Polycarbonates Interfacial Polymerizations Polymer Synthesis CHEM 421 Commercially
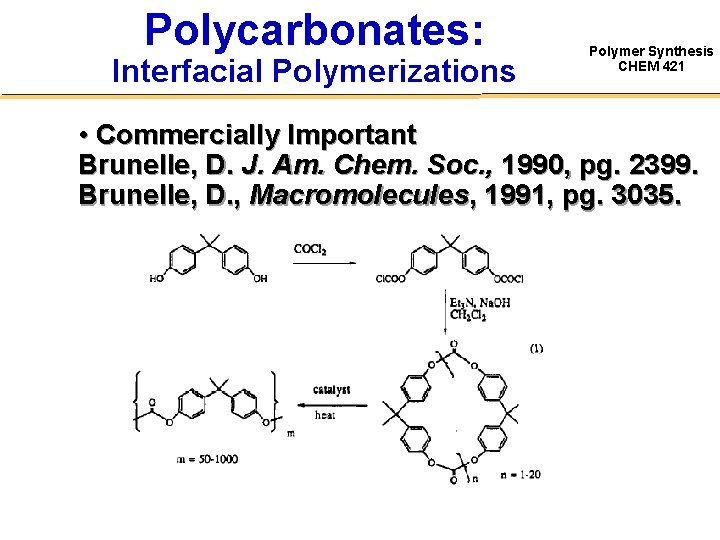
Polycarbonates: Interfacial Polymerizations Polymer Synthesis CHEM 421 • Commercially Important Brunelle, D. J. Am. Chem. Soc. , 1990, pg. 2399. Brunelle, D. , Macromolecules, 1991, pg. 3035.
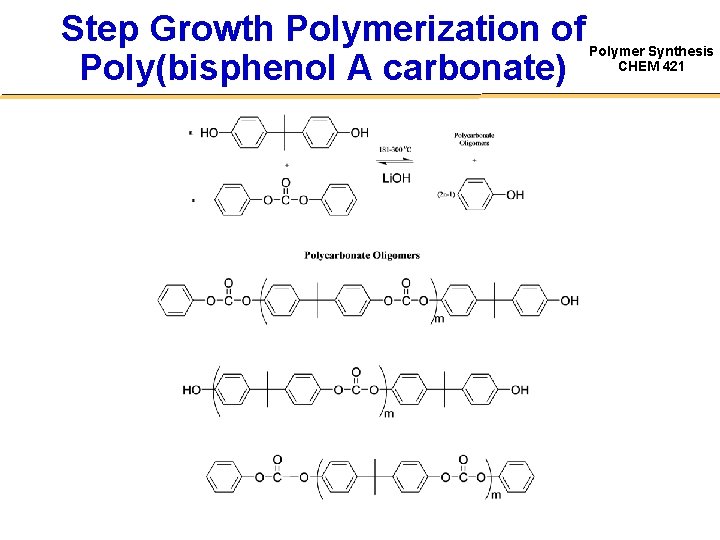
Step Growth Polymerization of Poly(bisphenol A carbonate) Polymer Synthesis CHEM 421
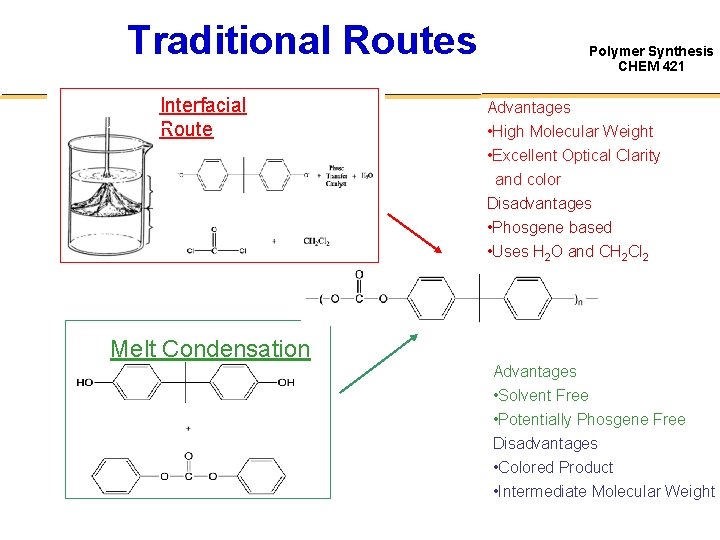
Traditional Routes Interfacial Route Polymer Synthesis CHEM 421 Advantages • High Molecular Weight • Excellent Optical Clarity and color Disadvantages • Phosgene based • Uses H 2 O and CH 2 Cl 2 Melt Condensation Advantages • Solvent Free • Potentially Phosgene Free Disadvantages • Colored Product • Intermediate Molecular Weight
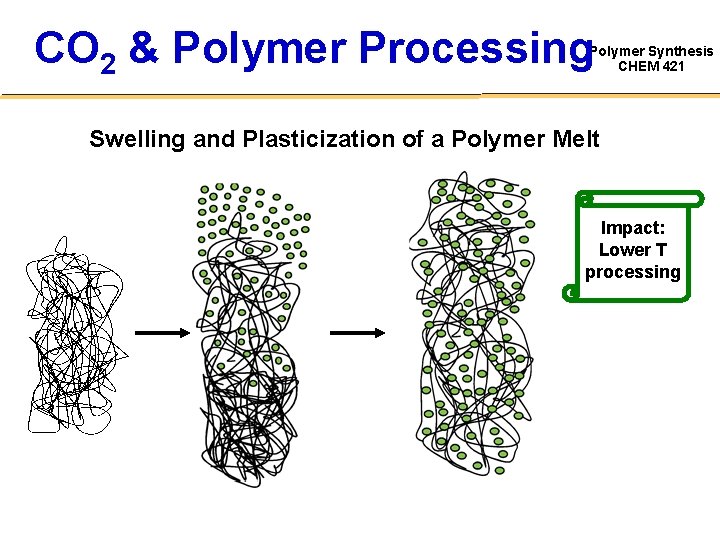
CO 2 & Polymer Processing Polymer Synthesis CHEM 421 Swelling and Plasticization of a Polymer Melt Impact: Lower T processing
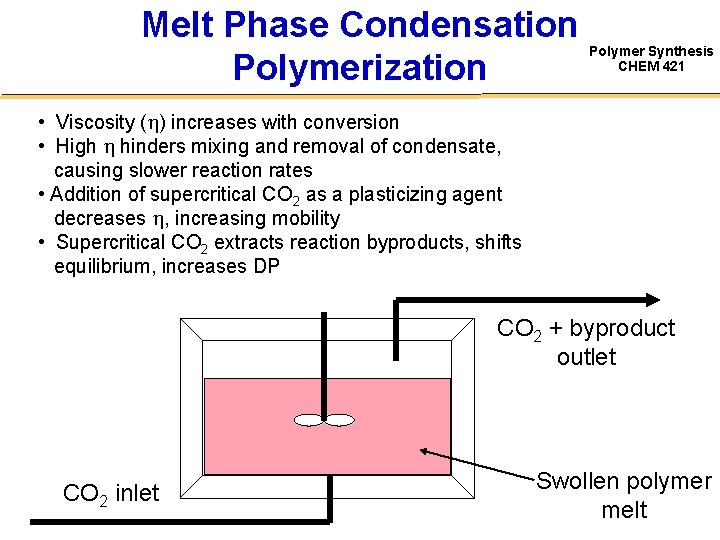
Melt Phase Condensation Polymerization Polymer Synthesis CHEM 421 • Viscosity ( ) increases with conversion • High hinders mixing and removal of condensate, causing slower reaction rates • Addition of supercritical CO 2 as a plasticizing agent decreases , increasing mobility • Supercritical CO 2 extracts reaction byproducts, shifts equilibrium, increases DP CO 2 + byproduct outlet CO 2 inlet Swollen polymer melt

Swelling Measurements 0 psi 2000 psi 3000 psi Polymer Synthesis CHEM 421 4000 psi 5000 psi • In a closed system, the polymer swelling correlates to CO 2 mass uptake by the polymer • Swelling measurements allow for the determination of the diffusion coefficient of CO 2 in the polymer

Polymer Synthesis CHEM 421

Solvent-Induced (CO 2) Crystallization of Polycarbonate Polymer Synthesis CHEM 421 Polycarbonate Pellets Crystallized with Supercritical CO 2 Amorphous Polycarbonate Pellets

Solid State Polymerization Polymer Synthesis CHEM 421 Synthesize amorphous prepolymer Crystallize the prepolymer with supercritical CO 2 to eliminate the need for organic solvents Heat the semicrystalline prepolymer between Tg and Tm Flow sweep fluid past the surface of the polymer particle to remove condensation byproduct Investigate the use of supercritical CO 2 as a sweep fluid Amorphous region Crystalline region

Solid State Polymerization: Solvent. Polymer Synthesis CHEM 421 Induced Crystallization Solvent induced crystallization presents a unique opportunity to study solid state polymerization PET PC -Thermally crystallizes crystallize -Fixed level of crystallinity -Uniform crystallinity -Does not readily thermally -Can control crystallinity -Can control morphology Solvent front
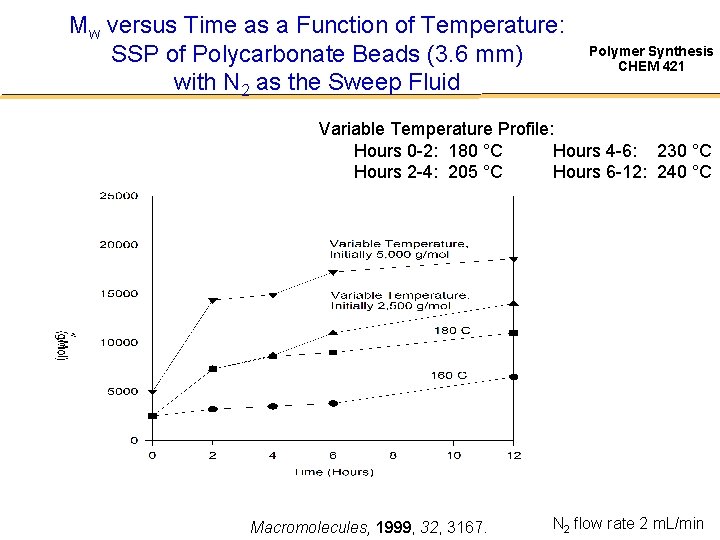
Mw versus Time as a Function of Temperature: SSP of Polycarbonate Beads (3. 6 mm) with N 2 as the Sweep Fluid Polymer Synthesis CHEM 421 Variable Temperature Profile: Hours 0 -2: 180 °C Hours 4 -6: 230 °C Hours 2 -4: 205 °C Hours 6 -12: 240 °C Macromolecules, 1999, 32, 3167. N 2 flow rate 2 m. L/min

Role of Phenol Diffusion Polymer Synthesis CHEM 421 Phenol must diffuse from the particle to increase polymer molecular weight. core intermediate shell Segment Radius Core = 0. 0 to 0. 4 mm Inter = 0. 4 to 1. 4 mm Shell = 1. 4 to 1. 8 mm The sample was separated into three regions for analysis. . . -molecular weight -percent crystallinity -melting point (Tm) What is the role of…. -phenol diffusivity -tortuosity -end group mobility
![MW as a Function of Diameter? Polymer Synthesis CHEM 421 [Condensate] Macromolecules, 2000, 33, MW as a Function of Diameter? Polymer Synthesis CHEM 421 [Condensate] Macromolecules, 2000, 33,](http://slidetodoc.com/presentation_image_h2/4ef479b7ed8d49e2d5321ee0901a9630/image-13.jpg)
MW as a Function of Diameter? Polymer Synthesis CHEM 421 [Condensate] Macromolecules, 2000, 33, 40. 1 —— Xn 2 PC Beads=3. 6 mm N 2 Flow=2 m. L/min
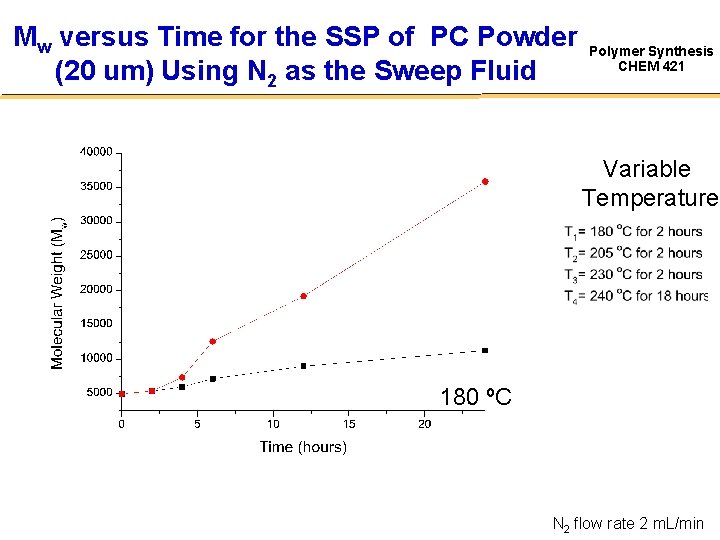
Mw versus Time for the SSP of PC Powder (20 um) Using N 2 as the Sweep Fluid Polymer Synthesis CHEM 421 Variable Temperature 180 ºC N 2 flow rate 2 m. L/min
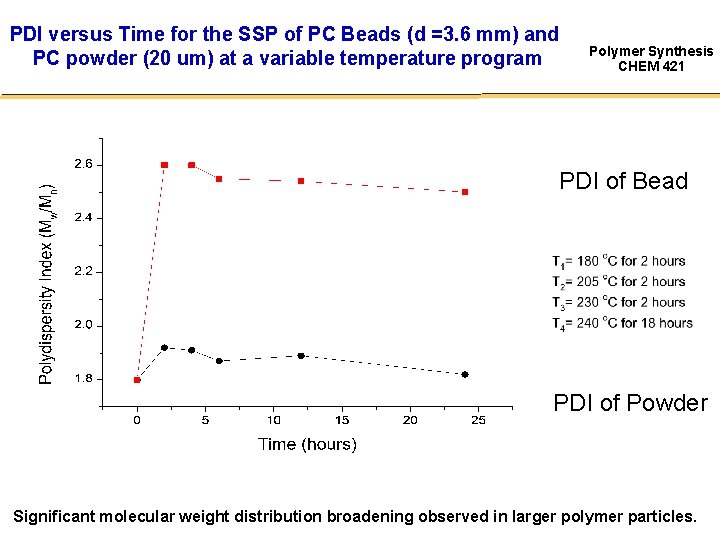
PDI versus Time for the SSP of PC Beads (d =3. 6 mm) and PC powder (20 um) at a variable temperature program Polymer Synthesis CHEM 421 PDI of Bead PDI of Powder Significant molecular weight distribution broadening observed in larger polymer particles.
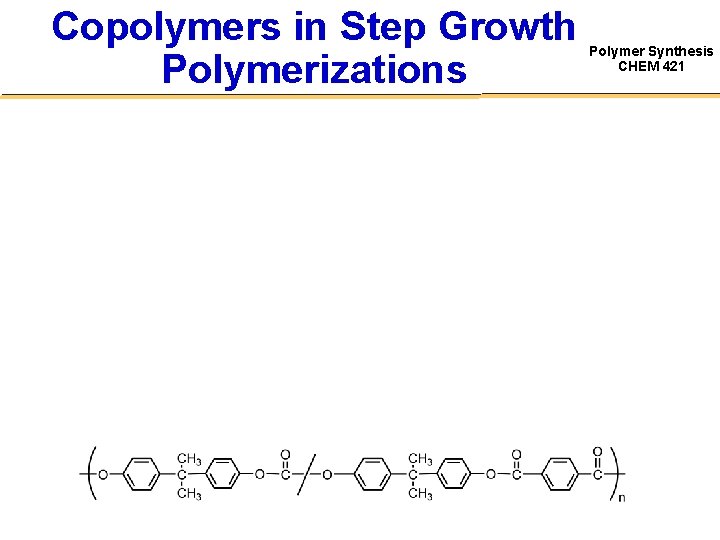
Copolymers in Step Growth Polymerizations Polymer Synthesis CHEM 421
- Slides: 16