Polymer Synthesis CHEM 421 Reading Odian Book Chapter

Polymer Synthesis CHEM 421 Reading (Odian Book): Chapter 2 -1, 2 -2, 2 -4
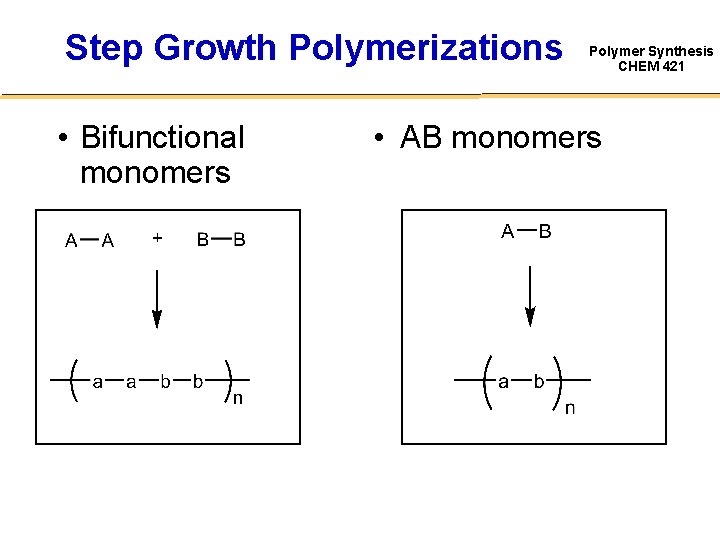
Step Growth Polymerizations • Bifunctional monomers Polymer Synthesis CHEM 421 • AB monomers
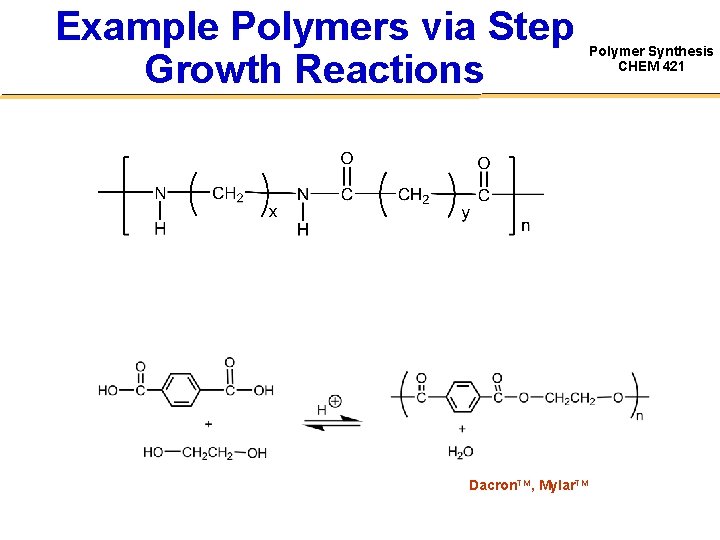
Example Polymers via Step Growth Reactions Dacron. TM, Mylar. TM Polymer Synthesis CHEM 421

Importance of Equal Reactivity Polymer Synthesis CHEM 421 A—B + A—B + A—B + A—B A—ba—B + A—ba—B + FA° = # of A groups at the beginning FA = # of A groups at any given time p = conversion = 1 – FA / FA° = 1 – (6 / 10) Xn = average degree of polymerization total # of molecules present initially Xn = ———————— total # of molecules present at time t A—B

Importance of Equal Reactivity Polymer Synthesis CHEM 421 A—B + A—B + A—B + A—B p = 0. 0 A—ba—B + A—ba—B + p = 0. 4 A—ba—ba—ba—B + + + A—ba—B + A—ba—ba—B + + Xn = 1 A—B Xn = 1. 67 A—B p = 0. 6 Xn = 2. 5 p = 0. 7 Xn = 3. 33 p = 0. 8 Xn = 5 A—B A—ba—ba—ba—B
![MW and Conversion Polymer Synthesis CHEM 421 Given that: [M] = [M]0 - [M]0 MW and Conversion Polymer Synthesis CHEM 421 Given that: [M] = [M]0 - [M]0](http://slidetodoc.com/presentation_image_h2/0339a81bbc741c0da9bc5b29a62e97a7/image-6.jpg)
MW and Conversion Polymer Synthesis CHEM 421 Given that: [M] = [M]0 - [M]0 p Rewriting: [M] = [M]0 (1 – p) Rewriting: 1 (1 – p) = Knowing: [M]0 Xn = [M] Xn = 1 (1 – p) [M]0 [M]

Implications of Carothers Equation • Conversion Xn – 50% 2 – 80% 5 – 90% 10 – 95% 20 – 99% 100 – 99. 5% 200 Xn = Polymer Synthesis CHEM 421 1 (1 – p)
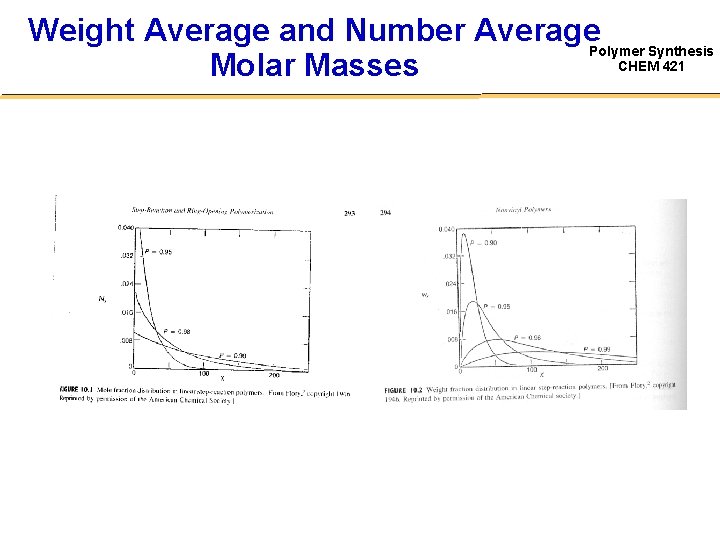
Weight Average and Number Average. Polymer Synthesis CHEM 421 Molar Masses

Factors Involved in the Synthesis of High MW Linear Polymers via Step-Growth Reactions Polymer Synthesis CHEM 421 • High purity monomers • Di-functionality • Proper stoichiometry • Very high conversions • No side reactions • Accessibility of mutually reacting groups • In general: – Suitable for bulk reactions – Moderate viscosity during much of the reaction – Incredible effort ($$) goes to pushing the reaction forward in last stages

Methods for Polyester Synthesis • Direct reaction • Acid halide / hydroxyl • Transesterification • Melt acidolysis Polymer Synthesis CHEM 421
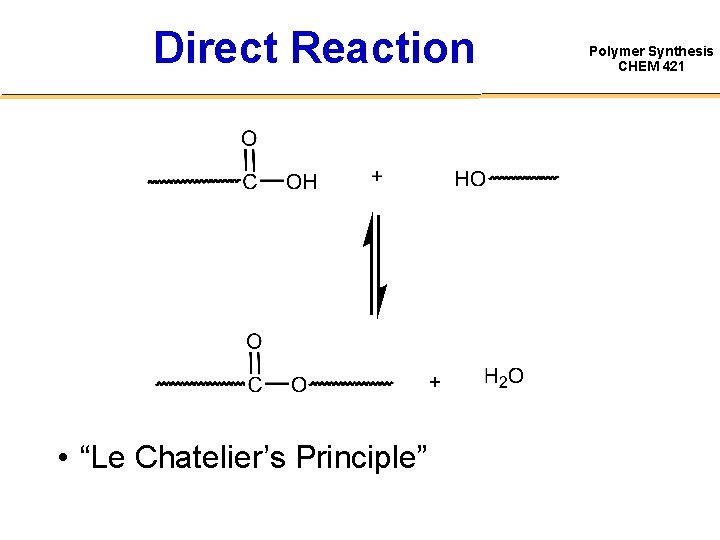
Direct Reaction • “Le Chatelier’s Principle” Polymer Synthesis CHEM 421
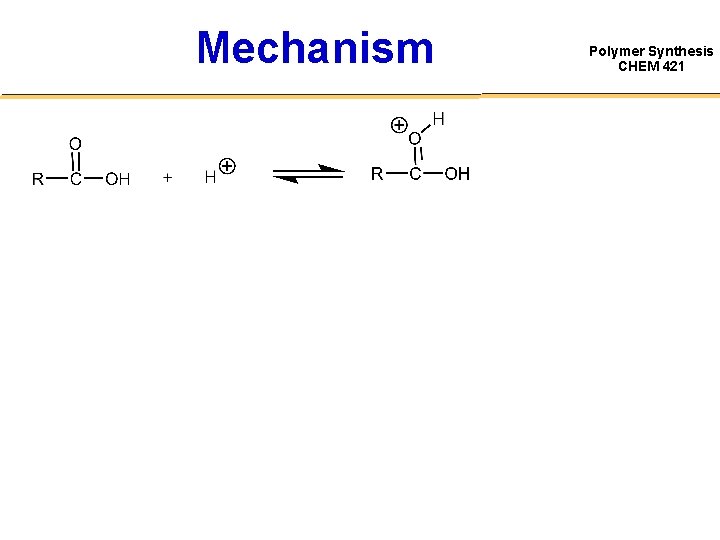
Mechanism Polymer Synthesis CHEM 421
![Overall Reaction Polymer Synthesis CHEM 421 • Self-catalyzed Rp [OH] [COOH] • Catalyzed by Overall Reaction Polymer Synthesis CHEM 421 • Self-catalyzed Rp [OH] [COOH] • Catalyzed by](http://slidetodoc.com/presentation_image_h2/0339a81bbc741c0da9bc5b29a62e97a7/image-13.jpg)
Overall Reaction Polymer Synthesis CHEM 421 • Self-catalyzed Rp [OH] [COOH] • Catalyzed by added acid ( [H+] = constant)
![Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] = Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] =](http://slidetodoc.com/presentation_image_h2/0339a81bbc741c0da9bc5b29a62e97a7/image-14.jpg)
Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] = ———— [RCOOH] [ROH] • Initial hydroxyl and carboxyl concentrations are [M]0 • Concentration of ester groups @ equilibrium is p [M]0 where p = extent of reaction @ equilibrium • The concentrations of hydroxyl and carboxyl groups @ equilibrium must therefore be: ( [M]0 – p [M]0) • Therefore Keq ( p [M]0)2 p 2 [M]02 p 2 = ———————— = ———— ( [M]0 – p [M]0)2 [M]02 ( 1– p )2
![Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] = Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] =](http://slidetodoc.com/presentation_image_h2/0339a81bbc741c0da9bc5b29a62e97a7/image-15.jpg)
Equilibrium Considerations: Closed System Keq Polymer Synthesis CHEM 421 [ester] [H 2 O] = ———— [RCOOH] [ROH] • Solve for p yields: K½ p = ———— 1+ K½ • Knowing that: Xn = 1 (1 – p) Xn = 1 + K ½
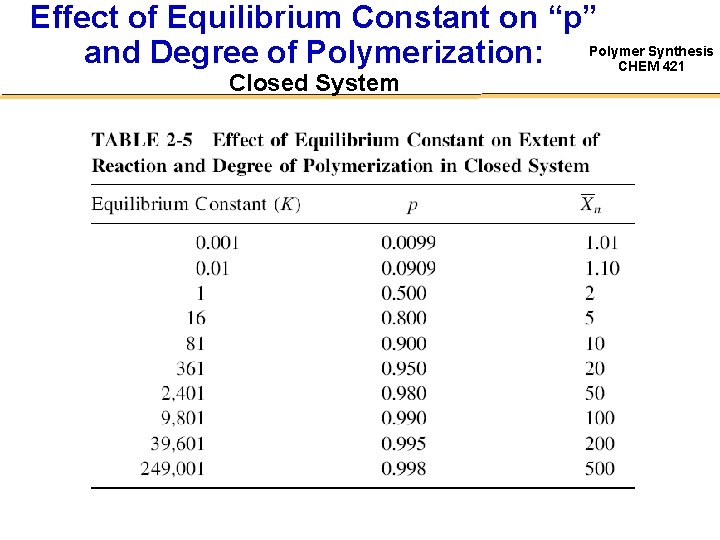
Effect of Equilibrium Constant on “p” Synthesis and Degree of Polymerization: Polymer CHEM 421 Closed System

Effect of Equilibrium Constant on “p” Synthesis and Degree of Polymerization: Polymer CHEM 421 Closed System • Indicates the limitation imposed by equilibrium on the synthesis of high MW polymer – @ Xn = 100 (corresponding to Mn ≈ 10 k) can be obtained in a closed system only if K is 104!!! – Can not be done in a closed system for most polymers… Polymer Polyesters Polycarbonates Transesterifications K 1 - 10 15 0. 1 – 1. 0 Polyamidation 102 - 103 • Therefore must drive the equilibrium

Open System Polymer Synthesis CHEM 421 • Extent to which one must drive the system in the forward reaction – can be seen by calculating the lowering of the small molecule condensate concentration that is necessary to achieve a particular MW – Knowing that [H 2 O] = p [M]0 Keq Xn = 1 (1 – p) ( p [M]0)2 p [H 2 O] (Xn)2 = —————— ( [M]0 – p [M]0)2 [M]0 ( 1– p )2 [M]0 – Solve for [H 2 O] K [M]0 [H 2 O] = ————— Xn ( Xn – 1) [H 2 O] 1 —— Xn 2

Drive the Equilibrium Polymer Synthesis CHEM 421 • Need to remove the small molecule condensate – H 2 O – HCl • Small molecule condensate needs to diffuse through and eventually out of the reaction mixture – Not easy because of high viscosity – Can lead to reactions becoming diffusion controlled
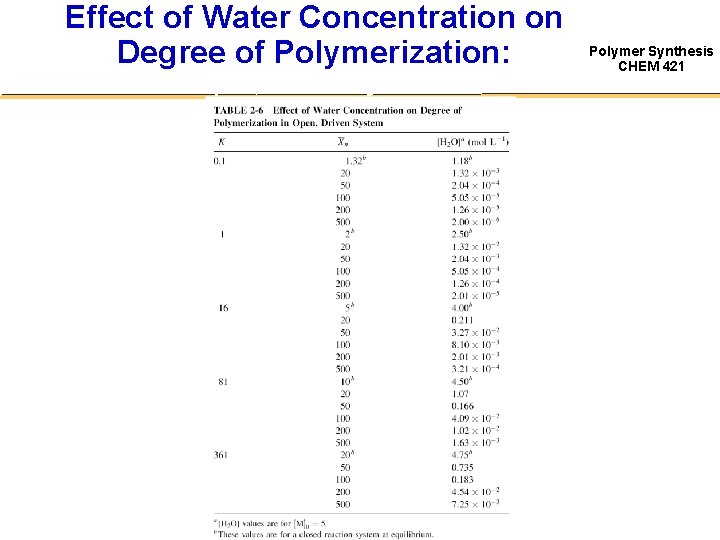
Effect of Water Concentration on Degree of Polymerization: Open, Driven System Polymer Synthesis CHEM 421

How best to drive the Equilibrium? Polymer Synthesis CHEM 421 • Mixing is energy and capital intensive – Wiped film reactors to increase surface area • Increase diffusivity of the condensate – Raise the temperature to lower the viscosity of the melt » Potential for side reactions – Swell the melt with solvents » Supercritical CO 2
- Slides: 21