Recognizing DrugInduced Liver Injury DILI in Exposed Populations






































- Slides: 48

Recognizing Drug-Induced Liver Injury (DILI) in Exposed Populations John R. Senior, M. D. Associate Director for Science Office of Pharmacoepidemiology and Statistical Science Food and Drug Administration (FDA) 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 1

Material presented here is based on the observations of the speaker for 20 years in academic hepatology and gastroenterology, 5 years as a senior executive in the pharmaceutical industry, 11 years in private consulting to industry, and upon 9. 5 years at the FDA: 4. 5 years as a medical reviewer for new gastrointestinal drugs, 3 as the Senior Scientific Advisor for hepatology in the Office of Drug Safety, 2 years as Associate Director for Science, Office of Pharmacoepidemiology and Statistical Science. Comments made here do not reflect official policies or positions of the Agency, but are personal opinions of the presenter based on his diverse experiences mentioned. 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 2

First, Detect it ! – (if you don’t ask or look, you won’t get or find) • Ask: Is there liver injury or disease? –serum transaminases, other enzymes, bilirubin, INR • Is it progressive or serious? –progressive = getting worse or likely to do so –serious = disabling, life-threatening, fatal • Drug-induced or some other cause? – no pathognomonic test for DILI, including biopsy – DILI may mimic any known liver disease 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 3

“some other causes” • What are they? – mainly acute hepatitis : viral A or B, seldom C, alcoholic, biliary stones, ischemic, or autoimmune; other causes rarer • How can they be ruled out? – what information should be collected? • What are the odds of other causes? – It’s really a problem of differential diagnosis. 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 4

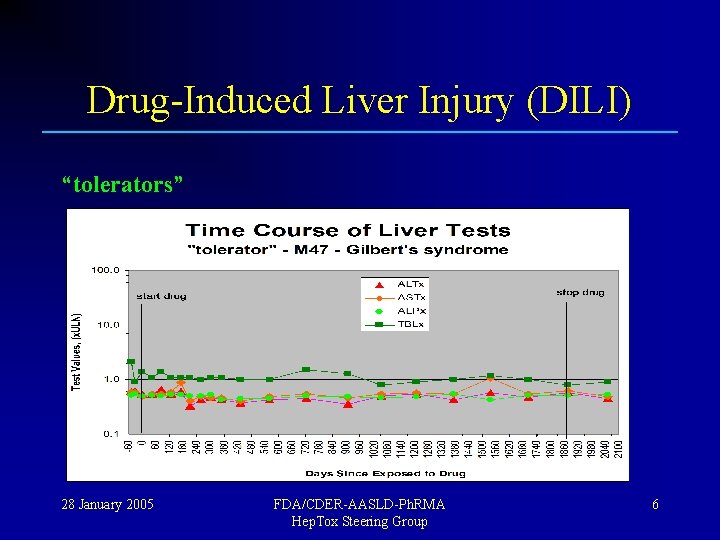
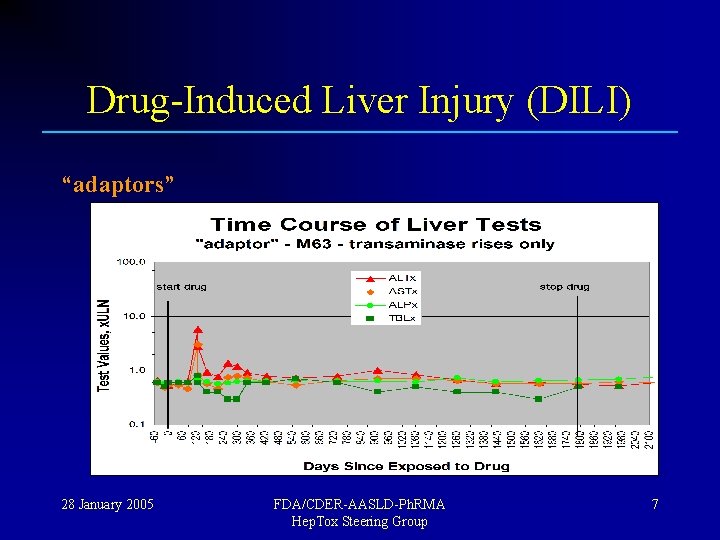
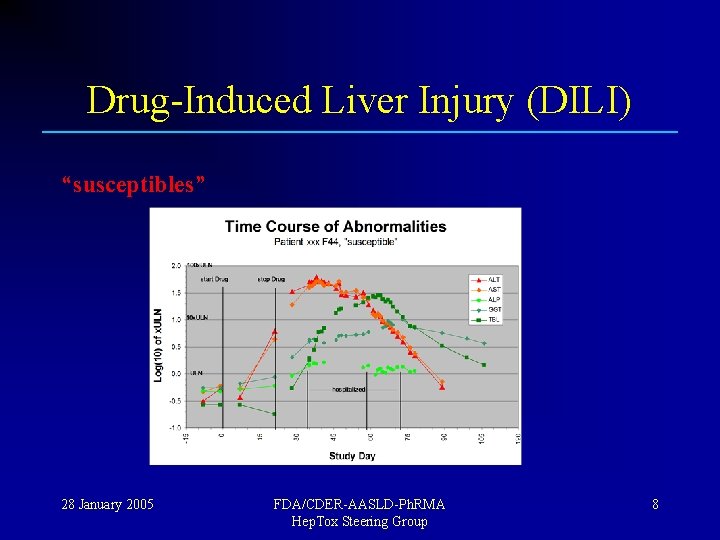
Drug-Induced Liver Injury (DILI) Most people exposed to a new drug show no injury; “tolerators” Some people show transient injury, but adapt; “adaptors” A few fail to adapt and show serious toxicity ! “susceptibles” 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 5

Drug-Induced Liver Injury (DILI) “tolerators” 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 6

Drug-Induced Liver Injury (DILI) “adaptors” 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 7

Drug-Induced Liver Injury (DILI) “susceptibles” 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 8

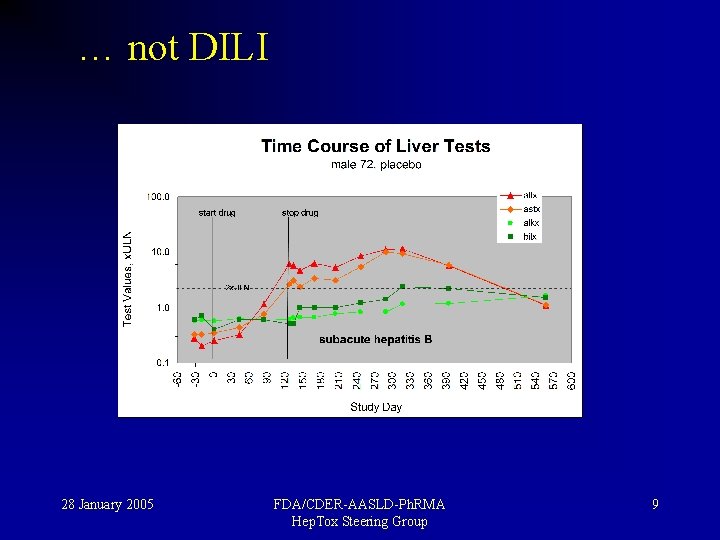
… not DILI 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 9

Is It a Bad Drug or Is It a Susceptible Patient? • • Why do a few people not tolerate it? What’s different about them? Can we find out? A drug tolerated by nearly everybody except a very few cannot be considered “toxic. ” • And not give them this drug? 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 10

Given: Some Drugs are More Toxic • • many or most eliminated in development molecular variations in class differing potencies of variants therapeutic index varies variable PK ADME among patients variable PD responses in patients one dose may (will) not suit all patients 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 11

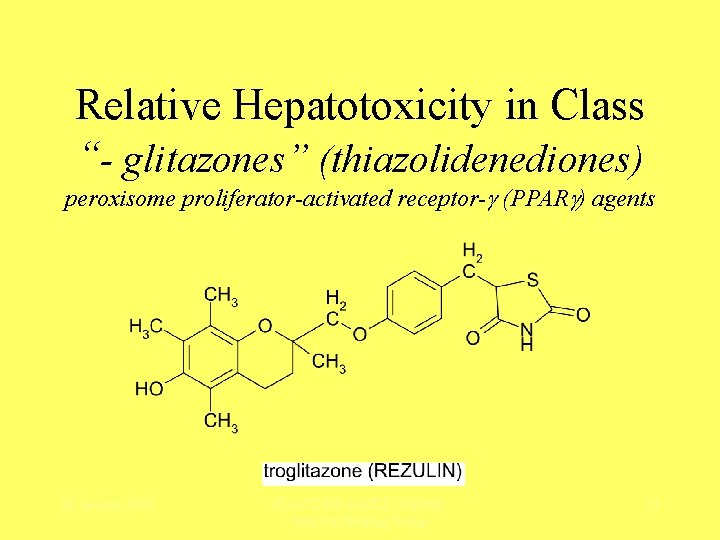
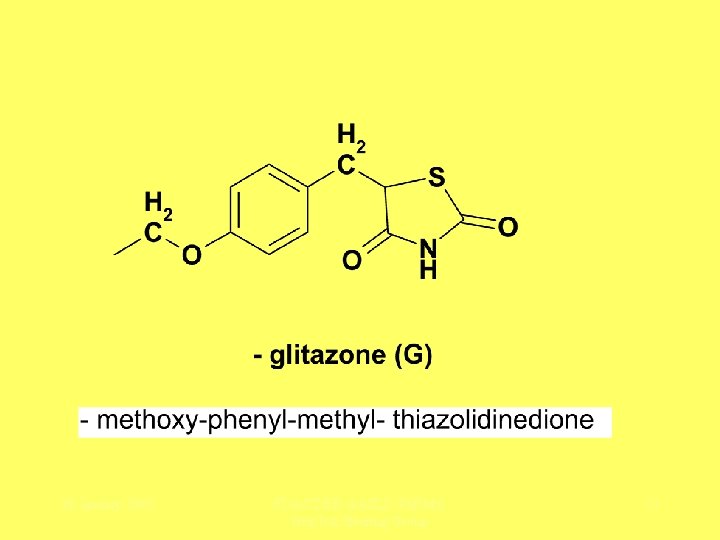
Relative Hepatotoxicity in Class “- glitazones” (thiazolidenediones) peroxisome proliferator-activated receptor- (PPAR ) agents 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 12

28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 13

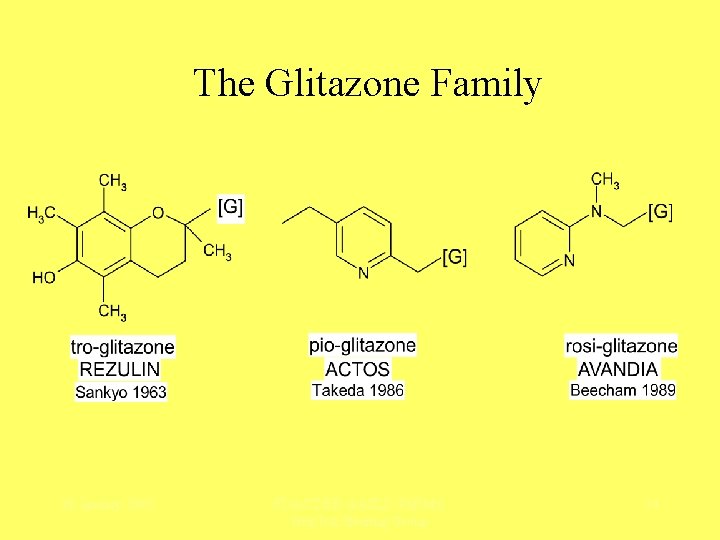
The Glitazone Family 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 14

Other Drug “Families” • “-profens” – benoxaprofen – flurbiprofen – ibuprofen • “-pidems” – alpidem – zolpidem • “-navirs” • “-fenacs” – ibufenac – bromfenac – diclofenac 28 January 2005 – – ritonavir indinavir nefinavir saquinavir FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 15

Need research on. . . 1) which cytochrome isoforms attack what part of the molecules? 2) which metabolites are more toxic? 3) proof that efficacy and toxicity are in different parts of same molecule ? 4) some rules to predict DILI 5) mechanisms causing the DILI 6) not just in animals, but in humans 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 16

Should We Be Looking More Carefully at the Patients ? • • Humans are often very different than animal models They are incredibly diverse genetically They resist being standardized or controlled They take a lot of drugs, diets, supplements, alcohol They have lots of other diseases/disorders/activities The ones treated are not the same as the ones studied They don’t always report troubles promptly 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 17

" Our study is man, as the subject of accidents or diseases. Were he always, inside and outside, cast in the same mould, instead of differing from his fellow man as much in constitution and in his reaction to stimulus as in feature, we should ere this have reached some settled principles in our art. The practice of medicine is an art, based on science. . . it has not reached, perhaps never will, the dignity of a complete science, with exact laws, like astronomy or engineering. ” William Osler, M. D. (1849 -1919) “Teacher and Student, ”Minneapolis 1892 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 18

Hippocrates 460 -377 B. C. The Father of Medicine 28 January 2005 " …he who aspires to treat correctly of human regimen must first acquire knowledge and discernment of the nature of man in general – knowledge of its primary constituents and discernment of the components by which it is controlled. … though they are made from the same materials, no two are alike…” FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 19

Hippocrates “Nature of Man” elements qualities fire hot water wet air cold earth dry constituents blood phlegm bile - yellow bile - black “Airs, Waters, Places” “Epidemics” “Nutriment” 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 20

Why Are They Susceptible ? “idio-sug-krasia” (Hippocrates, ~ 400 B. C. ) idios ( ) - one’s own, self syn ( ) - together crasis ( ) - mixing, mixture therefore, a person’s own individual mixture of characteristics, factors; “nature and nurture, ” unique in the world [it does NOT mean rare, unexpected, unexplained, although it may or may not be any or all of them] 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 21

4*4@FL(6 DVF 4 V (idiosugkrasia) s. f. , (substantive, feminine) temperament; constitution Pocket Oxford Greek Dictionary Oxford University Press, Oxford UK, 2000 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 22

Factors in Idiosyncrasy • genetic, bestowed at conception –gender –cytochromes, enzymes, transport systems • acquired in life since conception –age, activities, travels –infections, immunities, diseases –diet, obesity, dietary supplements –other drugs, chemical exposures 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 23

Especially Susceptible Patients • • adverse effect, often not dose-related may not be duration-related may not depend on prior disease unexpected, unpredictable (up to now) risk factors not well known toxicity often uncommon or rare who are they? how can they be identified? 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 24

Drug Development Issues: Idiosyncratic Drug Toxicity • Occurs despite careful preclinical testing in animals • … and despite careful and costly clinical trials • Some problem (perhaps wrong) assumptions: – – Assuming drug variability is the key to safety Obsolete concepts of “safe and effective” – for all? Assuming one dose/regimen suits all patients Focus on efficacy, little on safety (especially if rare) 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 25

Needles in the Haystack Tyranny of the Numbers - I • if only 1 patient in 1000 reacts adversely to a drug, should we call the drug “toxic”? • If 1 person in 1000 is idiosyncratically hyper -susceptible to show drug-induced liver injury, how many patients need to be observed to have 95% chance of finding at least 1? 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 26

A Binomial Answer. . . - if i = incidence of a rare event, and u = usual absence of the event, then (i + u) = 1, it happens or it doesn’t. And (i + u)N, where N = number of persons - when u. N, the chance of seeing no events in a group of N persons, is <0. 05, then the chance of seeing at least one is >95%, because 1 – u. N = 1 - <0. 05 = >95% - for (0. 001 + 0. 999)N, need 2995 people to make (0. 999)2995 = 0. 04996, which is <0. 05; - in general, N 3/i for rare events. 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 27

Detection of Rare Safety Problems • Before approval and marketing – NDA review: seldom have enough patients, so rare but serious event may not be observed; but if they are seen, investigate! • After marketing and clinical use – spontaneous, voluntary reporting useful, but severely underreported and incomplete data – problems usually worse after marketing 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 28

Liver “Function” Tests • what are they ? • what do they mean ? Important: distinguish between liver function and liver injury • they’re the biomarkers we use 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 29

Commonly Used Tests “transaminases”: ALT enzymes AST alkaline phosphatase gamma-glutamyl transferase bilirubin (total, “direct”) substances 28 January 2005 injury hepatocellular cholestatic function excretory albumin synthetic prothrombin (INR) synthetic FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 30

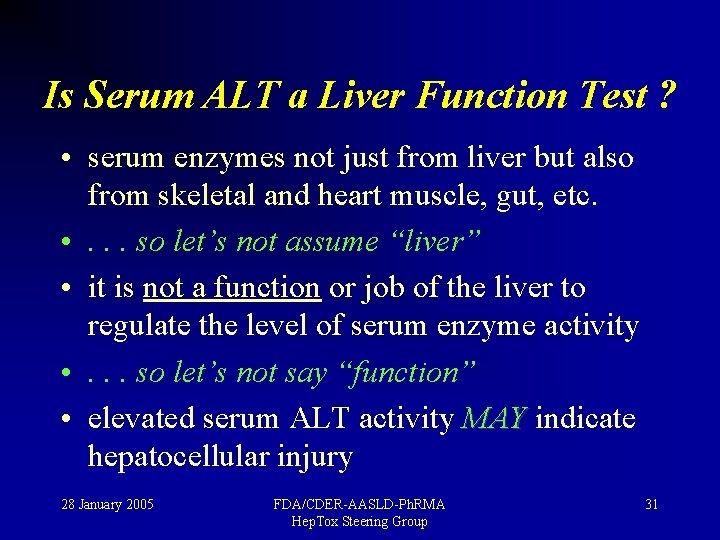
Is Serum ALT a Liver Function Test ? • serum enzymes not just from liver but also from skeletal and heart muscle, gut, etc. • . . . so let’s not assume “liver” • it is not a function or job of the liver to regulate the level of serum enzyme activity • . . . so let’s not say “function” • elevated serum ALT activity MAY indicate hepatocellular injury 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 31

Is Serum Transaminase Enough? 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 32

Need Very, Very Specific Tests Tyranny of the Numbers - II • For relatively low prevalence or incidence events, need extremely high specificity of test to avoid “finding” many false positives • Almost impossible to find a hyper-specific test; compound tests may help: ALT+TBL • Probably need to add clinical differential diagnostic data and methods to attain 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 33

Hy Zimmerman 1917 -1999 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 34

ESTIMATED CASE FATALITY RATE IN DRUGINDUCED ACUTE HEPATOCELLULAR INJURY WITH NON-OBSTRUCTIVE JAUNDICE Zimmerman. HEPATOTOXICITY, 1978 & 1999 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 35

Intrinsic vs Idiosyncratic Toxicity • “intrinsic” – – – • “idiosyncratic” predictable dose-related similar in animals high incidence short interval types • directly destructive • indirect, metabolic • cholestatic 28 January 2005 – – – unpredictable not dose-related not seen in animals low incidence, rare variably longer interval types • hypersensitivity • metabolic …H. Zimmerman, 1978 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 36

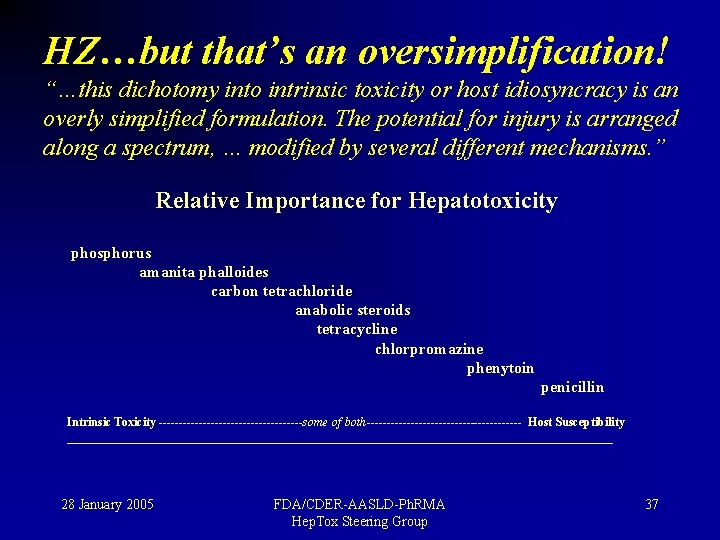
HZ…but that’s an oversimplification! “…this dichotomy into intrinsic toxicity or host idiosyncracy is an overly simplified formulation. The potential for injury is arranged along a spectrum, … modified by several different mechanisms. ” Relative Importance for Hepatotoxicity phosphorus amanita phalloides carbon tetrachloride anabolic steroids tetracycline chlorpromazine phenytoin penicillin Intrinsic Toxicity ------------------some of both-------------------- Host Susceptibility ______________________________________________ 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 37

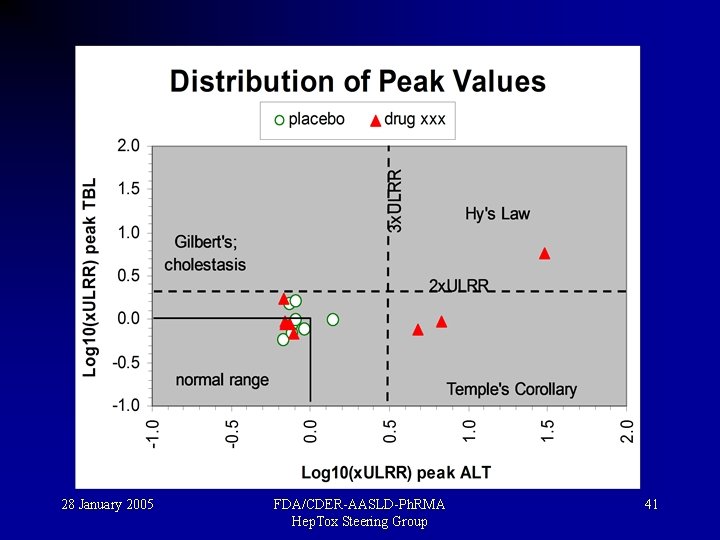
“Hy’s Law”. . . for drug-induced hepatotoxicity • If both hepatocellular injury and jaundice occur, look out for at least 10% mortality! • i. e. , when both transaminase and bilirubin elevations occur together. Robert Temple, FDA, 1999 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 38

Searching for Clues, Predictors • Can ways be found to search pre-approval clinical databases (NDA submissions) for clues or “signals” that predict liver failure in at least some who show the signal? • Hy Zimmerman was on to a key concept the combination of an indicator of injury and loss of overall function may be a valid predictor. It needs confirmation. 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 39

“Temple’s Corollary”. . . for drug-induced hepatotoxicity: Hy’s Law cases arise out of a background of increased incidence of transaminase elevations • Hy’s Rule-type serious cases of DILI usually arise from a background of increased injury rates, so • Look for more ALT elevations in those exposed to drug, compared to placebo or control drug. Robert Temple, FDA, 2004 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 40

28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 41

What to Do When a “Signal” Detected “signal” = laboratory abnormality, complaint, finding • immediately (within day or two) confirm by repeat tests, examination, questions • investigate possible causes AT ONCE ! r/o disease • start close observation (3 x/week) immediately • obtain more information about possible causes • obtain consultation as appropriate or needed • follow closely (weekly, prn) to normalization or worsening and management decision 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 42

It may be DILI if it’s nothing else • • • Diagnosis of exclusion; no test FOR DILI Must gather data to rule out other causes Need to educate “docs” to do it better Develop model for quantitative likelihood estimate Prospective large safety studies needed: • • • for true incidence for risk factors and to design risk management plans for ‘omic’ analyses (gen-, prote-, metabon-) specimens for elucidation of mechanisms 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 43

Tip of the Iceberg Death or Tx Acute Liver Failure Serious DILI – Threatening Detectable DILI – but Not Serious Patient Adaptation to New Agent Exposure Patients/People Tolerate Exposure Without Effects 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 44

Serious Adverse Events • • • Often have relatively low incidence rate, i Difficult to establish correct value for i Voluntary reporting gives uncertain values May be missed in the usual efficacy trials Must balance modest clinical benefits for many against serious risks for a few • Spontaneous reports (AERS) not enough 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 45

Retrospective Database Studies • • • Epidemiologic, observational – closer to reality A definite improvement on spontaneous reports Limited to who was included, data recorded No attribution of causality; statistical differences Can improve estimates of incidence, risk factors But cannot investigate mechanisms or causes or “do it right” in real time, collect specimens 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 46

How many patients to study? • • • Prospective, not retrospective Focus on safety, as drug actually prescribed Large and long enough to find rare events Full accounting, numbers of cases/exposed Use patient self-reporting, then investigate possible cases intensely; get needed info • Prospective case-control design • Impartial study conduct: NIH or AHRQ 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 47

Prospective Large Safety Studies • • • Offer best chance to learn, and to protect patients Need to establish true incidence of DILI Need to identify risk factors for rational RM Need to investigate mechanisms In best interests of everybody, including patients, regulators, and sponsors • In 2005, are we content with safety meaning “not poison, ” and withdrawing “toxic” drugs ? 28 January 2005 FDA/CDER-AASLD-Ph. RMA Hep. Tox Steering Group 48