Polymer Synthesis CHEM 421 Odian Book 2 12

Polymer Synthesis CHEM 421 • Odian Book 2 -12

Common Engineering Thermosets (Not elastomers) Polymer Synthesis CHEM 421 • Bismaleimides Step & Chain Growth • Epoxies Step & Chain Growth • Phenol / Formaldehyde Step Growth • Sheet Molding Compound Chain Growth • Polyurethanes** **Also thermoplastics Step Growth
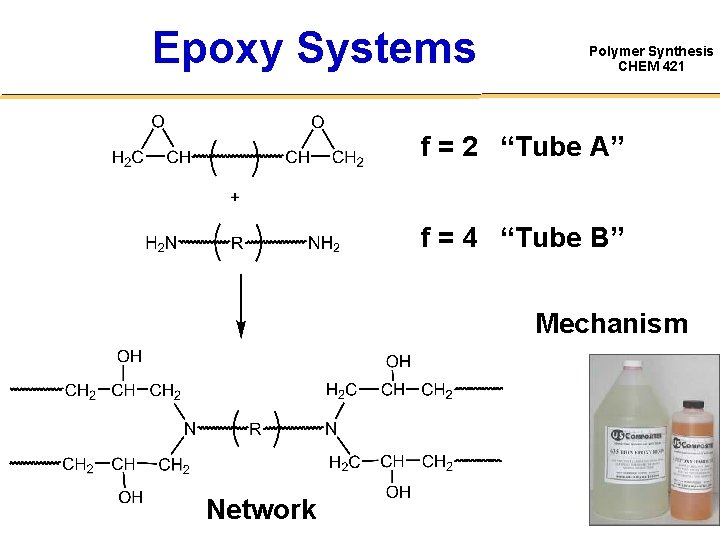
Epoxy Systems Polymer Synthesis CHEM 421 f = 2 “Tube A” f = 4 “Tube B” Mechanism Network
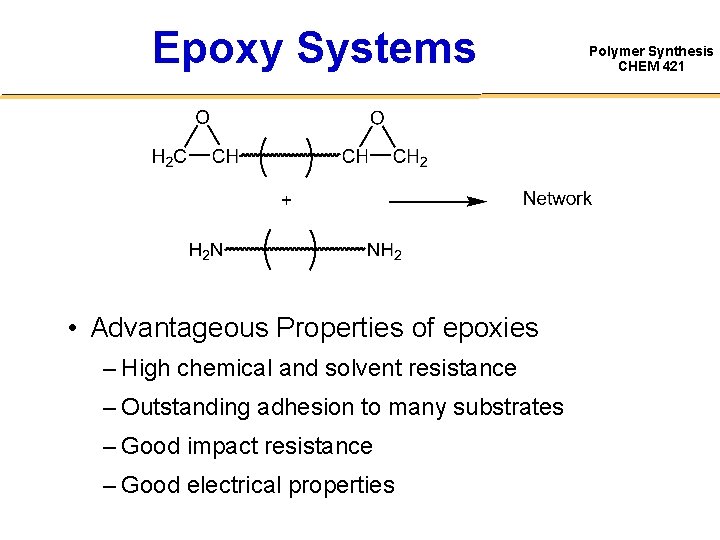
Epoxy Systems • Advantageous Properties of epoxies – High chemical and solvent resistance – Outstanding adhesion to many substrates – Good impact resistance – Good electrical properties Polymer Synthesis CHEM 421
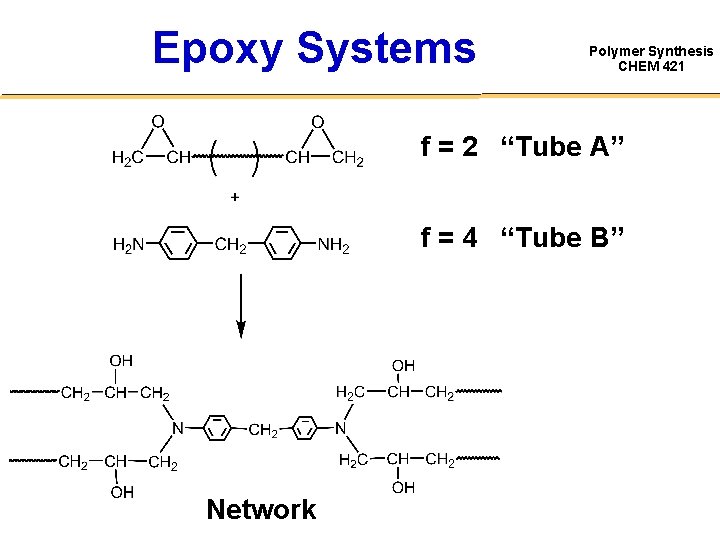
Epoxy Systems Polymer Synthesis CHEM 421 f = 2 “Tube A” f = 4 “Tube B” Network
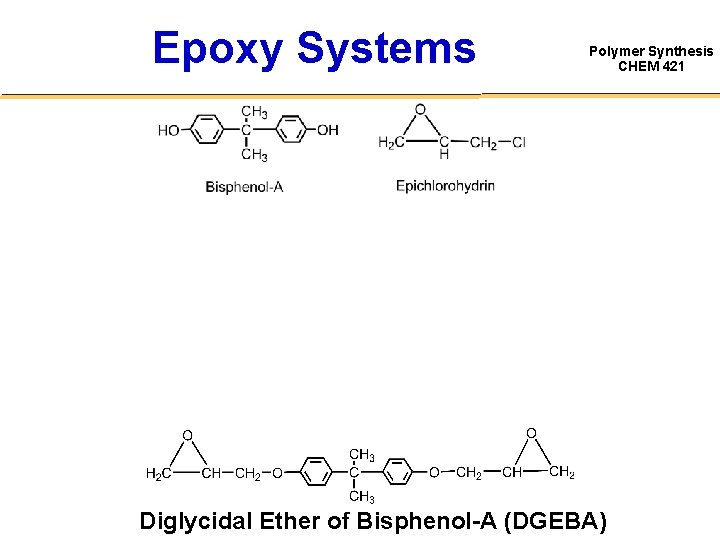
Epoxy Systems Polymer Synthesis CHEM 421 Diglycidal Ether of Bisphenol-A (DGEBA)
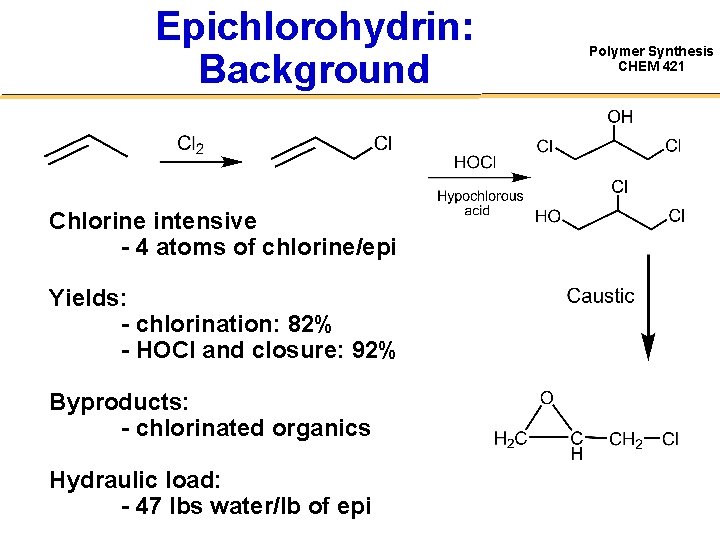
Epichlorohydrin: Background Chlorine intensive - 4 atoms of chlorine/epi Yields: - chlorination: 82% - HOCl and closure: 92% Byproducts: - chlorinated organics Hydraulic load: - 47 lbs water/lb of epi Polymer Synthesis CHEM 421
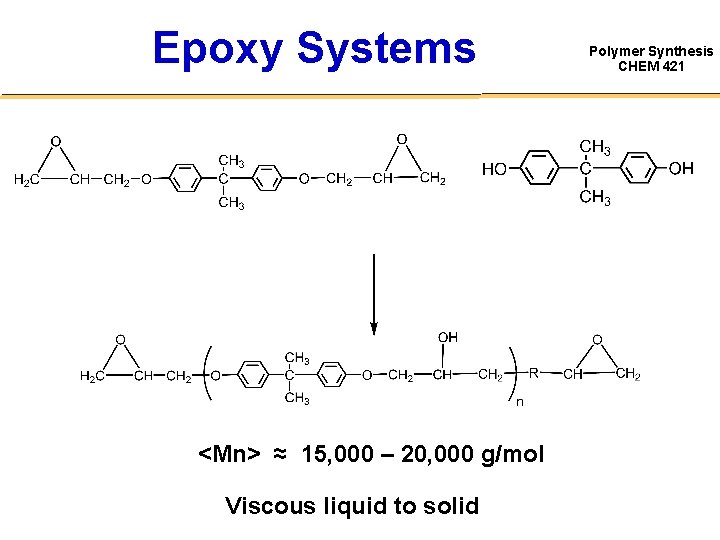
Epoxy Systems <Mn> ≈ 15, 000 – 20, 000 g/mol Viscous liquid to solid Polymer Synthesis CHEM 421
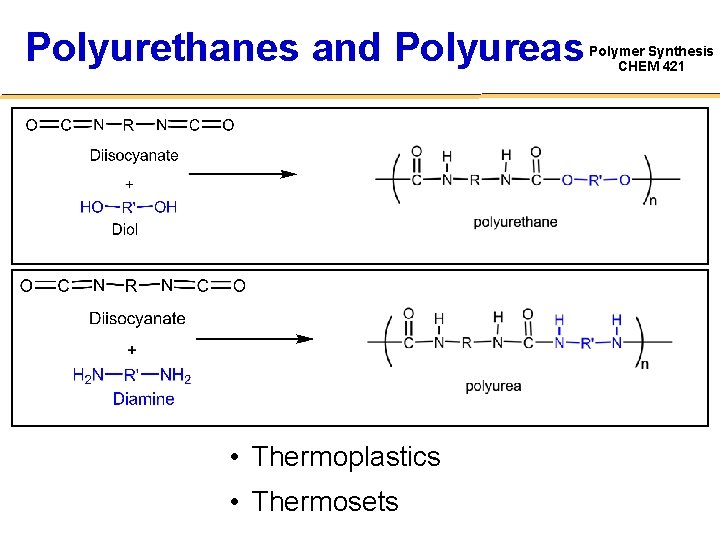
Polyurethanes and Polyureas • Thermoplastics • Thermosets Polymer Synthesis CHEM 421

Important Starting Materials for Polyurethanes Polymer Synthesis CHEM 421 • Diisocyantes • Polymeric Glycols (aka polyols) –MW < 3, 000 g/mol • Chain extenders • Catalysts –Trialkyl tin acetate –Dialkyl tin diacetate Relative rates 30, 000 x
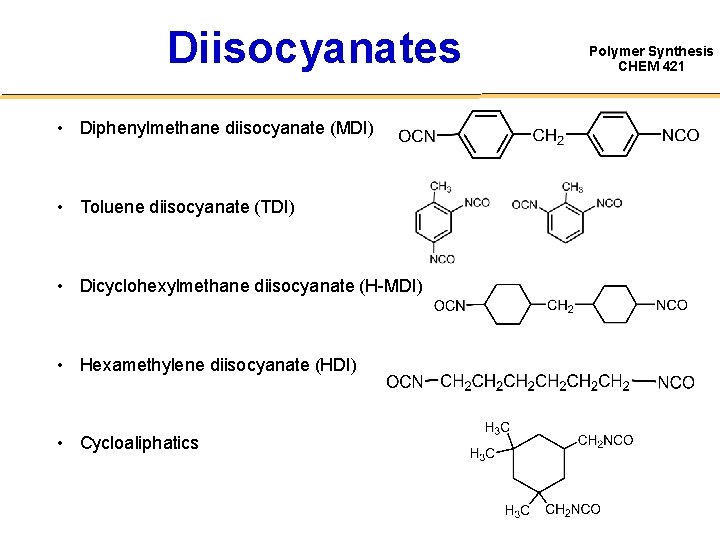
Diisocyanates • Diphenylmethane diisocyanate (MDI) • Toluene diisocyanate (TDI) • Dicyclohexylmethane diisocyanate (H-MDI) • Hexamethylene diisocyanate (HDI) • Cycloaliphatics Polymer Synthesis CHEM 421
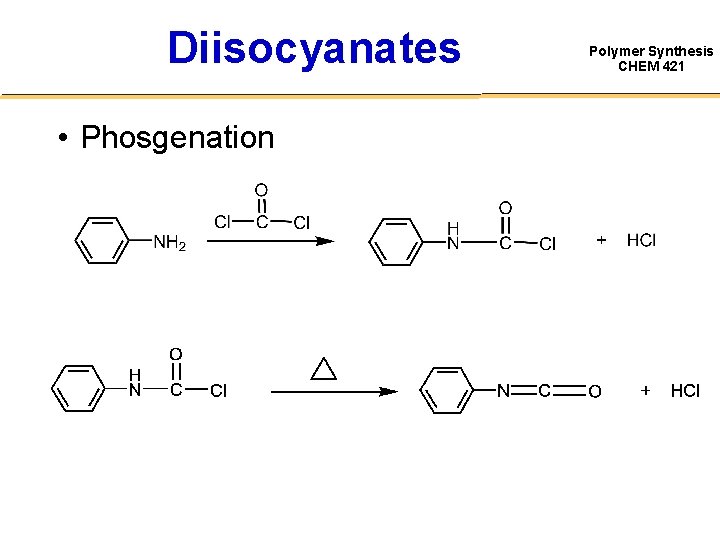
Diisocyanates • Phosgenation Polymer Synthesis CHEM 421
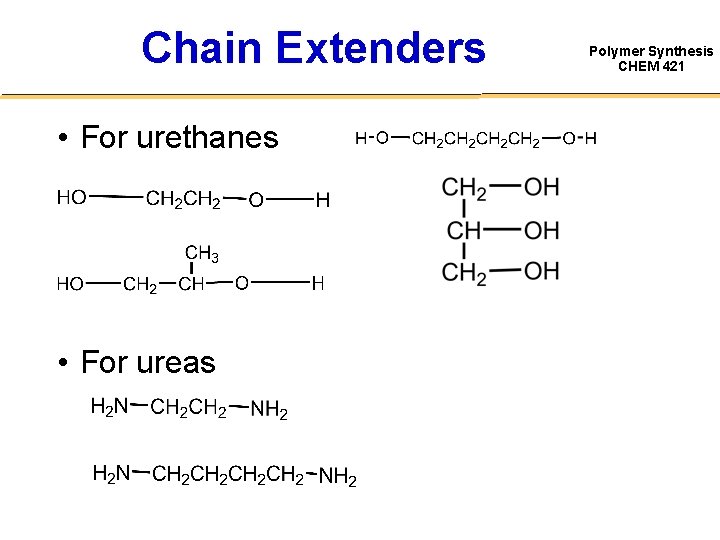
Chain Extenders • For urethanes • For ureas Polymer Synthesis CHEM 421
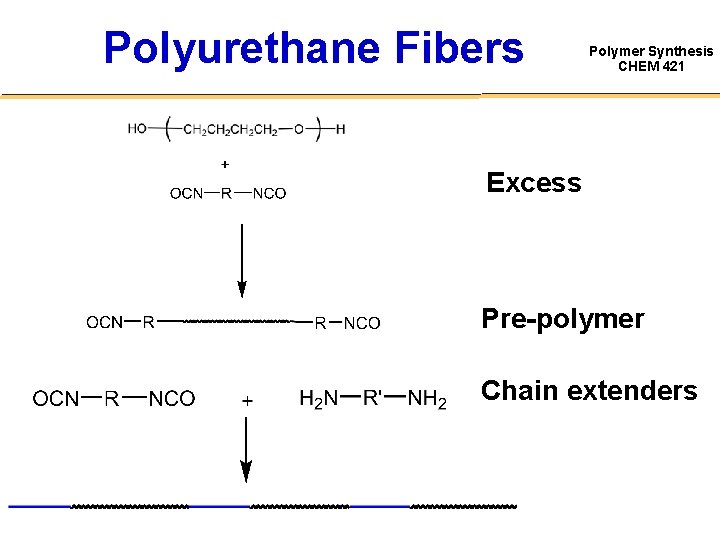
Polyurethane Fibers Polymer Synthesis CHEM 421 Excess Pre-polymer Chain extenders
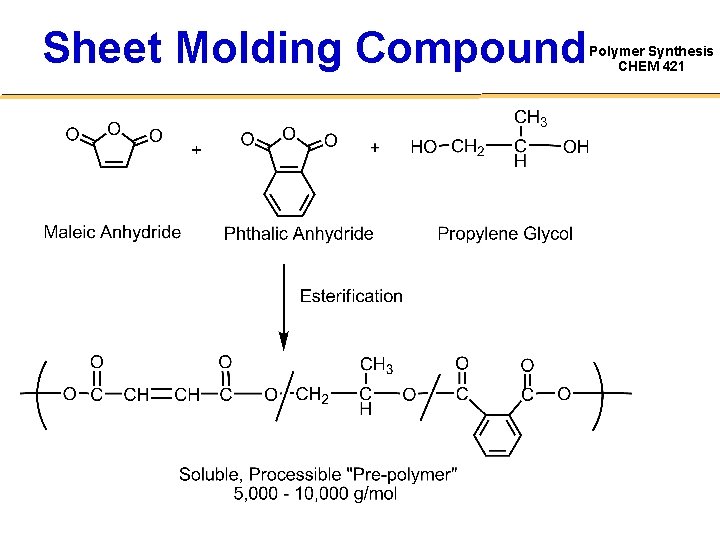
Sheet Molding Compound Polymer Synthesis CHEM 421

Sheet Molding Compound Polymer Synthesis CHEM 421

Phenol Formaldehyde Resins Polymer Synthesis CHEM 421 • 1872 – Invented by Bayer • 1907 – First patent and commercial process by Baekeland • Success: First wholly synthetic polymer used commercially

Phenol Formaldehyde Resins • Excellent thermal stability • High char yield • Low smoke generation • Low smoke toxicity Polymer Synthesis CHEM 421
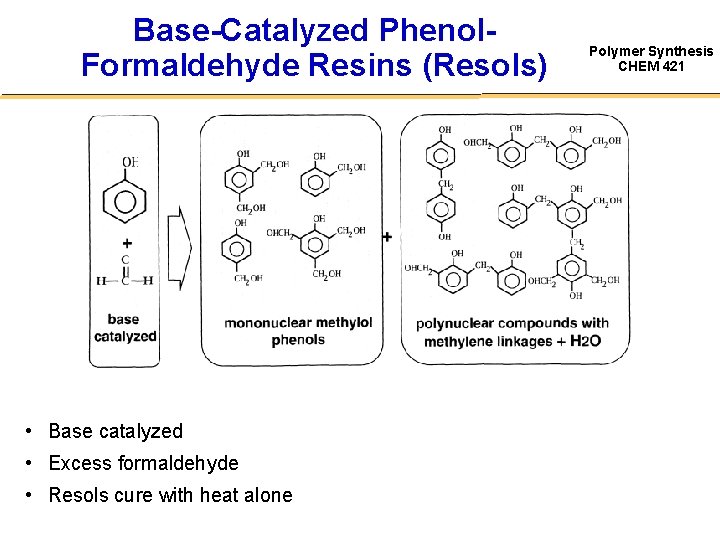
Base-Catalyzed Phenol. Formaldehyde Resins (Resols) • Base catalyzed • Excess formaldehyde • Resols cure with heat alone Polymer Synthesis CHEM 421
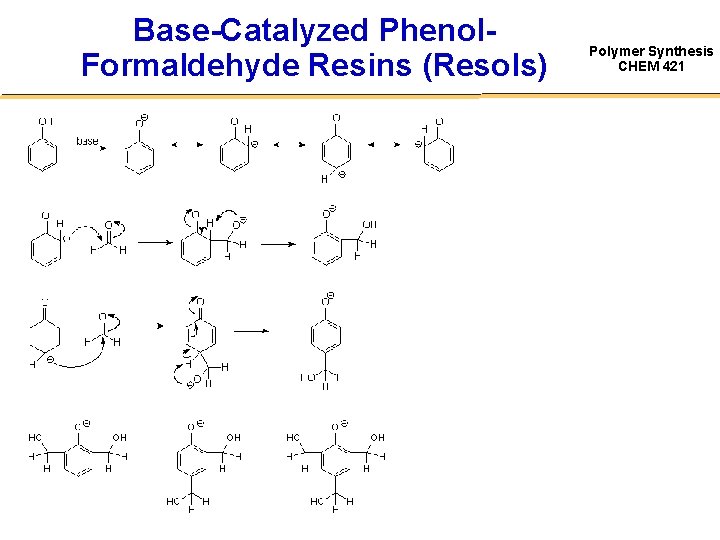
Base-Catalyzed Phenol. Formaldehyde Resins (Resols) Polymer Synthesis CHEM 421
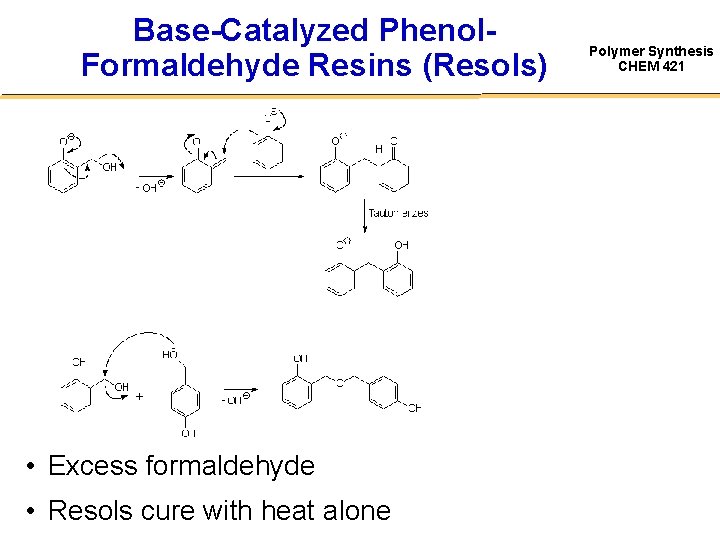
Base-Catalyzed Phenol. Formaldehyde Resins (Resols) • Excess formaldehyde • Resols cure with heat alone Polymer Synthesis CHEM 421
Polymer Synthesis CHEM 421
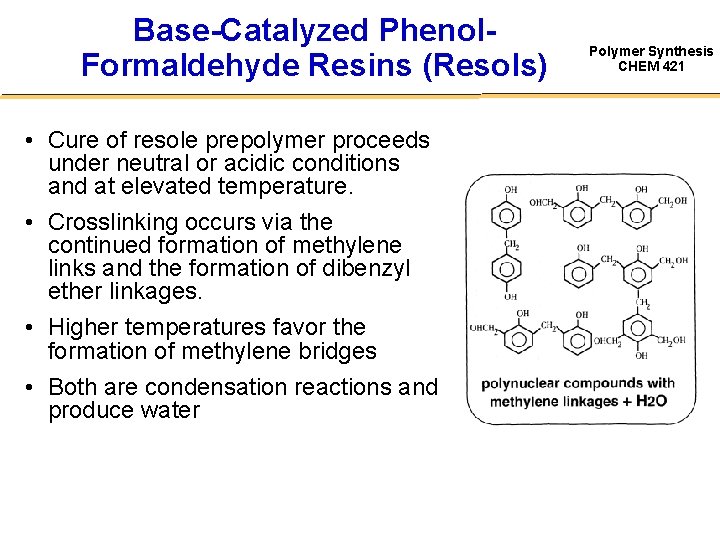
Base-Catalyzed Phenol. Formaldehyde Resins (Resols) • Cure of resole prepolymer proceeds under neutral or acidic conditions and at elevated temperature. • Crosslinking occurs via the continued formation of methylene links and the formation of dibenzyl ether linkages. • Higher temperatures favor the formation of methylene bridges • Both are condensation reactions and produce water Polymer Synthesis CHEM 421
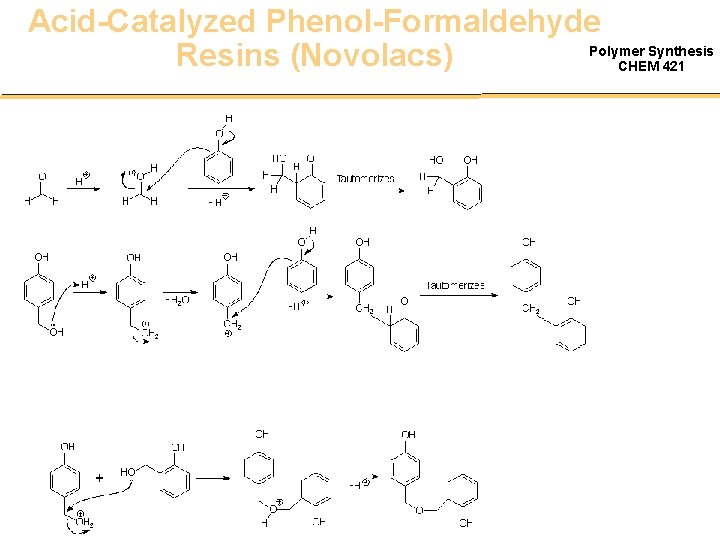
Acid-Catalyzed Phenol-Formaldehyde Polymer Synthesis Resins (Novolacs) CHEM 421
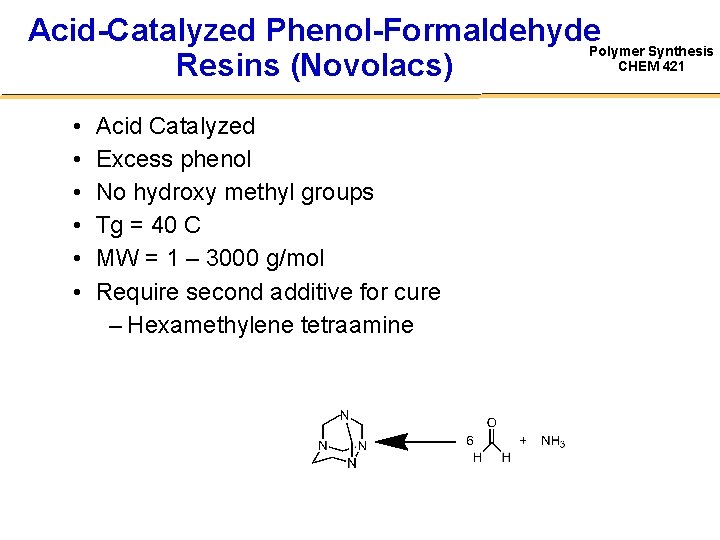
Acid-Catalyzed Phenol-Formaldehyde. Polymer Synthesis CHEM 421 Resins (Novolacs) • • • Acid Catalyzed Excess phenol No hydroxy methyl groups Tg = 40 C MW = 1 – 3000 g/mol Require second additive for cure – Hexamethylene tetraamine
- Slides: 25