Quantum Mechanics In quantum mechanics mechanics the electrons









- Slides: 15

Quantum Mechanics In quantum mechanics, mechanics the electrons occupy specific energy levels (as in Bohr's model) but they also exist within specific probability volumes called orbitals with specific orientations in space. The electrons within each orbital has a distinct spin. n = The principle quantum number Describes the possible energy levels and pictorially it describes the orbital size. n = 1, 2, 3…. where an orbital with the value of 2 is larger than an orbital with the value of 1. 1 s 2 s

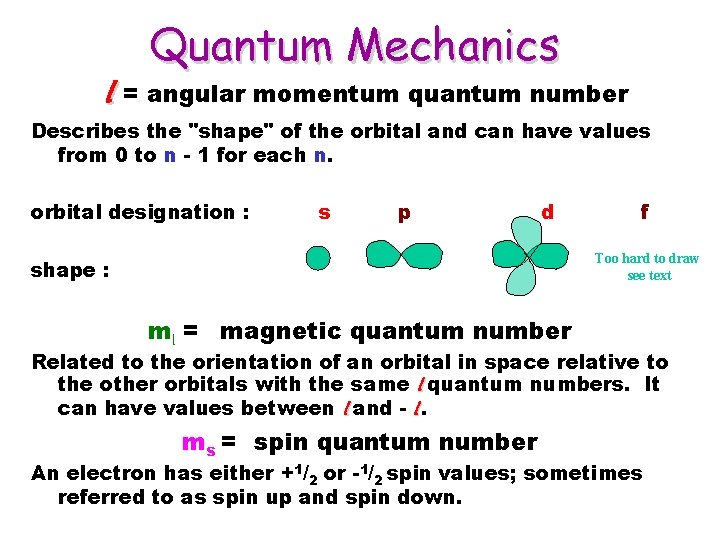
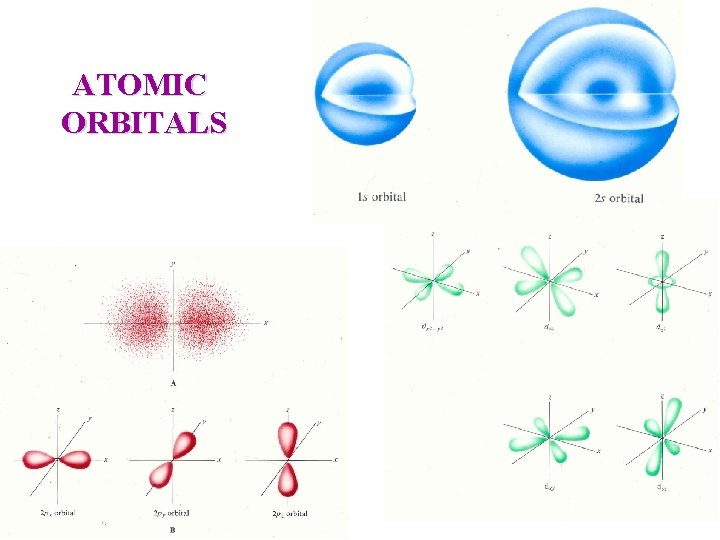
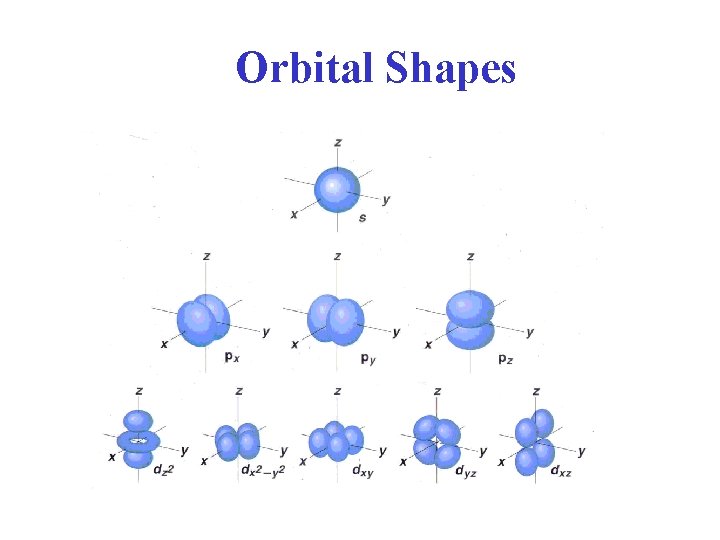
Quantum Mechanics l = angular momentum quantum number Describes the "shape" of the orbital and can have values from 0 to n - 1 for each n. orbital designation : s p d f Too hard to draw see text shape : ml = magnetic quantum number Related to the orientation of an orbital in space relative to the other orbitals with the same l quantum numbers. It can have values between l and - l. ms = spin quantum number An electron has either +1/2 or -1/2 spin values; sometimes referred to as spin up and spin down.

ATOMIC ORBITALS

Electron Configuration Electron configuration is a shorthand notation for describing the arrangement of the electrons about the nucleus. General Format using the quantum numbers: n = principle quantum number l = angular momentum quantum n l enumber e- = number of electrons RULES: 1. Fill the lowest energy levels first. Lowest 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 2. No more than two electrons per orbital.

Electron Configuration Examples: H : 1 s 1 He: 1 s 2 Li : 1 s 2 2 s 1 Co: 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 7 Br: 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 5 The condensed electron configuration distinguishes the core electrons from the valence electrons. CORE electrons are tightly held to the nucleus and resemble a noble gas configuration. VALENCE electrons are the outer most electrons and are involved in chemical reactions. Examples of the condensed configuration: Li: [He] 2 s 1 Co: [Ar] 4 s 2 3 d 7 2 10 5

Orbital Shapes

Electron Configuration Practice Problems 1. Give the full electron configuration: C O Ne Na Si Cl Ar K 2. Give the condensed electron configuration: C O Ne Na Si Cl Ar K Answers on next slide

Electron Configuration Answers 1 & 2. Give the full & condensed electron configuration: C 1 s 2 2 p 2 or [He] 2 s 2 2 p 2 O 1 s 2 2 p 4 or [He] 2 s 2 2 p 2 Ne 1 s 2 2 p 6 or [Ne] Na 1 s 2 2 p 6 3 s 1 or [Ne] 3 s 1 Si 1 s 2 2 p 6 3 s 2 3 p 2 or [Ne] 3 s 2 3 p 2 Cl 1 s 2 2 p 6 3 s 2 3 p 5 or [Ne] 3 s 2 3 p 5 Ar 1 s 2 2 p 6 3 s 2 3 p 6 or [Ar] K 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 or [Ar]4 s 1

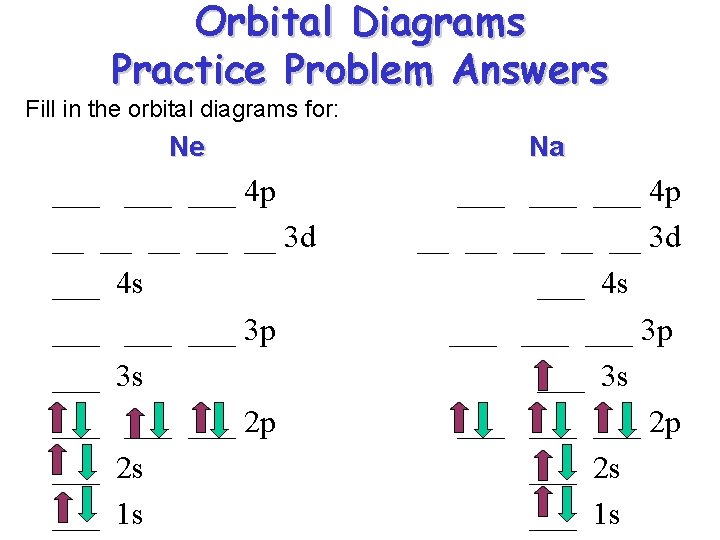
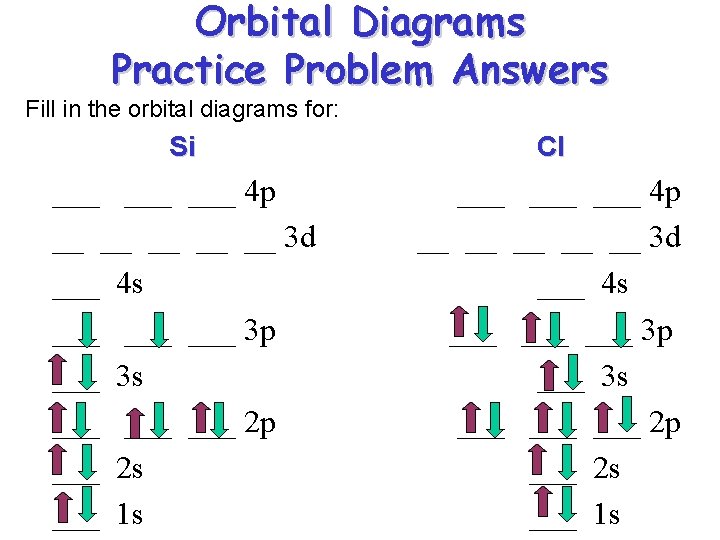
Orbital Diagrams Orbital diagrams are written in order of increasing energy levels starting with the lowest energy level the 1 s orbital. ___ ___ ___ 4 p ___ ___ 3 d 4 s ___ 3 p 3 s ___ 2 p 2 s Remember the order!! 1 s RULES: (1) fill the lowest energy level first (2) fill each orbital in a subshell with one electron first before you double up. (3) Completely fill each subshell before proceeding to the next energy level.


Orbital Diagrams Practice Problems 1. Fill in the orbital diagrams for: C O Ne Na ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ 3 p ___ 3 s ___ ___ 2 p ___ 2 s ___ 1 s Si Cl Ar K ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ 3 p ___ 3 s ___ ___ 2 p ___ 2 s ___ 1 s

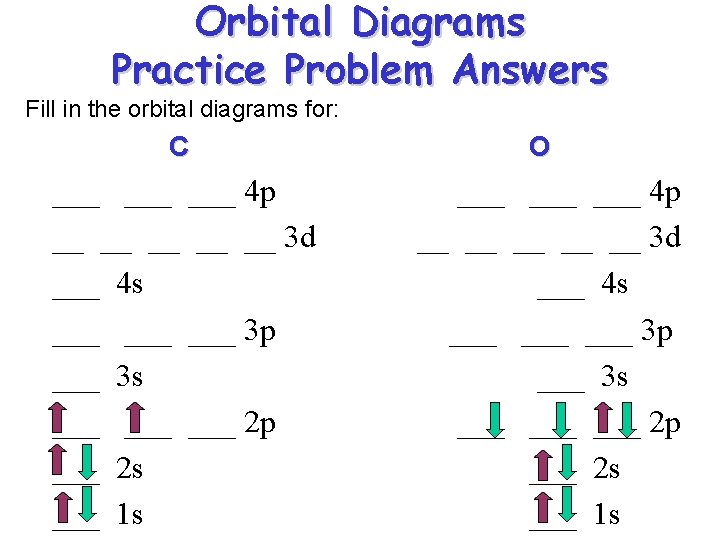
Orbital Diagrams Practice Problem Answers Fill in the orbital diagrams for: C ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ 3 p ___ 3 s ___ ___ 2 p ___ 2 s ___ 1 s O ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ 3 p ___ 3 s ___ ___ 2 p ___ 2 s ___ 1 s

Orbital Diagrams Practice Problem Answers Fill in the orbital diagrams for: Ne Na ___ ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ ___ 3 p ___ 3 s ___ ___ ___ 2 p ___ 2 s ___ 1 s

Orbital Diagrams Practice Problem Answers Fill in the orbital diagrams for: Si Cl ___ ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ ___ 3 p ___ 3 s ___ ___ ___ 2 p ___ 2 s ___ 1 s

Group Orbital Diagrams Study Problems Fill in the orbital diagrams and then write the electron configuration (both full and condensed) for: B F Ca P ___ ___ 4 p __ __ __ 3 d ___ 4 s ___ ___ 3 p ___ 3 s ___ ___ 2 p ___ 2 s ___ 1 s S As Zn Pb