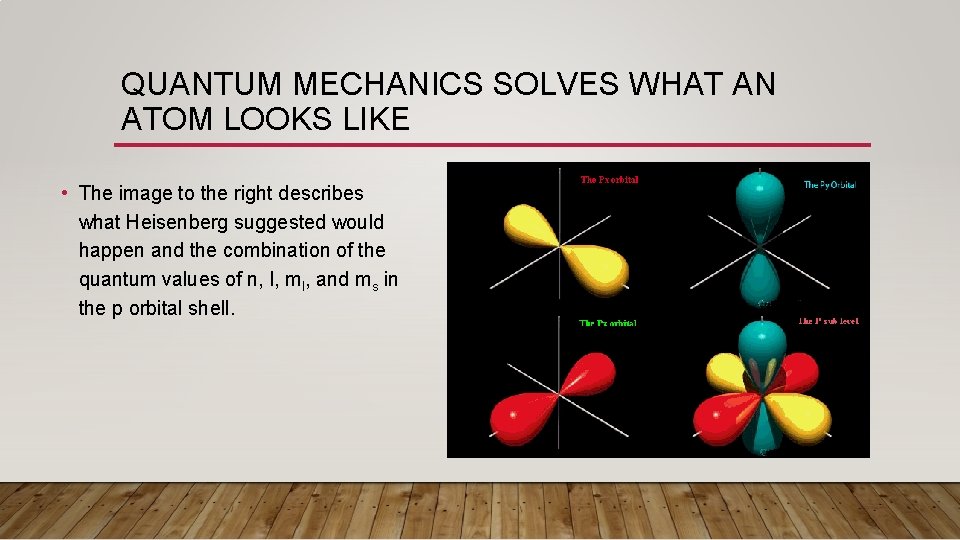
QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • The image to the right describes what Heisenberg suggested would happen and the combination of the quantum values of n, l, ml, and ms in the p orbital shell.

QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • The image to the right describes what Heisenberg suggested would happen and the combination of the quantum values of n, l, ml, and ms in the d orbital shell.

QUANTUM MECHANICS SOLVES WHAT AN ATOM LOOKS LIKE • The image to the right describes what Heisenberg suggested would happen and the combination of the quantum values of n, l, ml, and ms in the d orbital shell. • Disclaimer note – my brain DEFINTELY cannot handle what this would look like exactly.

SIGNIFICANCE OF QUANTUM MECHANICS • All the things we know to understand in life – electronics, chemistry, biology, organic molecules, the table you are writing on and etc… has all been calculated through mathematical equations. • If you think about it, everything we see, observe and do has been calculated mathematically. • There is a great movie called “What the Bleep” where they use our understanding of quantum mechanics to actually understand depression and what actually happens in our brains.

SCIENCE AND THE CORE COMPETENCIES • Using mathematics we deduce something is happening – critical thinking. • Then we use the math to draw out and describe what we are observing - creative thinking. • We then take make a personal decision to either share this information or not – personal awareness. • We then decide what kind of responsibility we have in sharing this information with society – social responsibility (solar panels, atomic bomb, computers, electronics, biological warfare, etc…) • In sharing this information we have to write it out so our peers can agree with our findings, and then express the ideas so the regular lay person can understand - communications. • In doing all this we then reflect on what we have done – personal and cultural identity.

CONGRATS • You have come to the end of the Physics 11 journey. • Now lets get in a circle and discuss the year that has unfolded.