Polymer Synthesis CHEM 421 Chapter 3 6 Odian



![Routes to Oligomers DP = [ M ]o Polymer Synthesis CHEM 421 Use large Routes to Oligomers DP = [ M ]o Polymer Synthesis CHEM 421 Use large](https://slidetodoc.com/presentation_image_h/178fe1f9fdbce172dcd50be98aae7681/image-4.jpg)









- Slides: 25

Polymer Synthesis CHEM 421 • Chapter 3 -6 (Odian)

Oligomers Polymer Synthesis CHEM 421 “Oligomer” – Greek: oligos, “few” mer, “parts” • Find commercial application in a variety of fields: Elastomers - poly(ethylene oxide) oligomers in Spandex® Coatings & Adhesives - acrylic oligomers Lubricants - fluorinated oligomers used as lubricants on satellites, disk drives, etc…

Free Radical Solution Polymerizations • Initiation • Propagation • Termination Polymer Synthesis CHEM 421
![Routes to Oligomers DP M o Polymer Synthesis CHEM 421 Use large Routes to Oligomers DP = [ M ]o Polymer Synthesis CHEM 421 Use large](https://slidetodoc.com/presentation_image_h/178fe1f9fdbce172dcd50be98aae7681/image-4.jpg)
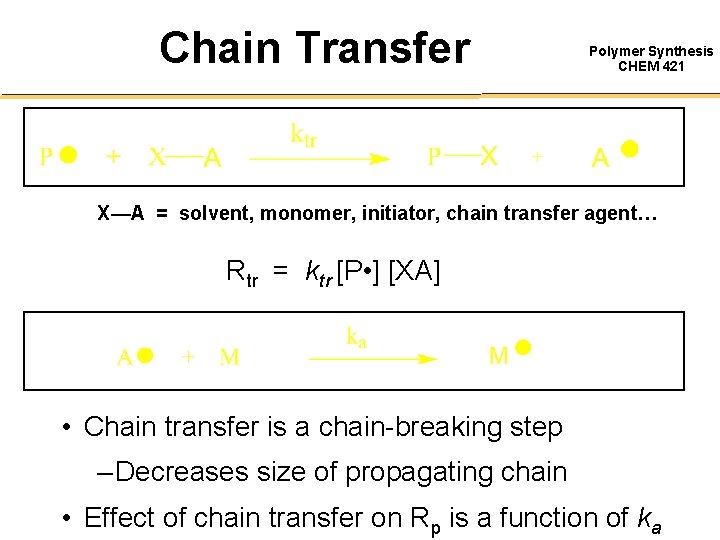
Routes to Oligomers DP = [ M ]o Polymer Synthesis CHEM 421 Use large amounts of initiator: • very expensive • high levels of azo-initiators leads to toxic cross-coupling products [ I ]o Use low monomer concentrations: • low productivity • requires lots of solvents Alternative is to use ‘Chain Transfer” Processes…

Chain Transfer Polymer Synthesis CHEM 421 X—A = solvent, monomer, initiator, chain transfer agent… Rtr = ktr [P • ] [XA] • Chain transfer is a chain-breaking step – Decreases size of propagating chain • Effect of chain transfer on Rp is a function of ka

Chain Transfer Polymer Synthesis CHEM 421 • Aliphatic hydrocarbons with strong C—H bonds show low CT • Benzene even lower • Alkyl aromatics (benzylic H’s) – t-butyl benzene↓, no benzylic H • Butyl iodide (weak C—I bond) • Acids, ethers, amines, alcohols >> than aliphatics due to heteroatom stabilization • Weak S—S bond • Halogenated solvents, weak C —X bond • Thiols the largest!

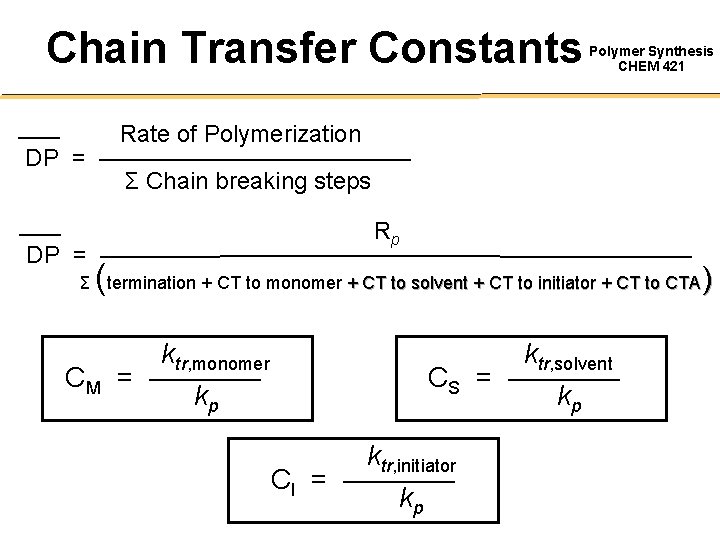
Chain Transfer Constants Polymer Synthesis CHEM 421 Rate of Polymerization DP = ——————— Σ Chain breaking steps Rp DP = ———————————— ( ) Σ termination + CT to monomer + CT to solvent + CT to initiator + CT to CTA CM ktr, monomer = ———— kp ktr, solvent CS = ———— kp ktr, initiator CI = ———— kp

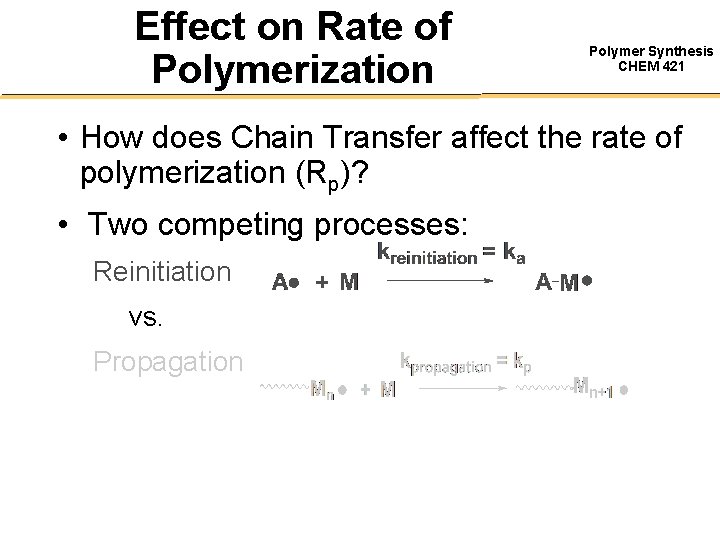
Effect on Rate of Polymerization Polymer Synthesis CHEM 421 • How does Chain Transfer affect the rate of polymerization (Rp)? • Two competing processes: Reinitiation vs. Propagation

Effect on Degree of Polymerization Polymer Synthesis CHEM 421 • How does Chain Transfer affect the degree of polymerization (DP)? • Two competing processes: Transfer vs. Propagation

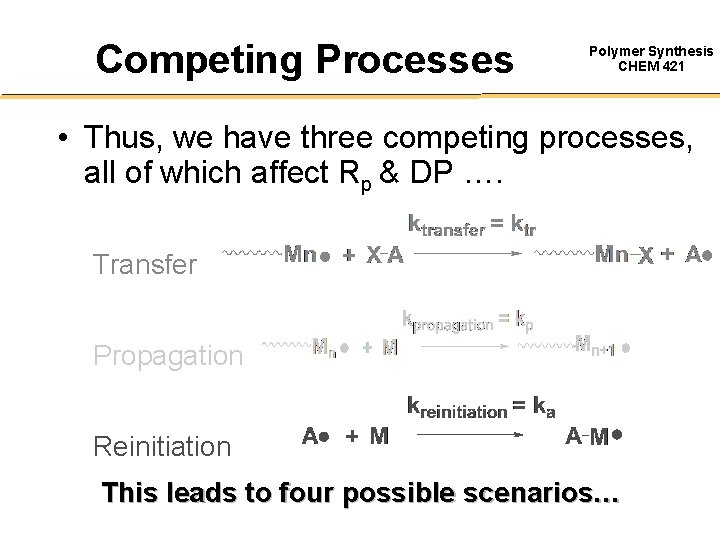
Competing Processes Polymer Synthesis CHEM 421 • Thus, we have three competing processes, all of which affect Rp & DP …. Transfer Propagation Reinitiation This leads to four possible scenarios…

1 st Case Polymer Synthesis CHEM 421 “ Normal Chain Transfer ” Rate of Propagation >> Rate of Transfer kp >> ktr Rate of Reinitiation ≈ Rate of Propagation ka ≈ kp • No effect on Rate of Polymerization (Rp) ie. same # of monomers consumed / unit time • Decrease in Degree of Polymerization (DP)

2 nd Case Polymer Synthesis CHEM 421 “ Telomerization ” Rate of Propagation << Rate of Transfer kp << ktr Rate of Reinitiation ≈ Rate of Propagation ka ≈ kp • Still no effect on Rate of Polymerization (Rp) ie. same # of monomers consumed / unit time • Huge Decrease in Degree of Polymerization (DP) DP = 1 -5 repeat units!!! Unlike 1 st case, transfer (kt) is more rapid than propagation (kp) !!

3 rd Case Polymer Synthesis CHEM 421 “ Retardation ” Rate of Propagation >> Rate of Transfer kp >> ktr Rate of Reinitiation < Rate of Propagation ka < kp • Decrease in Rate of Polymerization (Rp) Rp is decreased b/c reinitiation (kr) is slower!! • Decrease in Degree of Polymerization (DP)

4 th Case Polymer Synthesis CHEM 421 “ Degradative Chain Transfer ” Rate of Propagation << Rate of Transfer kp << ktr Rate of Reinitiation < Rate of Propagation ka < kp • Decrease in Rate of Polymerization (Rp) Like Retardation, re-initiation is slow… • Large decrease in Degree of Polymerization (DP) Different from Retardation, Transfer (kt) is rapid

Chain Transfer Constant (Cs) Polymer Synthesis CHEM 421 Mathematical Definition: Cs = ktr kp Transfer Propagation The magnitude of Cs reflects the activity of the chain transfer agent …

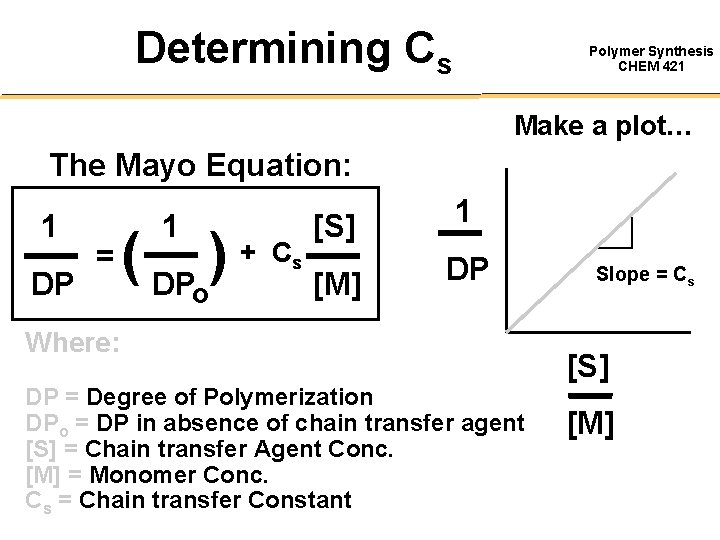
Determining Cs Polymer Synthesis CHEM 421 Make a plot… The Mayo Equation: 1 DP = 1 ( DPo) + C s [S] 1 [M] DP Where: DP = Degree of Polymerization DPo = DP in absence of chain transfer agent [S] = Chain transfer Agent Conc. [M] = Monomer Conc. Cs = Chain transfer Constant Slope = Cs [S] [M]

Common Chain Transfer Agents Polymer Synthesis CHEM 421 Advantages Disadvantages • Very reactive • Toxicity • Commercially available • Stench (Thiols) • Some able to functionalize polymer end groups • Non-Catalytic (ie. very low MWs require high conc. ) • Some are inexpensive

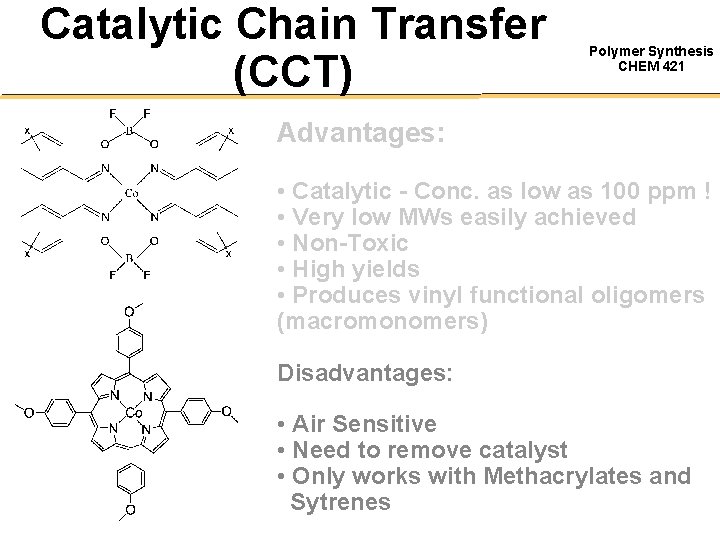
Catalytic Chain Transfer (CCT) Polymer Synthesis CHEM 421 Advantages: • Catalytic - Conc. as low as 100 ppm ! • Very low MWs easily achieved • Non-Toxic • High yields • Produces vinyl functional oligomers (macromonomers) Disadvantages: • Air Sensitive • Need to remove catalyst • Only works with Methacrylates and Sytrenes

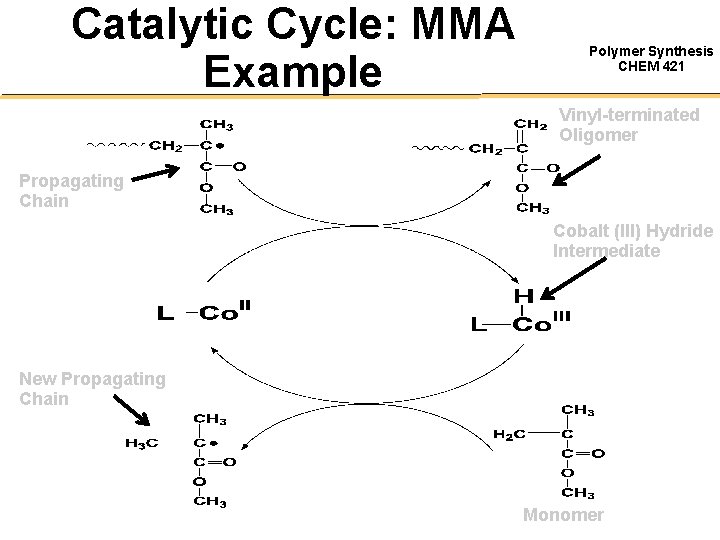
Catalytic Cycle: MMA Example Polymer Synthesis CHEM 421 Vinyl-terminated Oligomer Propagating Chain Cobalt (III) Hydride Intermediate New Propagating Chain Monomer

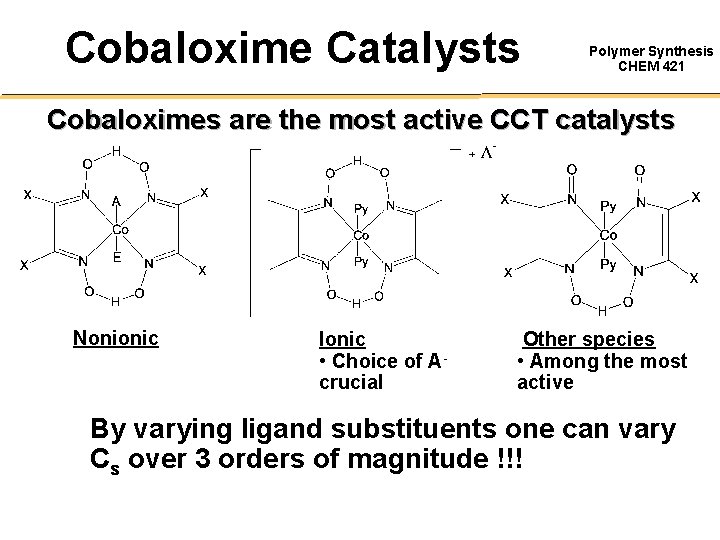
Cobaloxime Catalysts Polymer Synthesis CHEM 421 Cobaloximes are the most active CCT catalysts Nonionic Ionic • Choice of Acrucial Other species • Among the most active By varying ligand substituents one can vary Cs over 3 orders of magnitude !!!

BF 2 Bridging Ligands in CCT Catalysts Polymer Synthesis CHEM 421 First reported in 1981 by Nonaka et. al. Nonaka, T. ; Hamada, K. Bull. Chem. Soc. Jpn. 1981, 54 (10), 3185 • Current CCT catalyst of choice largely because of decreased sensitivity to oxygen • Crucial for CCT on industrial scales

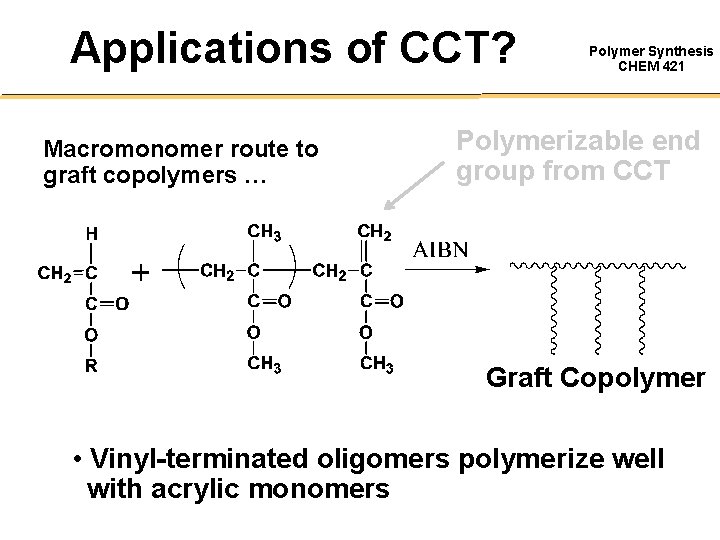
Applications of CCT? Macromonomer route to graft copolymers … Polymer Synthesis CHEM 421 Polymerizable end group from CCT Graft Copolymer • Vinyl-terminated oligomers polymerize well with acrylic monomers

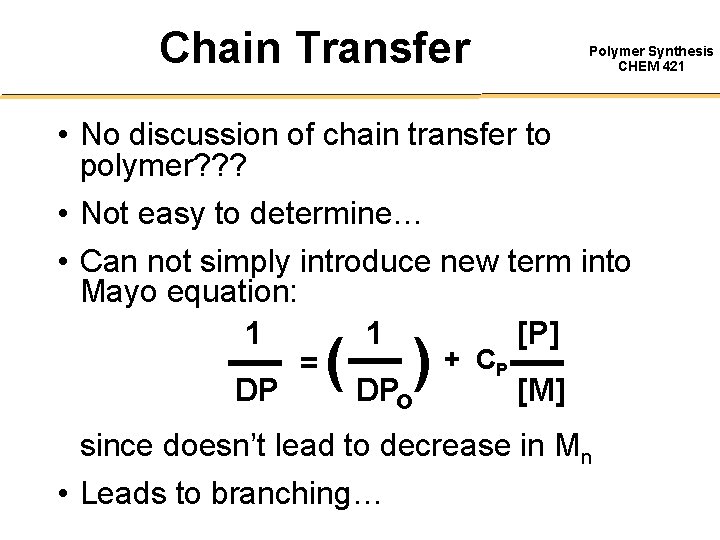
Chain Transfer Polymer Synthesis CHEM 421 • No discussion of chain transfer to polymer? ? ? • Not easy to determine… • Can not simply introduce new term into Mayo equation: 1 1 [P] + CP = DP DPo [M] ( ) since doesn’t lead to decrease in Mn • Leads to branching…

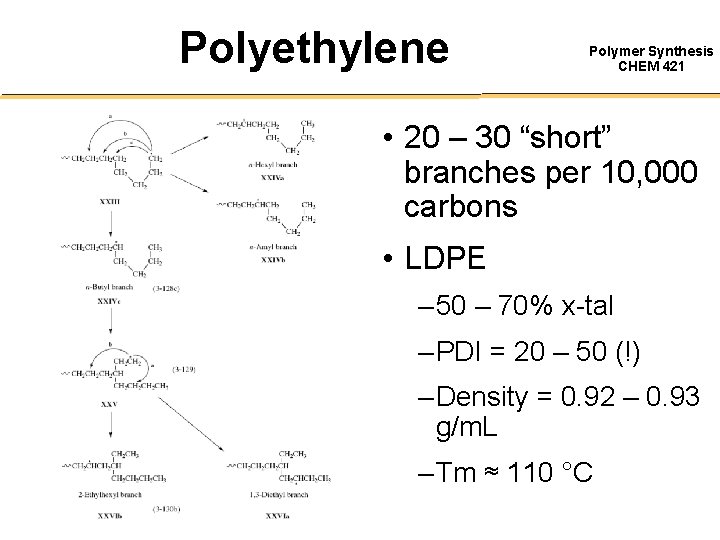
Polyethylene Polymer Synthesis CHEM 421 • 20 – 30 “short” branches per 10, 000 carbons • LDPE – 50 – 70% x-tal – PDI = 20 – 50 (!) – Density = 0. 92 – 0. 93 g/m. L – Tm ≈ 110 °C

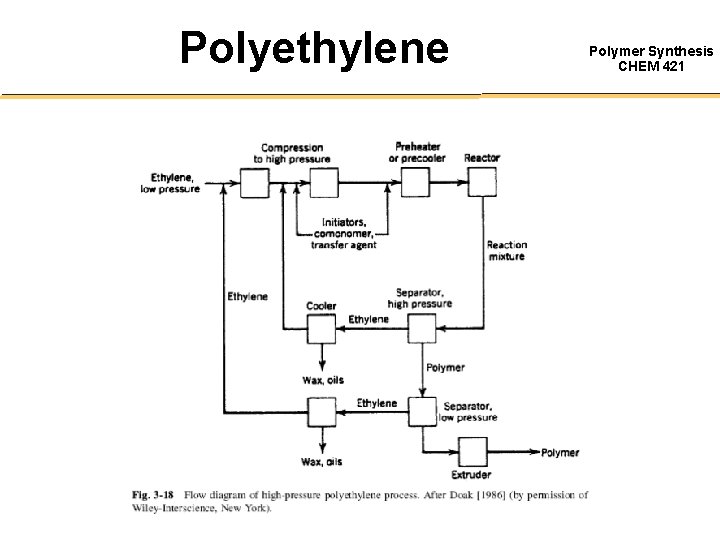
Polyethylene Polymer Synthesis CHEM 421